Abstract
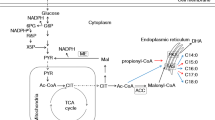
Docosahexaenoic acid (DHA) production in Schizochytrium sp. HX-308 was evaluated by detecting enzymatic activities of ATP:citrate lyase (EC 4.1.3.8), malic enzyme (EC 1.1.1.40) and glucose-6-phosphate dehydrogenase (EC 1.1.1.49) at different fermentation stages. According to the analysis, a regulation strategy was proposed which reinforced acetyl-CoA and NADPH supply at a specific fermentation stage. DHA content of total fatty acids was increased from 35 to 60% by the addition of 4 g/L malic acid at the rapid lipid accumulation stage. Total lipid content also showed an apparent increase of 35% and reached 19 g/L when 40 mL ethanol/L was added at the late lipid accumulation stage.






Similar content being viewed by others
References
Agostoni C, Riva E, Trojan S, Bellu R, Giovannini M (1995) Docosahexaenoic acid status and development quotient of healthy term infants. Lancet 346(8975):638
Lloyd AH, Young KY (1999) Health benefits of docohexaenoic acid (DHA). Pharmacol Res 40(3):211–225
Sijtsma L, Swaaf ME (2004) Biotechnological production and applications of the ω-3 polyunsaturated fatty acid docosahexaenoic acid. Appl Microbiol Biotechnol 64:146–153
Yokochi T, Honda D, Higashihara T et al (1998) Optimization of docosahexaenoic acid production by Schizochytrium limacinum SR21. Appl Microbiol Biotechnol 29:72–76
Barclay W, Weaver C, Metz J (2005) Development of a docosahexaenoic acid production technology using Schizochytrium: a historical perspective. In: Cohen Z, Ratledge C (eds) Single cell oils. AOCS Press, Champaign
Harrington GW, Holz GG Jr (1968) The monoenoic and docosahexaenoic fatty acids of a heterotrophic dinoflagellate. Biochim Biophys Acta 164:137–139
David HB, Glenn WH, Joannel G et al (1974) Biosynthesis of oleic acid and docosahexaenoic acid by a heterotrophic marine dinoflagellate Crypthcodinium cohnii. Biochim Biophys 369:16–24
Yaguchi T, Tanaka S, Yokochi T et al (1997) Production of high yields of docosahexaenoic acid by Schizochytrium sp strain SR21. J Am Oil Chem Soc 74:1431–1434
Ganuza E, Izquierdo MS (2007) Lipid accumulation in Schizochytrium G13/2S produced in continuous culture. Appl Microbiol Biotechnol 270:985–990
Ganuza E, Anderson AJ, Raledge C (2008) High-cell-density cultivation of Schizochytrium sp. in an ammonium/pH-auxostat fed-batch system. Biotechnol Lett 30(9):1559–1564
Ratledge C, Wynn JP (2002) The biochemistry and molecular biology of lipid accumulation in oleaginous microorganisms. Adv Appl Microbiol 51:1–51
Ratledge C (2004) Fatty acid biosynthesis in microorganisms being used for single cell oil production. Biochimie 86:807–815
Wynn JP, Ratledge C (1997) Malic enzyme is a major source of NADPH for lipid accumulation by Aspergillus nidulans. Microbiology 143:53–257
Randolph TW (1989) Malic enzyme of higher plants characteristics, regulation, and physiological function. Plant Physiol 90:367–371
de Swaaf ME, de Rijk TC et al (1999) Optimisation of docosahexaenoic acid production in batch cultivations by Crypthecodimium cohnii. J Biotechnol 70:185–192
de Swaaf ME, Sijtsma L, Pronk JT (2003) High-cell-density fedbatch cultivation of the docosahexaenoic-acid producing marine alga Crypthecodinium cohnii. Biotechnol Bioeng 81:666–672
de Swaaf ME, Pronk JT, Sijtsma L (2003) Fed-batch cultivation of the docosahexaenoic acid producing marine alga Crypthecodinium cohnii on ethanol. Appl Microbiol Biotechnol 61:40–43
Indarti E, Majid MIA, Hashim R, Chong A (2005) Direct FAME synthesis for rapid total lipid analysis from fish oil and cod liver oil. J Food Compost Anal 18:161–170
Rodrı guez-Ruiz J, Belarbi EH, Sa nchez JLG et al (1998) Rapid simultaneous lipid extraction and transesterification for fatty acid analyses. Biotechnol Technol 12:689–691
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Hsu RY, Lardy HA (1969) Malic enzyme. Methods Enzymol 13:230–235
Langdon RC (1966) Glucose 6-phosphate dehydrogenases from erythrocytes. Methods Enzymol 9:126–131
Takeda Y, Suzuki F, Inoue H (1969) ATP citrate lyase (citrate-cleavage enzyme). Methods Enzymol 13:153–160
Sijtsma L, Anderson AJ, Ratledge C (2005) Alternative carbon sources for heterotrophic production of docosahexaenoic acid by the marine alga Crythecodinium cohnii. In: Cohen Z, Ratledge C (eds) Single cell oils. AOCS Press, Champaign
Acknowledgments
This work was financed by the National Natural Science Foundation of China (No. 20606018), the Ministry of Science and Technology of China (National Basic Research Program of China (No. 2007CB707805)) and Scientific Research Project for Post-graduate in Jiangsu Province of China (No. CX07s_032z). We also thank Prof. Walt Johnson for his language assistance with this paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ren, LJ., Huang, H., Xiao, AH. et al. Enhanced docosahexaenoic acid production by reinforcing acetyl-CoA and NADPH supply in Schizochytrium sp. HX-308. Bioprocess Biosyst Eng 32, 837–843 (2009). https://doi.org/10.1007/s00449-009-0310-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-009-0310-4