Abstract
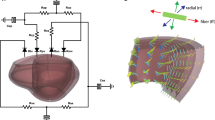
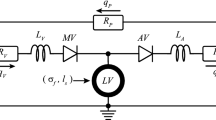
Ventricular growth is widely considered to be an important feature in the adverse progression of heart diseases, whereas reverse ventricular growth (or reverse remodeling) is often considered to be a favorable response to clinical intervention. In recent years, a number of theoretical models have been proposed to model the process of ventricular growth while little has been done to model its reverse. Based on the framework of volumetric strain-driven finite growth with a homeostatic equilibrium range for the elastic myofiber stretch, we propose here a reversible growth model capable of describing both ventricular growth and its reversal. We used this model to construct a semi-analytical solution based on an idealized cylindrical tube model, as well as numerical solutions based on a truncated ellipsoidal model and a human left ventricular model that was reconstructed from magnetic resonance images. We show that our model is able to predict key features in the end-diastolic pressure–volume relationship that were observed experimentally and clinically during ventricular growth and reverse growth. We also show that the residual stress fields generated as a result of differential growth in the cylindrical tube model are similar to those in other nonidentical models utilizing the same geometry.










Similar content being viewed by others
Notes
In this paper, we used the words “growth” and “remodeling” interchangeably although other authors have used “growth” and “remodeling” to specifically describe a change in mass and properties, respectively.
References
Ambardekar AV, Buttrick PM (2011) Reverse remodeling with left ventricular assist devices: a review of clinical, cellular, and molecular effects. Circ Hear Fail 4(2):33–224
Ambrosi D, Ateshian GA, Arruda EM, Cowin SC, Dumais J, Goriely A, Holzapfel GA, Humphrey JD, Kemkemer R, Kuhl E, Olberding JE, Taber LA, Garikipati K (2011) Perspectives on biological growth and remodeling. J Mech Phys Solid 59(4):863–883
Brinke EA, Klautz RJ, Tulner SA, Verwey HF, Bax JJ, Delgado V, Holman ER, Schalij MJ, van der Wall EE, Braun J, Versteegh MI, Dion RA, Steendijk P (2010) Clinical and functional effects of restrictive mitral annuloplasty at midterm follow-up in heart failure patients. Ann Thorac Surg 90(6):1913–1920
Burkhoff D, Klotz S, Mancini DM (2006) LVAD-induced reverse remodeling: basic and clinical implications for myocardial recovery. J Card Fail 12(3):227–239
Choi HF, D’hooge J, Rademakers FE, Claus P (2010) Influence of left-ventricular shape on passive filling properties and end-diastolic fiber stress and strain. J Biomech 43(9):1745–1753
Cohn JN, Ferrari R, Sharpe N (2000) Cardiac remodeling concepts and clinical implications: a consensus paper from an international forum on cardiac remodeling. J Am Coll Cardiol 35(3):569–582
Drakos SG, Terrovitis JV, Anastasiou-Nana MI, Nanas JN (2007) Reverse remodeling during long-term mechanical unloading of the left ventricle. J Mol Cell Cardiol 43(3):231–242
Frost HM (2003) Bone’s mechanostat: a 2003 update. Anat Rec A Discov Mol Cell Evol Biol 275(2):1081–1101
Fung Y, Liu S (1989) Change of residual strains in arteries due to hypertrophy caused by aortic constriction. Circ Res 65(5):1340–1349
Gaasch WH, Meyer TE (2008) Left ventricular response to mitral regurgitation: implications for management. Circulation 118(22):2298–303
Gerdes AM, Clark LC, Capasso JM (1995) Regression of cardiac hypertrophy after closing an aortocaval fistula in rats. Am J Physiol 268(6 Pt 2):51–H2345
Gerdes AM, Kellerman SE, Moore JA, Muffly KE, Clark LC, Reaves PY, Malec KB, McKeown PP, Schocken DD (1992) Structural remodeling of cardiac myocytes in patients with ischemic cardiomyopathy. Circulation 86(2):426–430
Göktepe S, Abilez OJ, Kuhl E (2010a) A generic approach towards finite growth with examples of athlete’s heart, cardiac dilation, and cardiac wall thickening. J Mech Phys Solids 58(10):1661–1680
Göktepe S, Abilez OJ, Parker KK, Kuhl E (2010b) A multiscale model for eccentric and concentric cardiac growth through sarcomerogenesis. J Theor Biol 265(3):42–433
Grossman W, Jones D, McLaurin LP (1975) Wall stress and patterns of hypertrophy in the human left ventricle. J Clin Invest 56(1):56–64
Guccione JM, McCulloch AD, Waldman LK (1991) Passive material properties of intact ventricular myocardium determined from a cylindrical model. J Biomech Eng 113(1):42–55
Kerckhoffs RCP (2012) Computational modeling of cardiac growth in the post-natal rat with a strain-based growth law. J Biomech 45(5):865–871
Kerckhoffs RCP, Omens J, McCulloch AD (2012) A single strain-based growth law predicts concentric and eccentric cardiac growth during pressure and volume overload. Mech Res Commun 42:40–50
Klepach D, Lee LC, Wenk JF, Ratcliffe MB, Zohdi TI, Navia Ja, Kassab GS, Kuhl E, Guccione JM (2012) Growth and remodeling of the left ventricle: a case study of myocardial infarction and surgical ventricular restoration. Mech Res Commun 42:134–141
Kroon W, Delhaas T, Arts T, Bovendeerd P (2009) Computational modeling of volumetric soft tissue growth: application to the cardiac left ventricle. Biomech Model Mechanobiol 8(4):301–309
Lee EH (1969) Elastic-plastic deformation at finite strains. J Appl Mech 36(1):1–6
Lee LC, Wall ST, Klepach D, Ge L, Zhang Z, Lee RJ, Hinson A, Gorman JH, Gorman RC, Guccione JM (2013) Algisyl-LVR with coronary artery bypass grafting reduces left ventricular wall stress and improves function in the failing human heart. Int J Cardiol 168(3):2022–2028
Legrice IJ, Hunter PJ, Smaill BH (1997) Laminar structure of the heart: a mathematical model. Am J Physiol 272(5 Pt 2):76–H2466
Levin H, Oz M, Chen J, Packer M, Rose E, Burkhoff D (1995) Reversal of chronic ventricular dilation in patients with end-stage cardiomyopathy by prolonged mechanical unloading. Circulation 91(11):2717–2720
Madigan JD, Barbone A, Choudhri AF, Morales DL, Cai B, Oz MC, Burkhoff D (2001) Time course of reverse remodeling of the left ventricle during support with a left ventricular assist device. J Thorac Cardiovasc Surg 121(5):902–908
Omens JH (1998) Stress and strain as regulators of myocardial growth. Prog Biophys Mol Biol 69(2–3):559–572
Omens JH, McCulloch AD, Criscione JC (2003) Complex distributions of residual stress and strain in the mouse left ventricle: experimental and theoretical models. Biomech Model Mechanobiol 1(4):267–277
Omens JH, Vaplon SM, Fazeli B, Mcculloch AD (1998) Left ventricular geometric remodeling and residual stress in the rat heart. J Biomech Eng 120:715–719
Opie LH, Commerford PJ, Gersh BJ, Pfeffer MA (2006) Controversies in cardiology. Lancet 367(9519):1315; author reply 1315–6
Rausch MK, Dam A, Göktepe S, Abilez OJ, Kuhl E (2011) Computational modeling of growth: systemic and pulmonary hypertension in the heart. Biomech Model Mechanobiol 10(6):799–811
Rodriguez EK, Hoger A, Mcculloch AD (1994) Stress-dependent finite growth in soft elastic tissues. J Biomech 27(4):455–467
Rutz AK, Ryf S, Plein S, Boesiger P, Kozerke S (2008) Accelerated whole-heart 3D CSPAMM for myocardial motion quantification. Magn Reson Med 59(4):755–763
Taber LA (1995) Biomechanics of growth, remodeling, and morphogenesis. Appl Mech Rev 48(8):487–545
Taber LA (2001) Biomechanics of cardiovascular development. Annu Rev Biomed Eng 3:1–25
Taber LA, Chabert S (2002) Theoretical and experimental study of growth and remodeling in the developing heart. Biomech Model Mechanobiol 1(1):29–43
Wall ST, Walker JC, Healy KE, Ratcliffe MB, Guccione JM (2006) Theoretical impact of the injection of material into the myocardium: a finite element model simulation. Circulation 114(24):2627–35
Weis SM, Emery JL, Becker KD, McBride DJ, Omens JH, McCulloch aD (2000) Myocardial mechanics and collagen structure in the osteogenesis imperfecta murine (oim). Circ Res 87(8):663–669
Wells SM, Walter EJ (2010) Changes in the mechanical properties and residual strain of elastic tissue in the developing fetal aorta. Ann Biomed Eng 38(2):345–356
Wenk JF, Eslami P, Zhang Z, Xu C, Kuhl E, Gorman JH, Robb JD, Ratcliffe MB, Gorman RC, Guccione JM (2011) A novel method for quantifying the in-vivo mechanical effect of material injected into a myocardial infarction. Ann Thorac Surg 92(3):935–941
Wenk JF, Klepach D, Lee LC, Zhang Z, Ge L, Tseng EE, Martin A, Kozerke S, Gorman JH, Gorman RC, Guccione JM (2012) First evidence of depressed contractility in the border zone of a human myocardial infarction. Ann Thorac Surg 93(4):1188–1193
Zhang X, Javan H, Ms LL, Ms AS, Bs RZ, Deng Y, Selzman CH (2013) A modified murine model for the study of reverse cardiac remodelling. Exp Clin Cardiol 18(2):115–117
Acknowledgments
This work was supported by NIH Grants R01-HL-077921 and R01-HL-118627 (J.M. Guccione); K25-NS058573-05 (G. Acevedo-Bolton); NSF Grants 0952021 and 1233054 (E. Kuhl); and Marie Curie international outgoing fellowship within the 7th European Community Framework Program (M. Genet). We thank the reviewers for their valuable comments, which have helped us improve the presentation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, L.C., Genet, M., Acevedo-Bolton, G. et al. A computational model that predicts reverse growth in response to mechanical unloading. Biomech Model Mechanobiol 14, 217–229 (2015). https://doi.org/10.1007/s10237-014-0598-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-014-0598-0