Abstract
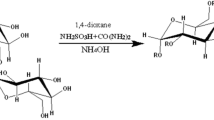
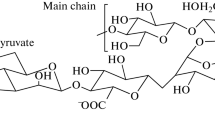
The synthesis of guar gum sulfates by a complex of sulfur trioxide with 1,4-dioxane was studied. The influence of temperature, process duration, and the volume of chlorosulfonic acid on the degree of substitution of guar gum sulfates was studied. The sulfation process has been optimized using the Box-Behnken design. It was shown that the optimal conditions for sulfation of guar gum with a complex of sulfur trioxide-1.4-dioxane: temperature 60 °C, duration 2.9 h, and a volume of chlorosulfonic acid of 3.1 ml. Sulfate groups embedding into the structure of guar gum was confirmed by elemental analysis and FTIR. The initial and sulfated guar gum were also characterized by methods: X-ray diffraction, scanning electron microscopy, and gel permeation chromatography. Using X-ray diffraction, it was shown that amorphization of guar gum occurs during sulfation. Using scanning electron microscopy, it was shown that the morphology of guar gum changes in the process of sulfation. Using gel permeation chromatography, it was shown in the process of guar gum sulfation by a complex of sulfur trioxide with 1,4-dioxane, the molecular weight decreases from 600 to 176 kDa. The geometric parameters of all complexes were carried out by using the DFT/B3PW91 method with a 6-31 + G (d,p) basis set. These structures are optimized to predict the important properties of a theme. MEP with contour map has been performed to obtain the electronic properties. Frontier molecular orbital HOMO-LUMO orbital diagram has been obtained for different energy levels and their band gap energies have been computed.











Similar content being viewed by others
References
Zhao X, Zhou H, Sikarwar VS, Zhao M, Park A-HA, Fennell PS, Shen L, Fan L-S (2017). Energy Environ Sci 10(9):1885–1910
Medvedeva EN, Babkin VA, Ostrouhova LA (2003). Chem Raw Plant Mater 1:27–37
Dushkin AV, Meteleva ES, Tolstikova TG, Pavlova AV, Khvostov MV (2013). Pharm Chem J 46(10):630–633
Cerqueira MA, Bourbon AI, Pinheiro AC, Martins JT, Souza BWS, Teixeira JA, Vicenta AA (2011). Trends Food Sci Technol 22:662–671
Prajapati VD, Jani GK, Moradiya NG, Randeria NP, Nagar BJ, Naikwadi NN, Variya BC (2013). Int J Biol Macromol 60:83–92
Filatova AV, Azimova LB, Turaev AS (2020). Chem Plant Raw Mater 1:33–39
Mercier T, Guldentops E, Lagrou K, Maertens J (2018). Front Microbiol 9:661
Perera N, Yang FL, Chang CM, Lu YT, Zhan SH, Tsai YT, Hsieh JF, Li LH, Hua KF, Wu SH (2017). Org Lett 19(13):3486–3489
Tolstenkov AS, Drozd NN, Lapikova EU, Makarov VA, Mestechkina NM, Bannikova GE, Ilyina AB, Varlamov VP (2007). Clinical Hematol Hemorheol in Cardiovasc Surgery 7:242–243 (in Rus)
Mestechkina NM, Anulov OV, Scherbukhin VD (1998). Appl Biochem Microbiol (Rus) 34(5):549–552
Mestechkina NM, Dovletmuradov K, Scherbukhin VD (1991). Appl Biochem Microbiol (Rus) 27(3):435–441
Krishtanova NA, Safonova MJ, Bolotova VTS (2005). Proc Voronezh St Univ Ser: Chem Biol Pharm 1:212–221 (in Russ)
Mudgil D, Barak S, Khatkar BS (2014). J Food Sci Technol 51:409–418
Yamamoto Y (2001). J Japan Soc Nutr Food Sci 54:139–145
Zhang LM, Zhou JF, Hui PS (2005). J Sci Food Agric 85:2638–2644
Hasan AMA, Abdel-Raouf ME (2018). Egypt J Pet 27:1043–1050
Jano A, Lame A, Kokalari E (2012). Kemija u industriji (J Chem Chem Eng) 61(11–12):497–503
About S, Zouarhi M, Chebabe D, Damej M, Berisha A, Hajjaji N (2020). Heliyon 6(3):e03574
Mothé CG, Correia DZ, de França FP, Riga AT (2006). J Therm Anal Calorim 85:31–36
Adewole JK, Muritala KB (2019). J Petrol Explor Prod Technol 9(3):2297–2307
Mohayeji M, Farsi M, Rahimpour MR, Shariati A (2016). J Taiwan Inst Chem Eng 60:76–82
Caputo HE, Strau JE, Grinstaff MW (2019). Chem Soc Rev 48:2338–2365
Wang X, Wang J, Zhang J, Zhao B, Yao J, Wang Y (2009). Int J Biol Macromol 46(1):59–66
Mestechkina NM, Egorov AV, Shcherbukhin VD (2006). Appl Biochem Microbiol 4:326
Wang J, Zhao B, Wang X, Yao J, Zhang J (2012). Int J Biol Macromol 50(5):1201–1206
Al-Horani RA, Desai UR (2010). Tetrahedron 66(16):2907–2918
Kazachenko AS, NYu V, Sudakova IG, Levdansky VA, Lutoshkin MA, Kuznetsov BN (2020). J Sib Fed Univ Chem 13(2):232–246
Pen RZ (2014) Planning an experiment at Statgraphics Centurion. Krasnoyarsk: SibSTU 293
Becke AD (1988). Phys Rev A 38:3098–3100
Perdew JP, Burke K, Wang Y (1996). Phys Rev B 54:16533–16539
Young D (2001) Computational chemistry: a practical guide for applying techniques to real world problems, vol 408. Wiley, New York
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, et al (2010) Gaussian, Inc., Wallingford
Dennington R, Keith T, Millam J (2010) Gauss View, Version 5. Semichem Inc., Shawnee Mission
Parr RG, Pearson RG (1983). J Am Chem Soc 105:7512–7516
Noureddine O, Gatfaoui S, Brandan SA, Marouani H, Issaoui N (2020). J Mol Struct 1202:127351
Gatfaoui S, Issaoui N, Roisnel T, Marouani H (2019). J Mol Struct 1191:183–196
Mudgil D, Barak S, Khatkar BS (2012). J Biol Macromol 50(4):1035–1039
Wang Q, Ellisa PR, Ross-Murphy SB (2003). Carbohyd Polym 53:75–83
Sagaama A, Noureddine O, Brandan SA, Jedryka AJ, Flakus HT, Ghalla H, Issaoui N (2020). Comput Biol Chem 87:107311
Issa TB, Sagaama A, Issaoui N (2020). Comput Biol Chem 86:107268
Gatfaoui S, Sagaama A, Issaoui N, Roisnel T, Marouani H (2020). Solid State Sci:106326
Noureddine O, Gatfaoui S, Brandan SA, Sagaama A, Marouani H, Issaoui N (2020). J Mol Struct 1207:127762
Jomaa I, Noureddine O, Gatfaoui S, Issaoui N, Roisnel T, Marouani H (2020). J Mol Struct 1213:128186
Noureddine O, Gatfaoui S, Brandán SA, Marouani H, Issaoui N (2020). J Mol Struct 1202:127351
Politzer P, Murray JS (2018). J Comput Chem 39:464–471
Politzer P, Murray JS (2020). ChemPhysChem 21:579–588
Politzer P, Murray JS, Clark T (2015). Top Curr Chem 358:19–42
Acknowledgments
The devices of the Krasnoyarsk Regional Center of Research Equipment of Federal Research Center “Krasnoyarsk Science Center SB RAS” were used in the work. The authors are grateful to G.N. Bondarenko for obtaining X-ray data, Korolkova I.V. for obtaining FTIR spectra and Antonov A.V. for obtaining SEM images. Besides, the authors thank Bitlis Eren University for supporting the Gaussian 09W software and Bingol University for the server.
Availability of data and material
N/A.
Funding
This work was supported by the Ministry of Higher Education and Scientific Research of Tunisia. The publication also supported by RFBR, project № 20-33-70256.
Author information
Authors and Affiliations
Contributions
Conceptualization: Aleksandr S. Kazachenko, Feride Akman, Abir Sagaama, Noureddine Issaoui; methodology: Aleksandr S. Kazachenko, Feride Akman, Noureddine Issaoui; formal analysis and investigation: Feride Akman, Abir Sagaama, Noureddine Issaoui, Yuriy N. Malyar, Valentina S. Borovkova, Aleksandr S. Kazachenko; writing—original draft preparation: Noureddine Issaoui, Feride Akman, Abir Sagaama, Aleksandr S. Kazachenko; writing—review and editing: Aleksandr S. Kazachenko, Feride Akman, Abir Sagaama, Noureddine Issaoui; supervision: Feride Akman, Abir Sagaama, Noureddine Issaoui, Natalya Yu. Vasilieva.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent for publication
N/A.
Ethics approval
N/A. In the course of work on this article, the authors did not conduct research on animals and humans in any form.
Consent to participate
N/A.
Code availability
N/A.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kazachenko, A.S., Akman, F., Sagaama, A. et al. Theoretical and experimental study of guar gum sulfation. J Mol Model 27, 5 (2021). https://doi.org/10.1007/s00894-020-04645-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-020-04645-5