Abstract
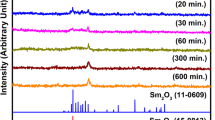
SmAlO3 nanocrystalline powders were successfully synthesized by the polymeric precursor method using ethylenediaminetetraacetic acid as a chelating agent. The precursor and the derived powders were characterized by thermogravimetry analysis (TG) and differential scanning calorimetry analysis (DSC), infrared spectroscopy (IR), X-ray diffractometry (XRD) and transmission electron microscopy (TEM). The results showed that pure SmAlO3 powder with orthorhombic perovskite structure could be synthesized at 800°C for 2 h without formation of any intermediate phase. The average particle size of the powder synthesized at 900°C was as low as 28 nm. Subsequently, the bulk SmAlO3 ceramics were prepared at various sintering temperatures using the synthesized powders calcined at 900°C for 2 h as starting materials. The sintering experiments indicated that the sample sintered at 1550°C for 2 h exhibited the highest relative density of 97.2% and possessed the best microwave dielectric properties of ε r=20.94, Q×f=78600 GHz and τ f=−71.8 ppm/°C.
Similar content being viewed by others
References
H. Takahashi, Y. Baba, K. Ezaki, Jpn. J. Appl. Phys. 30, 2339–2342 (1991)
M. Fu, X. Liu, X. Chen, J. Eur. Ceram. Soc. 28, 585–590 (2008)
H. Matsumoto, H. Tamura, K. Wakino, J. Appl. Phys. 30, 2347–2349 (1991)
L. Khalam, M. Sebastian, J. Am. Ceram. Soc. 90, 1467–1474 (2007)
S. Behera, P. Sahu, S. Pratihar, S. Bhattacharyya, Mater. Lett. 58, 3710–3715 (2004)
L. Vasylechko, A. Senyshyn, U. Bismayer, Handbook on the Physics and Chemistry of Rare Earths (Elsevier, Amsterdam, 2009)
C. Huang, Y. Chen, Mater. Res. Bull. 37, 563–574 (2002)
S. Cho, I. Kim, K. Hong, J. Mater. Res. 14, 114–119 (1999)
D. Suvorov, M. Valant, B. Jancar, S. Skapin, Acta Chim. Slov. 48, 87–99 (2001)
B. Jancar, D. Suvorov, M. Valant, Key Eng. Mater. 206–212, 1289–1292 (2002)
K. Vidyasagar, J. Gopalkrishnan, C. Rao, J. Solid State Chem. 58, 29–37 (1985)
P. Padmini, T. Kutty, J. Mater. Chem. 4, 1875–1882 (1994)
M. Kumar, T. Srinivasan, P. Ramasamy, C. Subramanian, Mater. Lett. 25, 171–174 (1995)
A. Douy, M. Capron, J. Eur. Ceram. Soc. 23, 2075–2080 (2003)
A. Ries, A. Simoes, M. Cilense, M. Zaghete, J. Varela, Mater. Charact. 50, 217–221 (2003)
Z. Tian, H. Yu, Z. Wang, Mater. Chem. Phys. 106, 126–129 (2007)
P. Rodrigues, O. Dantas, I. Santos, A. Souza, J. Alloys Compd. 397, 255–259 (2005)
G. Huang, D. Zhou, J. Xu, Z. Zheng, Mater. Res. Bull. 40, 13–19 (2005)
D. Petrov, B. Angelov, V. Lovchinov, J. Rare Earths 28, 602–605 (2010)
Y. Xu, G. Huang, Y. He, Ceram. Int. 31, 21–25 (2005)
H. Wang, M. Chung, Mater. Chem. Phys. 77, 853–859 (2003)
Y. Xu, X. Yuan, P. Lu, G. Huang, C. Zeng, Ceram. Int. 32, 57–60 (2006)
B. Hakki, P. Coleman, IRE Trans. Microwave Theor. Tech. MTT-8, 402–410 (1960)
W. Courtney, IEEE Trans. Microw. Theory Tech. MTT-18, 476–485 (1970)
M. Rajendran, M. Rao, J. Solid State Chem. 113, 239–247 (1994)
M. Couzi, P. Huong, J. Chem. Phys. Phys.-Chim. Biol. 69, 1339 (1972)
P. Lessing, Am. Ceram. Soc. Bull. 68, 1002–1006 (1989)
H. Lu, L. Burkhart, G. Schrader, J. Am. Ceram. Soc. 74, 968–972 (1991)
J. Choy, Y. Han, J. Sohn, M. Itoh, J. Am. Ceram. Soc. 78, 1169–1172 (1995)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, J., Qiu, T. Synthesis of SmAlO3 nanocrystalline powders by polymeric precursor method. Appl. Phys. A 104, 465–469 (2011). https://doi.org/10.1007/s00339-011-6263-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00339-011-6263-x