Abstract
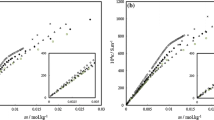
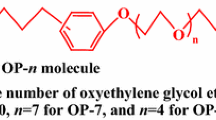
The thermodynamics of micellization of non-ionic surfactants (ethoxylated nonyl-phenols with different chain length) was studied as a function of ethoxy group number, electrolyte concentration and type, and concentration of short-chain alcohols at different temperature. On the basis of the thermodynamic data the enthalpy/entropy correlation and the ΔH/ΔS compensation temperature were calculated to characterize the solvent-solute and solute-solute interaction. The experimental results have definitely proved that a well-defined correlation exists for all investigated systems and the compensation temperature is independent of the ethoxy group number and the presence of alcohols. The inorganic electrolytes, however, decrease theT c compared to both alcohol-free and alcohol-containing systems, indicating the change in the mechanism of the micellization process. The new theoretical results significantly contribute to confirmation of the earlier conclusion concerning the stability and the structure of non-ionic surfactant solutions.
Similar content being viewed by others
References
Krishnan CV, Friedman HL (1973) J Solution Chem 2:37
Krishnan CV, Friedman HL (1973) J Solution Chem 2:119
Jolicoeur C, Philip PR (1974) Can J Chem 52:1834–1839
Bedö Zs, Berecz E, Lakatos I, Lakatos-Szabó J (1989) Acta Chim Hung 126:213–223
Bedö Zs, Berecz E, Lakatos I, Lakatos-Szabó J (1990) Progress in Colloid Polym Sci 82:229–235
Goto A, Takemoto M, Endo F (1985) Bull Chem Soc Jpn 58:247–251
Bedö Zs, Berecz E, Lakatos I (1986) Colloid Polym Sci 264:267–272
Lumry R, Rajender S (1970) Biopolymers 9:1125–1227
Bedö Zs, Berecz E, Lakatos I (1987) Colloid Polym Sci 265:715–722
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bedö, Z., Berecz, E. & Lakatos, I. Enthalpy-entropy compensation of micellization of ethoxylated nonyl-phenols. Colloid Polym Sci 270, 799–805 (1992). https://doi.org/10.1007/BF00776152
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00776152