Abstract
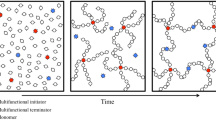
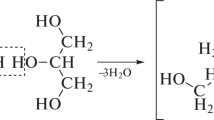
New molecular precursors to inorganic/organic hybrid network materials have been designed and synthesized. The molecules comprise multiple trialkoxysilane-terminated arms linked to an organic core. When hydrolyzed, these materials form network structures whose network elements are interconnected with flexible crosslinks. Clear, compliant glasses and thick films have been generated from a number of these precursors. They are significantly tougher and less brittle than purely inorganic glasses or conventional purely orgnaic thermosetting materials such as epoxy resins. The gels typically lose all surface-connected porosity during drying. Gelation rates for pure “star gel” precursors can be extremely high; the stars can also greatly enhance condensation rates for conventional sol-gel systems based on tetraalkoxysilanes.
Similar content being viewed by others
References
R.H. Glaser, G.L. Wilkes, and C.E. Bronnimann, J. Non Cryst. Solids113, 73–87 (1989).
W.G. Fahrenholtz, D.M. Smith, and D.W. Hua, J. Non-Cryst. Solids144, 45–52 (1992).
Y. Wei, R. Bakthavatchalam, and C.K. Whitecar, Chem. Mater.2, 337–339 (1990).
H. Schmidt, J. Non Cryst. Solids73, 681–691 (1985).
Y. Chujo, E. Ihara, S. Kure, and T. Saegusa, Macromolecules26, 5681–5686 (1993).
H.H. Huang, G.L. Wilkes, and J.G. Carlson, Polymer30, 2001–2012 (1989).
K.J. Shea, D.A. Loy, and O.W. Webster, Chem. Mater.1, 572–574 (1989).
K.J. Shea, D.A. Loy, and O.W. Webster, Polym. Mater. Sci. Eng.63, 281–285 (1990).
D.A. Loy, K.J. Shea, and E.M. Russick, inMater. Res. Soc. Symp. Proc, edited by M. Hampden-Smith, W. Klemperer, and C. Brinker, 1992, vol. 271, pp. 699–704.
K.J. Shea, D.A. Loy, and O. Webster, J. Am. Chem. Soc.114, 6700–6710 (1992).
J.H. Small, K.J. Shea, and D.A. Loy, J. Non-Cryst. Solids160, 234–246 (1993).
H.W. Oviatt, Jr., K.J. Shea, and J.H. Small, Chem. Mater.5, 943–950 (1993).
M.J. Michalczyk and K.G. Sharp, US Patent 5,378,790, assigned to the DuPont Co., 1/5/95.
The Pt catalyst of choice was Pt((ViMe2Si)2O) [Karstedt’s catalyst]; concentrations of about 20 ppm by weight were found effective. Caution should be used since hydrosilylation done without solyent can be highly exothermic. Soluble catalyst residues were typically removed with activated charcoal.
W.J. Simonsick, inStructure-Property Relationships in Polymers, edited by M.W. Urban and C.D. Craver (American Chemical Society, Washington, D.C., 1993), vol. 236.
C.J. Brinker and G.W. Scherer,Sol-Gel Science (Academic Press, San Diego, CA, 1990).
K.G. Sharp, J. Sol-Gel Sci. Tech.2, 35 (1994).
r is the molar ratio of gelling agent (water or formic acid) to silane; for the stars the same ratio of gelling agent to OR group was used.
A.J. Kinloch and R.J. Young,Fracture Behavior of Polymers (Applied Science Publishers, Essex, England, 1983).
Several reports now exist of silica gel which shows no appreciable nitrogen adsorption at 77°K over several hours but does adsorb CO2 at higher temperatures.
T. Woignier and J. Phalippou, J. Non-cryst. Solids93, 17 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sharp, K.G., Michalczyk, M.J. Star gels: New hybrid network materials from polyfunctional single component precursors. J Sol-Gel Sci Technol 8, 541–546 (1997). https://doi.org/10.1007/BF02436896
Issue Date:
DOI: https://doi.org/10.1007/BF02436896