Abstract
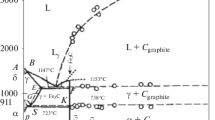
Carbon solubility and the composition of carbides in the Fe-Cr-C system up to 8.07 pct Cr were measured at 985 K by CH4/H2 gas equilibration. An iron-carbon binary alloy was included in the equilibration as a reference material. The chromium-carbon interaction inα-phase was analyzed by the central atoms model. The Wagner interaction coefficient was determined asε CrC = -72 ± 2, a significantly higher negative value than in austenitic alloy. The activity coefficient of carbon inα-Fe was determined as logϕ oC = 3.617, which supports the values reported by Swartz and by Chipman. The carbide phase was analyzed as a regular solution of two component carbides, FeC x and CrC x . M7C3 carbide was present in the carbon activity range up toa c = 0.8, while M3C carbide was present at higher carbon activities. Partitioning of chromium betweenα and the carbide phases was measured. The standard Gibbs energies of formation of the two component carbides and the interaction energy parameters were determined for both M7C3 and M3C carbides.
Similar content being viewed by others
References
K. Kuo:J. Iron Steel Inst., 1953, vol. 173, p. 363.
W. Jellinghaus and H. Keller:Arch. Eisenhüttenw., 1972, vol. 13, p. 319.
M. Small and E. Ryba:Metall. Trans. A, 1981, vol. 12A, p. 1389.
R.C. Sharma, G.R. Purdy, and J. S. Kirkaldy:Metall. Trans. A, 1979, vol. 10A, p. 1119.
M. Hillert and L.I. Staffansson:Acta Chem. Scand., 1970, vol. 24, p. 3618.
M. Waldenstrom and B. Uhrenius:Scand. J. Mat., 1977, vol. 6, p. 202; TRITA-MAC-0100, Oct. 1976.
R. Lundberg, M. Waldenstrom, and B. Uhrenius:CALPHAD, 1977, vol. 1, p. 159.
R. P. Smith:J. Am. Chem. Soc., 1946, vol. 68, p. 1163; Trans. TMS-AIME, 1962, vol. 224, p. 105.
J.C. Swartz:Trans. TMS-AIME, 1967, vol. 239, p. 68; 1969, vol. 245, p. 1083.
J.A. Lobo and G.H. Geiger:Metall. Trans. A, 1976, vol. 7A, p. 1347.
K. Narita, H. Hara, A. Miyamoto, and H. Iwakiri:Tetsu-to-Hagané, 1974, vol. 13, p. 1962.
I. Taguchi:Japan Analyst, 1973, vol. 22, p. 359.
“Atomic Absorption Spectrometric Analyses of Iron, Steel and Iron Ore”, Iron Steel Inst., Japan, 1974.
Dünwald and C. Wagner:Z. Anorg. Allgem. Chem., 1931, vol. 199, p. 321.
R.B. McLellan:Trans. TMS-AIME, 1965, vol. 233, p. 1664.
R.B. McLellan and W.W. Dunn:Metall. Trans., 1970, vol. 1, p. 535.
W.W. Dunn and R.B. McLellan:Metall. Trans., 1971, vol. 2, p. 1079.
E. Schumann, T. Schmidt, and F. Tillmann:Giesserei-Forschung, 1967, vol. 19, p. 35.
J. Chipman:Metall. Trans., 1972, vol. 3, p. 55.
E. H. Foo and C. H. P. Lupis:Acta Metall., 1973, vol. 21, p. 1409.
C.H. P. Lupis: “Chemical Thermodynamics of Materials”, North-Holland, Elsevier, NY, 1983.
M. Enomoto:Tetsu-to-Hagané, 1983, vol. 69, p. 1336.
M. Hillert, T. Wada, and H. Wada:J. Iron Steel Inst., 1967, vol. 205, p. 539.
L. Kaufman and H. Nesor:CALPHAD, 1978, vol. 2, p. 55, p. 81; p. 117, p. 295, p. 325; 1979, vol. 3, p. 45.
H. Kleykamp:Ber. Bunsenges. Phy. Chem., 1969, vol. 73, p. 354.
H. Tanaka, Y. Kishida, A. Yamaguchi, and J. Moriyama:J. Japan Inst. Metals, 1971, vol. 35, p. 523.
A.D. Kulkarni and W.L. Warrell:Metall. Trans., 1972, vol. 3, p. 2363.
V. I. Alekseev and L. A. Shvartsman:Fiz. Metal. Metalloved., Akad. Nauk SSSR, 1961, vol. 11, p. 545.
E. K. Storms: “The Refractory Carbides”, Academic Press, NY, 1967, p. 102.
T. Kunitake:Trans. Japan Inst. Metals, 1966, vol. 7, p. 253.
K. Kuo and A. Hultgren:Jernkont. Ann., 1951, vol. 135, p. 449.
F. Leiber, W. Kock, and E. Schumann:Arch. Eisenhüttenw., 1971, vol. 42, p. 106.
T. Sato and T. Nishizawa:J. Japan Inst. Metals, 1955, vol. 19, p. 385.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wada, H. Thermodynamics of the Fe-Cr-C system at 985 K. Metall Trans A 16, 1479–1490 (1985). https://doi.org/10.1007/BF02658680
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02658680