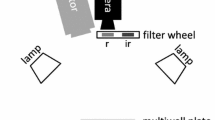
Abstract
Short-term variability in the photosynthetic activity of microphytobenthos assemblages was studied by measuring chlorophyll fluorescence rapid light curves (RLC), using pulse amplitude modulated (PAM) fluorometry. Measurements carried out on undisturbed samples under dark–light cycles revealed large diel oscillations in both the initial slope of the RLC (α) and in the maximum relative electron transport rate (ETRm). Short-term variations in RLC parameters were also observed, closely following changes in incident photon irradiance (E). Increases in irradiance were followed by decreases in α and increases in ETRm, resulting in significant correlations between the light-saturation parameter Ek and E. These results were interpreted as resulting from the onset of reversible energy-dissipating, non-photochemical quenching mechanisms and of compensatory high light-induced activation of carbon metabolism activity. Short-term RLC variability was shown to result mainly from physiological causes and to be detectable only by using short (10–20 s) light steps during RLC construction. Dark-adapted samples kept under constant conditions exhibited apparently endogenous rhythms in RLC parameters and in the maximum quantum yield, Fv/Fm, coincident with vertical migratory movements occurring during subjective photoperiods. These fluctuations appeared to result from the interaction between migratory rhythms and the physiological responses, and from the endogenous activation of processes affecting both the efficiency of energy transfer from light-harvesting antennae to the photosystem II (PSII) reaction centres or from non-radiative pathways (Fv/Fm, α) and the reactions downstream of PSII (ETRm).







Similar content being viewed by others
References
Barranguet C, Kromkamp J (2000) Estimating primary production rates from photosynthetic electron transport in estuarine microphytobenthos. Mar Ecol Prog Ser 204:39–52
Behrenfeld MJ, Kolber ZB (1999) Widespread iron limitation of phytoplankton in the South Pacific Ocean. Science 283:840–843
Behrenfeld MJ, Prasil O, Kolber ZS, Babin M, Falkowski PG (1998) Compensatory changes in photosystem II electron transport rate protect photosynthesis from photoinhibition. Photosynth Res 58:259–268
Behrenfeld MJ, Prasil O, Babin M, Bruyant F (2004) In search of a physiological basis for covariations in light-limited and light-saturated photosynthesis. J Phycol 40:4–25
Blanchard GF, Cariou-Le Gall V (1994) Photosynthetic characteristics of microphytobenthos in Marennes-Oléron Bay, France: preliminary results. J Exp Mar Biol Ecol 182:1–14
Blanchard GF, Montagna PA (1992) Photosynthetic response of natural assemblages of marine benthic microalgae to short- and long-term variations of incident irradiance in Baffin Bay, Texas. J Phycol 28:7–14
Dijkman NA, Kroon BMA (2002) Indications for chlororespiration in relation to light regime in the marine diatom Thalassiosira weissflogii. J Photochem Photobiol 66:179–187
Falkowski PG, Kolber Z (1995) Variations in chlorophyll fluorescence yields in phytoplankton in the world oceans. Aust J Plant Physiol 22:341–355
Falkowski PG, Greene R, Kolber Z (1994) Light utilization and photoinhibition of photosynthesis in marine phytoplankton. In: Baker NR, Bowes J (eds) Photoinhibition of photosynthesis: from molecular mechanisms to the field. Bios Scientific, Oxford, pp 407–432
Geider RJ, La Roche J, Greene RM, Olaizola M (1993) Response of the photosynthetic apparatus of Phaeodactylum tricornutum (Bacillariophyceae) to nitrate, phosphate, or iron starvation. J Phycol 29:755–766
Genty B, Briantais J-M, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990:87–92
Gévaert F, Créach A, Davoult D, Migné A, Levasseur G, Arzel P, Holl A-C, Lemoine Y (2003) Laminaria saccharina photosynthesis measured in situ: photoinhibition and xanthophyll cycle during a tidal cycle. Mar Ecol Prog Ser 247:43–50
Glud RN, Rysgaard S, Kühl M (2002) A laboratory study on O2 dynamics and photosynthesis in ice algal communities: quantification by microsensors, O2 exchange rates, 14C incubations and a PAM fluorometer. Aquat Microb Ecol 27:301–311
Green RM, Geider RJ, Falkowski PG (1991) Effect of iron limitation on photosynthesis in a marine diatom. Limnol Oceanogr 36:1772–1782
Guillard RRL, Ryther JH (1962) Studies of marine phytoplanktonic diatoms. I. Cyclotella nana Hustedt and Detonula confervaceae (Cleve) Gran. Can J Microbiol 8:229–239
Hartig P, Wolfstein K, Lippemeier S, Colijn F (1998) Photosynthetic activity of natural microphytobenthos populations measured by fluorescence (PAM) and 14C-tracer methods: a comparison. Mar Ecol Prog Ser 166:53–62
Jakob T, Goss R, Wilhem C (1999) Activation of diadinoxanthin de-epoxidase due to a chlororespiration proton gradient in the dark in the diatom Phaeodactylum tricornutum. Plant Biol 1:76–82
Kromkamp J, Barranguet C, Peene J (1998) Determination of microphytobenthos PSII quantum efficiency and photosynthetic activity by means of variable chlorophyll fluorescence. Mar Ecol Prog Ser 162:45–55
Kühl M, Glud RN, Borum J, Roberts R, Rysgaard S (2001) Photosynthetic performance of surface-associated algae below sea ice as measured with a pulse-amplitude-modulated (PAM) fluorometer and O2 microsensors. Mar Ecol Prog Ser 223:1–14
Lamontagne I, Cardinal A, Fortier L (1986) Intertidal microalgal production and the auxiliary energy of tides. Mar Biol 91:409–419
Lamontagne I, Cardinal A, Fortier L (1989) Environmental forcing versus endogenous control of photosynthesis in intertidal epilithic microalgae. Mar Ecol Prog Ser 51:177–187
Lavaud J, Rousseau B, van Gorkom H, Etienne A-L (2002) Influence of the diadinoxanthin pool size on photoprotection in the marine planktonic diatom Phaeodactylum tricornutum. Plant Physiol 129:1398–1406
Longstaff BJ, Kildea T, Runcie JW, Cheshire A, Dennison WC, Hurd C, Kana T, Raven JA, Larkum AWD (2002) An in situ study of photosynthetic oxygen exchange and electron transport rate in the marine macroalga Ulva lactuca (Chlorophyta). Photosynth Res 74:281–293
MacIntyre HL, Cullen JJ (1995) Fine-scale vertical resolution of chlorophyll and photosynthetic parameters in shallow-water benthos. Mar Ecol Prog Ser 122:227–237
MacIntyre HL, Kana TM, Geider RJ (2000) The effect of water motion on short-term rates of photosynthesis by marine phytoplankton. Trends Plant Sci 5:12–17
MacIntyre HL, Kana TM, Anning T, Geider RJ (2002) Photoacclimation of photosynthesis irradiance response curves and photosynthetic pigments in microalgae and cyanobacteria. J Phycol 38:17–38
Martino-Catt S, Ort DR (1992) Low-temperature interrupts circadian regulation of transcriptional activity in chilling-sensitive plants. Proc Natl Acad Sci USA 89:3731–3735
Meyer T, Hust M, Marquardt J, Krumbein WE, Rhiel E (2003) A methodological approach to investigate steady state fucoxanthin chlorophyll a/c binding protein mRNA levels in Wadden Sea sediments. Int Microbiol 6:33–39
Müller P, Li X-P, Niyogi K (2001) Non-photochemical quenching. A response to excess light energy. Plant Physiol 125:1558–1566
Owens TG (1986) Light-harvesting function in the diatom Phaeodactylum tricornutum. II. Distribution of excitation energy between the photosystems. Plant Physiol 80:739–746
Perkins RG, Oxborough K, Hanlon ARM, Underwood GJC, Baker NR (2002) Can fluorescence be used to estimate the rate of photosynthetic electron transport within microphytobenthic biofilms? Mar Ecol Prog Ser 228:47–56
Pichard SL, Campbell L, Kang JB, Tabita FR, Paul JH (1996) Regulation of ribulose bisphosphate carboxilase gene expression in natural phytoplankton communities. I. Diel rhythms. Mar Ecol Prog Ser 139:257–265
Pinckney J, Zingmark RG (1993) Photophysiological responses of intertidal benthic microalgal communities to in situ light environments: methodological considerations. Limnol Oceanogr 38:1373–1383
Platt T, Gallegos CL, Harrison WG (1980) Photoinhibition of photosynthesis in natural assemblages of marine phytoplankton. J Mar Res 38:687–701
Prèzelin BB (1992) Diel periodicity in phytoplankton productivity. Hydrobiologia 238:1–35
Quick WP, Orton P (1984) Studies on the induction of chlorophyll fluorescnce in barley protoplasts. 2. Resolution of fluorescence quenching by redox state and the transthylakoid pH gradient. Proc R Soc Lond B Biol Sci 220:371–382
Ralph PJ, Gademann R, Dennison WC (1998) In situ seagrass photosynthesis measured using a submersible, pulse-amplitude modulated fluorometer. Mar Biol 132:367–373
Ralph PJ, Gademann R, Larkum AWD, Schreiber U (1999) In situ underwater measurements of photosynthetic activity of coral zooxanthellae and other reef-dwelling dinoflagellate endosymbionts. Mar Ecol Prog Ser 180:139–147
Ralph PJ, Gademann R, Larkum AWD, Kühl M (2002a) Spatial heterogeneity in active chlorophyll fluorescence and PSII activity of coral tissues. Mar Biol 141:639–646
Ralph PJ, Polk SM, Moore KA, Orth RJ, Smith Jr WO (2002b) Operation of the xanthophyll cycle in the seagrass Zostera marina in response to variable irradiance. J Exp Mar Biol Ecol 271:189–207
Saburova MA, Polikarpov IG (2003) Diatom activity within soft sediments: behavioural and physiological processes. Mar Ecol Prog Ser 251:115–126
Schofield O, Evens TJ, Millie DF (1998) Photosystem II quantum yield and xanthophyll-cycle pigments of the macroalga Sargassum natans (Phaeophyceae): responses under natural sunlight. J Phycol 34:104–112
Schreiber U, Schliwa U, Bilger W (1986) Continuous recording of photochemical and nonphotochemical chlorophyll fluorescence quenching with a new type of modulation fluorometer. Photosynth Res 10:51–62
Schreiber U, Gademann R, Ralph PJ, Larkum AWD (1997) Assessment of photosynthetic performance of Prochloron in Lissoclinum patella in hospite by chlorophyll fluorescence measurements. Plant Cell Physiol 38:945–951
Serôdio J (2003) A chlorophyll fluorescence index to estimate short-term rates of photosynthesis by intertidal microphytobenthos. J Phycol 39:33–46
Serôdio J (2004) Analysis of variable chlorophyll fluorescence in microphytobenthos assemblages: implications of the use of depth-integrated measurements. Aquat Microb Ecol 36:137–152
Serôdio J, Marques da Silva J, Catarino F (1997) Nondestructive tracing of migratory rhythms of intertidal microalgae using in vivo chlorophyll a fluorescence. J Phycol 33:542–553
Serôdio J, Marques da Silva J, Catarino F (2001) Use of in vivo chlorophyll a fluorescence to quantify short-term variations in the productive biomass of intertidal microphytobenthos. Mar Ecol Prog Ser 218:45–61
Silva J, Santos R (2003) Daily variation patterns of seagrass photosynthesis along a vertical gradient. Mar Ecol Prog Ser 257:37–44
Strasser B, Dau H, Heinze I, Senger H (1999) Comparison of light induced and cell cycle dependent changes in the photosynthetic apparatus: a fluorescence induction study on the green alga Scenedesmus obliquus. Photosynth Res 60:217–227
Ting CS, Owens TG (1993) Photochemical and nonphotochemical fluorescence quenching processes in the diatom Phaeodactylum tricornutum. Plant Physiol 101:1323–1330
Underwood GJC (2002) Adaptations of tropical marine microphytobenthic assemblages along a gradient of light and nutrient availability in Suva Lagoon, Fiji. Eur J Phycol 37:449–462
White AJ, Critchley C (1999) Rapid light curves: a new fluorescence method to assess the state of the photosynthetic apparatus. Photosynth Res 59:63–72
Acknowledgements
We thank J. Marques da Silva for discussion and critical comments on the manuscript. We also thank A. Calado and A. Tim-Tim for kindly providing unialgal cultures of C. closterium. This work was supported by project PDCTM/MAR/15318/99, funded by Fundação para a Ciência e a Tecnologia. We thank three anonymous reviewers for critical comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Kühl, Helsingør
Rights and permissions
About this article
Cite this article
Serôdio, J., Vieira, S., Cruz, S. et al. Short-term variability in the photosynthetic activity of microphytobenthos as detected by measuring rapid light curves using variable fluorescence. Marine Biology 146, 903–914 (2005). https://doi.org/10.1007/s00227-004-1504-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-004-1504-6