Abstract
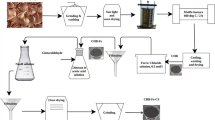
An aminated acrylic fiber waste has been utilized as an adsorbent material for the removal of Cd(II) ions from aqueous solution after treatment of acrylic fiber waste with hydroxylamine hydrochloride under basic conditions, and characterized for surface chemistry, surface morphology and textural properties. The ability of this sorbent to adsorb Cd(II) ions was examined via batch adsorption methods, studying the effect of pH, sorbent and sorbate concentrations, as well as contact time. Results obtained confirm that this sorbent was effective for Cd(II) ion adsorption, with uptakes promoted by high active site density, however, the adsorption process is independent of sorbent surface area. The values obtained exceed those previously reported within the literature. Isotherm analysis using arrange of two- and three- parameter models, evaluated using non-linear regression methods with error analysis, showed that the Langmuir isotherm model most appropriately described the experimental data obtained, indicating mono layer adsorption occurs. Kinetic analysis using arrange of models in their non-linear forms provided mechanistic information, showing that pseudo -second-order behavior is involved. The synthesized aminated acrylic fiber waste derived sorbents offer significant potential for the removal of Cd(II) ions from aqueous solution through a mechanism of chelation between the electron- donating oxygen-and nitrogen-containing groups in the sorbent and the electron-accepting Cd(II) ions.











Similar content being viewed by others
References
Meena AK, Kadirvelu K, Mishra GK, Rajagopal C, Nagar PN (2008) Adsorptive removal of heavy metals from aqueous solution by treated sawdust (Acacia arabica). J Hazard Mater 150(3):604–611
Moulick D, Santra SC, Ghosh D (2018) Effect of selenium induced seed priming on arsenic accumulation in rice plant and subsequent transmission in human food chain. Ecotoxicol Environ Saf 152:67–77
Meseguer VF, Ortuño JF, Aguilar MI, Pinzón-Bedoya ML, Lloréns M, Sáez J, Pérez-Marín AB (2016) Biosorption of cadmium (II) from aqueous solutions by natural and modified non-living leaves of Posidonia oceanica. Environ Sci Pollut Res 23(23):24032–24046
Ghodbane I, Nouri L, Hamdaoui O, Chiha M (2008) Kinetic and equilibrium study for the sorption of cadmium (II) ions from aqueous phase by eucalyptus bark. J Hazard Mater 152(1):148–158
Usman A, Sallam A, Zhang M, Vithanage M, Ahmad M, Al-Farraj A, Ok YS, Abduljabbar A, Al-Wabel M (2016) Sorption process of date palm biochar for aqueous Cd (II) removal: efficiency and mechanisms. Water Air Soil Pollut 227(12):449
Waalkes MP (2000) Cadmium carcinogenesis in review. J Inorg Biochem 79(1–4):241–244
Singh A, Prasad SM (2015) Remediation of heavy metal contaminated ecosystem: an overview on technology advancement. Int J Environ Sci Technol 12:353–366
Zhang S, Yu C, Liu N, Teng Y, Yin C (2019) Preparation of transparent anti-pollution cellulose carbamate regenerated cellulose membrane with high separation ability. Int J Biol Macromol 139:332–341
Gulgonul I, Çelik MS (2018) Understanding the flotation separation of Na and K feldspars in the presence of KCl through ion exchange and ion adsorption. Miner Eng 129:41–46
Li M, Xia X, Nie Z, Ma L, Liu Q (2019) Recovery of tungsten from WC–Co hard metal scraps using molten salts electrolysis. J Mater Res Technol 8(1):1440–1450
Wang T, Wang Q, Soklun H, Qu G, Xia T, Guo X, Jia H, Zhu L (2019) A green strategy for simultaneous Cu (II)-EDTA decomplexation and Cu precipitation from water by bicarbonate-activated hydrogen peroxide/chemical precipitation. Chem Eng J 370:1298–1309
Hasanzadeh V, Rahmanian O, Heidari M (2020) Cefixime adsorption onto activated carbon prepared by dry thermochemical activation of date fruit residues. Microchem J 152:104261
Ma L, He M, Fu P, Jiang X, Lv W, Huang Y, Liu Y, Wang H (2020) Adsorption of volatile organic compounds on modified spherical activated carbon in a new cyclonic fluidized bed. Sep Purif Technol 235:116146
Hashem A, Hussein HA, Sanousy MA, Adam E, Saad EE (2011) Monomethylolated thiourea–sawdust as a new adsorbent for removal of Hg (II) from contaminated water: equilibrium kinetic and thermodynamic studies. Polym Plast Technol Eng 50(12):1220–1230
Hashem A, Badawy SM (2015) Sesbania sesban L. biomass as a novel adsorbent for removal of Pb (II) ions from aqueous solution: non-linear and error analysis. Green Process Synth 4(3):179–190
Kwon J-S, Yun S-T, Lee J-H, Kim S-O, Jo HY (2010) Removal of divalent heavy metals (Cd, Cu, Pb, and Zn) and arsenic (III) from aqueous solutions using scoria: kinetics and equilibria of sorption. J Hazard Mater 174(1–3):307–313
Hashem A, Hammad HA, Al-Anwar A (2016) Modified Camelorum tree particles as a new adsorbent for adsorption of Hg (II) from aqueous solutions: kinetics, thermodynamics and non-linear isotherms. Desalin Water Treat 57(50):23827–23843
Hashem A, Al-Anwar A, Nagy NM, Hussein DM, Eisa SM (2016) Isotherms and kinetic studies on adsorption of Hg (II) ions onto Ziziphus spina-christi L. from aqueous solutions. Green Process Synth 5(2):213–224
Wu Y, Fan Y, Zhang M, Ming Z, Yang S, Arkin A, Fang P (2016) Functionalized agricultural biomass as a low-cost adsorbent: Utilization of rice straw incorporated with amine groups for the adsorption of Cr (VI) and Ni (II) from single and binary systems. Biochem Eng J 105:27–35
Xu C, Wang J, Yang T, Chen X, Liu X, Ding X (2015) Adsorption of uranium by amidoximated chitosan-grafted polyacrylonitrile, using response surface methodology. Carbohydr Polym 121:79–85
Abdouss M, Shoushtari AM, Shamloo N, Haji A (2013) Modified PET fibres for metal ion and dye removal from aqueous media. Polym Polym Compos 21(4):251–258
Racho P, Phalathip P (2017) Modified nylon fibers with amino chelating groups for heavy metal removal. Energy Procedia 118:195–200
Abdouss M, Shoushtari MA, Haji A, Moshref B (2012) Fabrication of chelating diethylenetriaminated pan micro-and nano-fibers for heavy metal removal. Chem Ind Chem Eng Q/CICEQ 18(1):27–34
Rangabhashiyam S, Anu N, Giri Nandagopal MS, Selvaraju N (2014) Relevance of isotherm models in biosorption of pollutants by agricultural byproducts. J Environ Chem Eng 2(1):398–414
Ng J, Cheung W, McKay G (2002) Equilibrium studies of the sorption of Cu (II) ions onto chitosan. J Colloid Interface Sci 255(1):64–74
Karaca S, Gürses A, Ejder M, Açikyildiz M (2004) Kinetic modeling of liquid-phase adsorption of phosphate on dolomite. J Colloid Interface Sci 277(2):257–263
Kapoor A, Yang R (1989) Correlation of equilibrium adsorption data of condensible vapours on porous adsorbents. Gas Sep Purif 3(4):187–192
Hossain M, Ngo H, Guo W (2013) Introductory of Microsoft Excel SOLVER function-spreadsheet method for isotherm and kinetics modelling of metals biosorption in water and wastewater. J Water Sustain 3:223–237
Hashem A, Badawy SM, Faraga S, Mohamed LA, Fletcher AJ, Tahaa GM (2020) Non-linear adsorption characteristics of modified pine wood sawdust optimised for adsorption of Cd(II) from aqueous systems. J Environ Chem Eng 8:103966
Sing KS (1982) Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (Provisional). Pure Appl Chem 54(11):2201–2218
Thommes M, Kaneko K, Neimark AV, Olivier JP, Rodriguez-Reinoso F, Rouquerol J, Sing KSW (2015) Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl Chem 87(9–10):1051–1069
Martín-Lara MÁ, Hernáinz F, Calero M, Blázquez G, Tenorio G (2009) Surface chemistry evaluation of some solid wastes from olive-oil industry used for lead removal from aqueous solutions. Biochem Eng J 44(2–3):151–159
Khalil A, Sokker HH, Al-Anwar A, Hashem A (2009) Preparation, characterization and utilization of amidoximated poly (AN/MAA)-grafted Alhagi residues for the removal of Zn (II) ions from aqueous solution. Adsorpt Sci Technol 27(4):363–382
Chen H, Zhao Y, Wang A (2007) Removal of Cu (II) from aqueous solution by adsorption onto acid-activated palygorskite. J Hazard Mater 149(2):346–354
Azouaou N, Sadaoui Z, Djaafri A, Mokaddem H (2010) Adsorption of cadmium from aqueous solution onto untreated coffee grounds: equilibrium, kinetics and thermodynamics. J Hazard Mater 184(1–3):126–134
Balkaya N, Cesur H (2008) Adsorption of cadmium from aqueous solution by phosphogypsum. Chem Eng J 140(1–3):247–254
Patterer MS, Bavasso I, Sambeth JE, Medici F (2017) Cadmium removal from acqueous solution by adsorption on spent coffee grounds. Chem Eng Trans 60:157–162
Ma F, Zhao B, Diao J (2016) Adsorption of cadmium by biochar produced from pyrolysis of corn stalk in aqueous solution. Water Sci Technol 74(6):1335–1345
Masoudi R, Moghimi H, Azin E, Taheri RA (2018) Adsorption of cadmium from aqueous solutions by novel Fe3O4-newly isolated Actinomucor sp. bio-nanoadsorbent: functional group study. Artif Cells Nanomed Biotechnol 46(sup3):S1092–S1101
Al-Anber ZA, Matouq MAD (2008) Batch adsorption of cadmium ions from aqueous solution by means of olive cake. J Hazard Mater 151(1):194–201
Hasan S, Krishnaiah A, Ghosh TK, Viswanath DS, Boddu VM, Smith ED (2006) Adsorption of divalent cadmium (Cd (II)) from aqueous solutions onto chitosan-coated perlite beads. Ind Eng Chem Res 45(14):5066–5077
Wang FY, Wang H, Ma JW (2010) Adsorption of cadmium (II) ions from aqueous solution by a new low-cost adsorbent—Bamboo charcoal. J Hazard Mater 177(1–3):300–306
Tajar AF, Kaghazchi T, Soleimani M (2009) Adsorption of cadmium from aqueous solutions on sulfurized activated carbon prepared from nut shells. J Hazard Mater 165(1–3):1159–1164
Langmuir I (1916) The constitution and fundamental properties of solids and liquids. Part I. Solids. J Am Chem Soc 38(11):2221–2295
Hall KR, Eagleton LC, Acrivos A, Vermeulen T (1966) Pore-and solid-diffusion kinetics in fixed-bed adsorption under constant-pattern conditions. Ind Eng Chem Fundam 5(2):212–223
Freundlich H (1907) Über die adsorption in lösungen. Zeitschrift für physikalische Chemie 57(1):385–470
Temkin M (1940) Kinetics of ammonia synthesis on promoted iron catalysts. Acta Physiochim URSS 12:327–356
Redlich O, Peterson DL (1959) A useful adsorption isotherm. J Phys Chem 63(6):1024–1024
Toth J (1971) State equation of the solid-gas interface layers. Acta Chim Hung 69:311–328
Sips R (1948) On the structure of a catalyst surface. J Chem Phys 16(5):490–495
Khan A, Ataullah R, Al-Haddad A (1997) Equilibrium adsorption studies of some aromatic pollutants from dilute aqueous solutions on activated carbon at different temperatures. J Colloid Interface Sci 194(1):154–165
Lagergren SK (1898) About the theory of so-called adsorption of soluble substances. Sven Vetenskapsakad Handingarl 24:1–39
Ho Y-S, McKay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34(5):451–465
Weber WJ, Morris JC (1963) Kinetics of adsorption on carbon from solution. J Sanit Eng Div 89(2):31–60
Unuabonah E, Adebowale K, Olu-Owolabi B (2007) Kinetic and thermodynamic studies of the adsorption of lead (II) ions onto phosphate-modified kaolinite clay. J Hazard Mater 144(1–2):386–395
Boparai HK, Joseph M, O’Carroll DM (2011) Kinetics and thermodynamics of cadmium ion removal by adsorption onto nano zerovalent iron particles. J Hazard Mater 186(1):458–465
Qi L, Teng F, Deng X, Zhang Y, Zhong X (2019) Experimental study on adsorption of Hg (II) with microwave-assisted alkali-modified fly ash. Powder Technol 351:153–158
Hu M, Tian H, He J (2019) Unprecedented selectivity and rapid uptake of CuS nanostructures toward Hg (II) ions. ACS Appl Mater Interfaces 11:19200–19206
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hashem, A., Nasr, M.F., Fletcher, A.J. et al. Aminated Acrylic Fabric Waste Derived Sorbent for Cd(II) Ion Removal from Aqueous Solutions: Mechanism, Equilibria and Kinetics. J Polym Environ 29, 175–186 (2021). https://doi.org/10.1007/s10924-020-01863-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-020-01863-z