Abstract
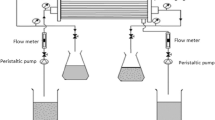
Separation of lanthanum and neodymium by supported liquid membrane has been studied. Synergistic extraction and recovery of lanthanum and neodymium with thenoyltrifluoroacetone (HTTA) in benzene have been found by the addition of Trioctylamine (TOA). Results indicate that percentage of extraction is highly dependent on pH of feed solution, which the maximum value is 2.5. When TOA was added to HTTA, the percentage of extraction and recovery considerably increased due to synergism. Lanthanum can be extracted and recovered more than neodymium because of the adduct formation constant,β 1 . Theβ 1 values decreased with an increase in atomic number of lanthanide and showed a difference between lanthanum and neodymium. Percentage of extraction and recovery is enhanced when the HTTA concentration is increased, but its difference is larger when TOA concentration is increased. Finally, multi-column module of supported hollow fiber membrane was used and the percentage and difference of extraction and recovery was found to be more increased due to resident time.
Similar content being viewed by others
References
Ching-Yet, S. and Pai-Zon, C., “Theoretical Analysis of Copper-ion Extraction through Hollow Fiber Supported Liquid Membranes”,Separation Science and Technology,28(13&14), 2149 (1993).
Cotton, F. A., “Advance Inorganic Chemistry”, 5th edition (1988).
Duyckaerts, G. and Desreux, J. F., “Recent Development and New Combinations of Extradant in Synergistic Process”, Proceeding of International Solvent Extraction Conference (1977).
Farbu, L., Alstad, J. and Auguston, J. H., 1974. cited in Duyckaerts, G. and Desreux, J. F., 1977, “Recent Development and New Combinations of Extradants in Synergistic Processes”, Proceeding of International Solvent Extraction Conference (1977).
Jeong, S. K. and Ju, C. S., “Extraction of Strontium Ion from Sea Water by Contained Liquid Membrane Permeator”,Korean J. Chem. Eng.,19(1), 93 (2002).
Loiacono, O., Drioli, E. and Molinari, R., “Metal Ions Separation and Concentration with Supported liquid Membranes”,Journal of Membrane Science,28, 123 (1986).
Mathur, J. N., “Synergism of Trivalent Actinides and Lanthanides Solvent Extraction and Ions Exchange”,Solvent Extraction and Ions Exchange,1(2), 349 (1983).
Naohiro, K. and Hisanori, I., “Synergistic Effect of Tris(4-isopropyl-tropolonato) Cocolt(III) on The Extraction of Lanthanoid(III) with 2-Thenoyltrifluoroacetone”,Analytical Science,17, 957 (2001).
Peppard, D. F., 1970. dted in Duyckaerts and Desreux, J. F., 1977, “Recent Development and New Combinations of Extradants in Synergistic Proesses”, Proceedings of International Solvent Extraction Conference (1977).
Porter Mark, C., “Handbook of Industrial Technology Membrane” New Jersey, Noyes publications (1990).
Ramakul, P., M.D.Thesis, Chulalongkorn University at Bangkok Thailand (2002).
Rovira, M. and Sastre, A. M., “Modeling of Mass Transfer in the Facilitated Supported Liquid Membrane Transport of Palladium(II) Using Di-2-ethylehexyl Thiophosphooric Acid”,Journal of Membrane Science,149, 241 (1988).
Schultz, G., “Separation Techniques with Supported Liquid Membrane”,Desalination,68, 191 (1988).
Yi, J. H., “Effect of the Boundary Layer and Interfacial Reaction on the Time Lag in Support Liquid Membranes”,Korean J. Chem. Eng.,12, 391 (1995).
Yang, X. J. and Fane, A. G., “Performance and Stability of Supported Liquid Membranes Using LIX 984N for Copper Transport”,Journal of Membrane Science,156, 251 (1999).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Prakorn, R., Ura, P. Synergistic extraction and separation of mixture of lanthanum and neodymium by hollow fiber supported liquid membrane. Korean J. Chem. Eng. 20, 724–730 (2003). https://doi.org/10.1007/BF02706915
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02706915