Abstract
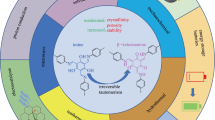
First, an intermediate ionic liquid [Bmim]Cl and Keggin-type heteropolyacids with different metal vacancies were prepared, and then prepared six heteropolyacid ionic liquids by the ion exchange method, and these liquids were tested and analyzed for chemical structures, crystal structures, thermal stability, and apparent morphology. After that, the six heteropolyacid ionic liquids were used to catalyze the transesterification reaction between phenol and dimethyl carbonate (DMC) to synthesize diphenyl carbonate (DPC), to explore the effects of reaction temperature, reaction time, and catalyst dosage on the catalytic performance, and reveal the catalytic mechanism. The results showed that the six heteropolyacid ionic liquids prepared were the target products with good thermal stability. Compared with traditional catalysts Cp2TiCl2 and C16H36O4Ti, they had better catalytic performance in the catalytic process. Among them, Bmim4[Ti(H2O)TiMo11O39] manifested the best catalytic performance. Under the conditions that the molar ratio of raw materials DMC to phenol was 2:1, reaction temperature 180 °C, catalyst consumption 1.5% of the total mass of the raw materials, and reaction time 8 h, the conversion rate of phenol reached 46.17%, and the overall selectivity of products monophenyl carbonate (MPC) and DPC was 98.89%. As a phase transfer catalyst characterized by a "homogeneous phase at high temperature and separation at low temperature," the catalyst can be self-assembled in the catalytic process with the temperature change. Therefore, they can be recycled and reused through simple washing and filtering at the end of the reaction. After five times of recycling, the catalytic performance of the catalyst reduces significantly, in which case the fresh catalyst should be added to maintain a good catalytic performance.
Graphical Abstract















Similar content being viewed by others
References
Pyo SH, Park JH, Chang TS (2017) Curr Opin Green Sust 5:61
Liu T, Hu J, Yong L (2019) J Anal Methods Chem 11:1
Baral ER, Lee JH, Kim JG (2018) J Org Chem 83:11768
Kim WB, Joshi UA, Lee JS (2004) Ind Eng Chem Res 43:1897
Fukuoka S, Fukawa I, Adachi T (2019) Org Process Res Dev 23:145
Mishra V, Sharma R, Mahajan K (2021) JoMM 4:61
Yilbas BS, Abubakar AA, Al-Qahtani H (2021) Polymers 13:1449
Figueiredo MC, Trieu V, Eiden S (2018) ACS Catal 8:3087
Zhao Y, Zhao X, Zhang M (2019) Mater Sci Eng R Rep 472:012055
Peng M, Hong C, Cai N (2020) Mol Catal 482:110684
Yang X, Bai H, Qian MM (2019) Mater Chem Phys 234:48
Qu Y, Yang H, Wang S (2017) Chem Res Chin Univ 33:804
Sun Y, Gao X, Yang N (2019) Ind Eng Chem Res 58:7937
Zhang Y, Wang S, Xiao Z (2016) Res Chem Intermediat 42:7213
Biying AO, Yuanting KT, Hosmane NS (2017) J Organomet Chem 849:195
Dibenedetto A, Angelini A, Bitonto L (2014) Chemsuschem 7:1155
Jia B, Cao P, Zhang H (2019) J Mater Sci 54:9466
Dibenedetto A, Angelini A, Dibitonto L (2014) Chemsuschem 7:1155
Li Z, Wang Y, Ding X (2009) J Nat Gas Chem 218:104
Weiqing Z, Xinqiang Z, Yanji W (2004) Appl Catal A-Gen 260:19
Wang S, Bai R, Mei F (2009) Catal Commun 11:202
Yang H, Xiao Z, Qu Y (2018) Res Chem Intermediat 44:99
Tang R, Chen T, Chen Y (2014) Chin J Catal 35:457
Wang Q, Li C, Guo M (2015) Inorg Chem Front 2:47
Niu H, Yao J, Wang Y (2005) J Mol Catal A-Chem 235:240
Dsta B, Jie YA, Yue WA (2007) J Mol Catal A-Chem 268:120
Wang C, Guo T, Sun M (2018) Sci Adv Mater 10:779
Xiao Z, Yang H, Zhang H (2018) Chem Pap 72:2347
Wang S, Li C, Xiao Z (2016) J Mol Catal A-Chem 420:26
Cao M, Meng Y, Lu Y (2005) Catal Commun 6:802
Wang S, Niu H, Wang J (2019) J Alloys Compd 777:18
Zhang J, Gao Y, Zhang J (2018) Chem Cent J 12:1
Qu Y, Wang S, Chen T (2017) Res Chem Intermediat 43:2725
Chen T, Han H, Yao J (2007) Catal Commun 8:1361
E YS, Wang Q, Zhao WZ (2020) Contemporary chemical industry 49:802
Yin G, Jia C, Kitamura T (2000) Catal Letters 69:89
Du Z, Kang W, Cheng T (2006) J Mol Catal A-Chem 246:200
Lee H, Kim SJ, Ahn BS (2003) Catal Today 87:139
Su KM, Li ZH, Ding M (2011) Adv Mat Res 233:124
Kore R, Kelley SP, Aduri P (2018) Dalton Trans 47:7795
Deshmukh KM, Qureshi ZS, Dhake KP (2010) Catal Commun 12:207
Ghasemi MH, Kowsari E, Shafiee A (2016) Tetrahedron Lett 57:150
Yang GP, Wu X, Yu B (2019) ACS Sustain Chem Eng 7:3727
Rajabi F, Wilhelm C, Thiel WR (2020) Green Chem 22:4438
Yang G, Li K, Zeng K (2021) RSC Adv 11:10610
Han X, Ouyang K, Xiong C (2017) Appl Catal A-Gen 543:115
Guo LY, Ran WT, Wang HY (2021) Preparation method and application of a titanium-based heteropolyacid ionic liquid catalyst, CN: 202111357048.4.
Guo LY, Deng LL, Jin XC (2018) Acta Petrolei Sin 34:71
Li J, Yang Z, Li S (2020) J Ind Eng Chem 82:1
Liu Y, Yao X, Liu X (2021) J Phys Chem C 125:25478
Shahabi M, Tabatabaee M, Keshavarz M (2019) Catal Commun 131:105748
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant Nos. 52103225, 51973051). Key project of Education Department of Liaoning Province (Grant No. LZGD2020005), and general project of Education Department of Liaoning Province (Grant No. LJKZ0164).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jiang, Z., Wang, H., Shan, L. et al. Preparation of Temperature-Controlled Heteropolyacid Ionic Liquids and Their Application for Synthesis of Diphenyl Carbonate. Catal Lett 153, 1308–1318 (2023). https://doi.org/10.1007/s10562-022-04068-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-022-04068-2