Abstract
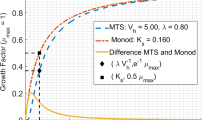
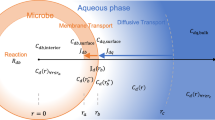
The Monod equation has been widely applied to describe microbial growth, but it has no mechanistic basis and is purely empirical. Extensive efforts have been dedicated to develop theoretical approaches for derivation of the Monod equation, which can be classified into three major groups, i.e., kinetic, thermodynamic, and substance transport approaches. In this review, four representative approaches are thus discussed. Due to the fact that different assumptions are made in each approach, no universal physical meaning of the Monod constant (K s) can be revealed. However, it seems that the Monod constant would be free energy-dependent and have nonequilibrium thermodynamic characteristics.

Similar content being viewed by others
References
Alexander M (1999) Biodegradation and bioremediation, 2nd edn. Academic, New York
Bailey JE, Ollis DF (1986) Biochemical engineering fundamentals. McGraw-Hill, Singapore
Bekins BA, Warren AE, Godsy EM (1998) A comparison of aero-order, first-order and Monod biotransformation models. Ground Water 36:261–268
Bitton G (1999) Wastewater microbiology. Wiley-Liss, New York
Blok J (2001) A single model for mass transfer and growth for biodegradation rates in activated sludge. Ecotoxicol Environ Saf 48:148–160
Boudart M (1976) Consistency between kinetics and thermodynamics. J Phys Chem 80:2869–2870
Brady JE, Russell JW, Holum JR (2000) Chemistry: matter and its changes. Wiley, Singapore
Button DK (1998) Nutrient uptake by microorganisms according to kinetic parameters from theory as related to cytoarchitecture. Microbiol Mol Biol Rev 62:636–645
Ferenci T (1999) Growth of bacterial cultures’ 50 years on: towards an uncertainty principle instead of constants in bacterial growth kinetics. Res Microbiol 150:431–438
Flickinger MC, Drew SW (1999) Encyclopedia of bioprocess technology: fermentation, biocatalysis and bioseparation. Wiley, New York
Gaudy AF, Gaudy ET (1980) Microbiology for environmental scientists and engineers. McGraw-Hill, New York
Grady CPL, Daigger GT, Lim HC (1999) Biological wastewater treatment. Marcel Dekker, New York
Heijnen JJ, Romein B (1995) Derivation of kinetic equation for growth on single substrates based on general properties of a simple metabolic network. Biotechnol Prog 11:712–716
Jin Q, Bethke CM (2002) Kinetics of electron transfer through the respiratory chain. Biophys J 83:1797–1808
Jin Q, Bethke CM (2003) A new rate law describing microbial respiration. Appl Environ Microbiol 69:2340–2348
Kovarova-Kovar K, Egli T (1998) Growth kinetics of suspended microbial cells: from single-substrate-controlled growth to mixed-substrate kinetics. Microbiol Mol Biol Rev 62:646–666
Merchuk JC, Asenjo JA (1995) The Monod equation and mass transfer. Biotechnol Bioeng 45:91–94
Monod J (1949) The growth of bacterial cultures. Annu Rev Microbiol 3:371–394
Rittmann B, McCarty P (2001) Environmental biotechnology: principles and applications. McGraw Hill, New York
Roels JA (1983) Energetics and kinetics in biotechnology. Elsevier, New York
Russell JB (1993) Effect of amino acids on the heat production and growth efficiency of Streptococcus bovis: balance of anabolic and catabolic rates. Appl Environ Microbiol 59:1747–1751
Shulter ML, Kargi F (2002) Bioprocess engineering: basic concepts. Prentice Hall PTR, Englewood Cliffs
Tan Y, Wang ZX, Schneider RP, Marshall KC (1994) Modelxling microbial growth: a statistical thermodynamic approach. J Biotechnol 32:97–106
Westerhoff HV, Lolkema JS, Otto R, Hellingwerf KJ (1982) Thermodynamics of growth. Non-equilibrium thermodynamics of bacterial growth. The phenomenological and the mosaic approach. Biochim Biophys Acta 683:181–220
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, Y. Overview of some theoretical approaches for derivation of the Monod equation. Appl Microbiol Biotechnol 73, 1241–1250 (2007). https://doi.org/10.1007/s00253-006-0717-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-006-0717-7