Abstract
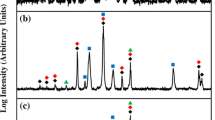
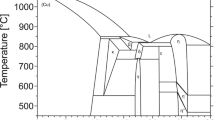
The thermodynamic activities of Ti at dilution in a series of Ag-Cu alloys and eutectic Ag-Cu alloys containing In or Sn were measured using a galvanic cell technique employing a ThO2-8 pct Y2O3 electrolyte. The equilibrium oxide phase formed by the reaction of Ti (XTi > 0.004) in the Ag-Cu alloy melts with an A12O3 or ZrO2 crucible was Ti2O (s). The free energy of formation of Ti2O (s) was estimated from available thermodynamic data. Titanium activities were calculated from measured oxygen potentials and the free energy of formation of Ti2O (s). Titanium in the eutectic Ag-Cu melt showed a positive deviation from ideal solution behavior at 1000°C, and its activity coefficient at infinite dilution was about 6.5 relative to pure solid Ti. Indium and Sn did not increase the activity coefficient of Ti in eutectic Ag-Cu melts. Silver increased the Ti activity coefficient in the Ag-Cu-Ti melts significantly. The Ti activity coefficient value in liquid Ag was about 20 times higher than in eutectic Ag-Cu melt at 1000 °C.
Similar content being viewed by others
References
G. Economo and W.D. Kingery:J. Am Ceram. Soc., 1953, vol. 36, pp. 403–09.
M. Humenik, Jr. and W.D. Kingery:J. Am. Ceram. Soc., 1954, vol. 37, pp. 18–23.
B.C. Allen and W.D. Kingery:Trans. AIME, 1959, vol. 215, pp. 30–36.
J.E. McDonald and J.G. Eberhart:Trans. AIME, 1965, vol. 233, pp. 512–17.
Yu.V. Naidich, V.S. Zhuravlev, V.G. Chuprina, and L.V. Strashinskaya:Sov. Powder Metall. Met. Ceram., 1973, vol. 12, pp. 895–99.
M.G. Nicholas, T.M. Valentine, and M. J. Waite:J. Mater. Sci., 1980, vol. 15, pp. 2197–2206.
A.J. Moorhead:Adv. Ceram. Mater., 1987, vol. 2, pp. 159–66.
A.J. Moorhead, H.M. Henson, and T.J. Henson: inCeramic Microstructures ’86, J.A. Pask and A.G. Evans, eds., Plenum Publishing Co., New York, NY, 1988, pp. 949–58.
M.G. Nicholas:Br. Ceram. Trans. J., 1986, vol. 85, pp. 144–46.
T.B. Massalski:Binary Alloy Phase Diagrams, ASM, Metals Park, OH, 1986, vols. 1 and 2.
A.R. Romero, J. Härkki, and D. Janke:Steel Res., 1986, vol. 57 (12), pp. 636–44.
H. Schmalzried:Z. Phys. Chem., 1963, vol. 38 (1-2), pp. 87–102.
D. Janke: inAdvances in Ceramics, Volume 12’, Science and Technology of Zirconia II, N. Claussen, M. Riihle, and A.H. Heuer, eds., The American Ceramic Society, Columbus, OH, 1984, pp. 636–45.
M.W. Chase, J.L. Curnutt, H. Prophet, and R.A. McDonald:J. Phys. Chem. Ref. Data, 1975, vol. 4 (1), pp. 137–47.
M.L. Santella, A.T. Fisher, and C.P. Haltom:J. Electron Microsc. Tech., 1988, vol. 8, pp. 211–15.
A.D. Mah, K.K. Kelley, N.L. Geliert, E.G. King, and C.J. O’Brien: Bur. Mines Rep. Invest. 5316, 1957.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pak, J.J., Santella, M.L. & Fruehan, R.J. Thermodynamics of Ti in Ag-Cu alloys. Metall Trans B 21, 349–355 (1990). https://doi.org/10.1007/BF02664203
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02664203