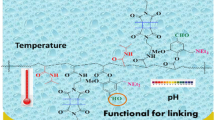
Abstract
The present work emphasizes the preparation of new monomer from the family of vanillin acrylate. It has been prepared in two steps the name of the final product is [2-((ditert-butylamino) methyl)-4-formyl-6-methoxyphenyl acrylate] and abbreviated by TBAMVA, both products were evaluated by 1H, 13C NMR and FTIR and they were in the logic case with the chemical structure. The next step was the preparation of three different pH and temperature responsive copolymers by free radical polymerization of 5, 10, 20 mol% of TBAMVA and 5 mol% of maleimide photo-cross-linker with N-isopropylacrylamide. Polymers were chemically and physically characterized indicating the presence of the aldehyde group in the main chain. The lower critical solution temperature of polymers has been determined by two methods; the first one using turbidity measurements by UV–Vis Spectroscopy, another method using micro-DSC. The last step was the formation of pH-Temperature responsive hydrogel layer over gold and studying the swelling properties response to pH and temperature using surface plasmon resonance with the optical waveguide. In the present work, we focused on synthesizing new environmental functional polymers and hydrogel thin films used as biosensor as a target for many biological molecules in biosynthesis, drug delivery, and bio-separation.




















Similar content being viewed by others
References
Fengjuan JA, Shuo C, Ziquan C, Guojie W (2016) Polymer 83:91. https://doi.org/10.1016/j.polymer.2015.12.027
Xiaojie Y, Jiawei L, Lingmin Y (2017) Mater Lett 208:49. https://doi.org/10.1016/j.matlet.2017.05.029
Jialing C, Zhaoyang D, Hongxia P, Xuejun C (2017) Talanta 174:256–264. https://doi.org/10.1016/j.talanta.2017.05.027
Chang K, Rubright N, Lowery PD, Taite LJ (2013) J Polym Sci A Polym Chem 51:2078. https://doi.org/10.1002/pola.26596
Heskins M, Guillet JE (1968) J Macromol Sci Chem A 2:1455. https://doi.org/10.1080/10601326808051910
Iatridi Z, Mattheolabakis G, Avgoustakis K, Tsitsilianis C (2011) Soft Matter 7:11168. https://doi.org/10.1039/C1SM06185B
Abdelaty MSA (2018) J Polym Environ 26:2256. https://doi.org/10.1007/s10924-017-1126-y
Abdelaty MSA (2018) J Polym Environ 26:646. https://doi.org/10.1007/s10924-017-0960-2
Young-Jin K, Yukiko TM (2017) J Mater Chem B 5:4321. https://doi.org/10.1039/C7TB00157F
Abdelaty MSA (2018) Polym Bull 11:4858. https://doi.org/10.1007/s00289-018-2297-y
Abdelaty MSA (2018) Biomolecules 8:154. https://doi.org/10.3390/biom8040138
Abdelaty MSA, Kuckling D (2016) Gels 2: 76. https://doi.org/10.3390/gels2010003
Andrés Díaz L (2017) Polymers 9:19
Renjith P, Johnsona S, Uthamanb R, Augustinea Yu, Zhanga, Hua Jina, Chang In Choic, Parkb I-K, Il Kima (2017) Reactive Functional Polymers 119: 47 https://doi.org/10.1016/j.reactfunctpolym.2017.07.010
Elvan Yilmaza Z, Yalincaa (2016) Kovan Yahyaa,b, Uliana Sirotinaa. Int J Biol Macromol 90:74. https://doi.org/10.1016/j.ijbiomac.2015.10.003
Soppimath KS, Tan DCW, Yang YY (2005) Adv Mater 17:323. https://doi.org/10.1002/adma.200401057
Delcea M, Möhwald H, Skirtach AG (2011) Adv Drug Deliv Rev 63:747. https://doi.org/10.1016/j.addr.2011.03.010
Uhlig K, Boysen B, Lankenau A, Jaeger M, Wischerhoff E, Lutz JF, Laschewsky A, Duschl C (2012) Biomicrofluidics 6:21. https://doi.org/10.1063/1.4729130
Sepehrifar R, Reinhard I, Boysen B, Danylec Y, Yang K, Milton S, Hearn TW (2017) Anal Chim Acta 963:163. https://doi.org/10.1016/j.aca.2017.01.061
Kanazawa H, Okano TJ (2011) Chromatogr A 1218:8747
Han X, Zhang X, Zhu H, Yin Q, Liu H, Hu Y (2013) Langmuir 29:1034
Xiong Z, Peng B, Han X, Peng C, Liu H, Hu Y (2011) J Colloid Interface Sci 356:565
Feng X, Wu H, Sui X, Hempenius MA, JuliusVancso G, Vancso (2015) Eur Polym J 72:542. https://doi.org/10.1016/j.eurpolymj.2015.05.022
Tokarev I, Minko S (2009) Soft Matter 5:524. https://doi.org/10.1039/B813827C
Yang HW, Chena JK, Cheng CC, Kuo SW (2013) Appl Surf Sci 271:69. https://doi.org/10.1016/j.apsusc.2013.01.074
Gauthier MA, Gibson MI, Klok HA (2009) Angew Chem Int Ed 48:58. https://doi.org/10.1002/anie.200801951
Fuchs AD, Tiller JC (2006) Angew Chem Int Ed 45:6762. https://doi.org/10.1002/anie.200602738
Zolotukhin MG, Colquhoun HM, Sestiaa LG, Rueda DR, Flot D (2003) Macromolecule 36:4771. https://doi.org/10.1021/ma0216977
Kanazawa H, Yamamoto K, Matsushima Y, Takai N, Kikuchi A, Sakurai Y, Okano T (1996) Anal Chem 68:105. https://doi.org/10.1021/ac950359j
Kuckling D, Harmon ME, Frank CW (2002) Macromolecules 35:6004. https://doi.org/10.1021/ma010985k
Harmon ME, Kuckling D, Frank CW (2003) Macromolecules 36:172. https://doi.org/10.1021/ma021025g CCC
Nan Zhang N, Knoll W (2009) Anal Chem 81: 2617. https://doi.org/10.1021/ac802527j
Aulasevich A, Junk MJN, Jakubowicz P, Roskamp RF, Menges B, Jonas U, Knoll W (2010) Macrmol Chem Phys 211:1025. https://doi.org/10.1002/macp.200900533
Chen JK, Chan CH, Chang FC (2008) Appl Phys Lett 92:053108. https://doi.org/10.1063/1.2939218
Chan CH, Chen JK, Chang FC (2008) Sens Actuators B 133:332https://doi.org/10.1016/j.snb.2008.02.041
Chen JK, Li JY (2010) Sens Actuatators B 150: 320. https://doi.org/10.1016/j.snb.2010.06.067
Hosoya K, Kubo T, Tanaka N, Haginaka JA (2003) J Pharm Biomed Anal 30: 1919. https://doi.org/10.1016/S0731-7085(02)00535-6
Lu Y, Mei Y, Drechsler M, Ballauff M (2006) Angew Chem Int Ed 45:816. https://doi.org/10.1002/anie.200502731
Chen JK, Wang JH, Chang JYF (2012) Appl Phys Lett 101:123701. https://doi.org/10.1063/1.4754135
Costa E, Coelho M, Ilharco LM, Aguiar-Ricardo A, Hammond PT (2011) Macromolecules 44:621. https://doi.org/10.1021/ma1025016
Acknowledgements
The authors are grateful acknowledge to Egyptian culture and missions, and The Deutscher Akademischer Austauch (DAAD) for financial assistance during the post doctor work in Germany of Momen S.A. Abdelaty.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Abdelaty, M.S.A. Layer by Layer Photo-Cross-Linked Environmental Functional hydrogel Thin Films Based on Vanillin: Part 3. J Polym Environ 27, 1212–1225 (2019). https://doi.org/10.1007/s10924-019-01421-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-019-01421-2