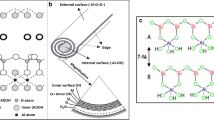
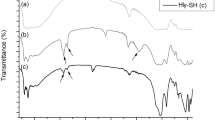
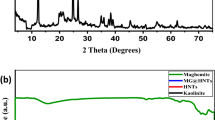
Abstract
In this study, natural halloysite nanotubes (HNTs) were applied to remove radiocobalt from wastewaters under various environmental parameters such as contact time, pH, ionic strength, foreign ions and temperature by using batch technique. The results indicated that the sorption of Co(II) on HNTs was dependent on ionic strength at pH < 8.5 and independent of ionic strength at pH > 8.5. Langmuir and Freundlich models were applied to simulate the sorption isotherms of Co(II) at three different temperatures of 293, 313 and 333 K. Langmuir model fitted the sorption isotherms of Co(II) on HNTs better than Freundlich model. The thermodynamic parameters (ΔG 0, ΔS 0 and ΔH 0) calculated from the temperature-dependent sorption isotherms manifested that the sorption of Co(II) on HNTs was an endothermic and spontaneous process. The sorption of Co(II) was dominated by outer-sphere surface complexation or ion exchange at low pH, whereas inner-sphere surface complexation or precipitation was the main sorption mechanism at high pH. The experimental results show that HNTs have good potentialities for cost-effective disposal of cobalt-bearing wastewaters.







Similar content being viewed by others
References
Guo ZQ, Xu DP, Zhao DL, Zhang SW, Xu JZ (2011) J Radioanal Nucl Chem 287:505–512
Sheng GD, Sheng J, Yang ST, Hu J, Wang XK (2011) J Radioanal Nucl Chem 289:129–135
Yang ST, Sheng GD, Tan XL, Hu J, Du JZ, Montavon G, Wang XK (2011) Geochim Cosmochim Acta 75:6520–6534
Yang ST, Sheng GD, Guo ZQ, Tan XL, Xu JZ, Wang XK (2012) Sci China Chem 55:632–642
Sheng GD, Yang ST, Sheng J, Hu J, Tan XL, Wang XK (2011) Environ Sci Technol 45:7718–7726
Chen L, Yu SM, Zuo LM, Liu B, Huang LL (2011) J Radioanal Nucl Chem 289:511–520
Liu ZJ, Chen L, Dong YH, Zhang ZC (2011) J Radioanal Nucl Chem 289:851–859
Tan LQ, Jin YL, Chen J, Cheng XC, Wu J, Feng LD (2011) J Radioanal Nucl Chem 289:601–610
Leggett RW, Eckerman KF (2008) Sci Total Environ 389:259–269
Rengaraj S, Moon SH (2002) Water Res 36:1783–1793
Wen T, Chen YT, Cai LS (2011) J Radioanal Nucl Chem 290:437–446
Chen L, Huang Y, Huang LL, Liu B, Wang G, Yu SM (2011) J Radioanal Nucl Chem 290:675–684
Hu J, Shao DD, Chen CL, Sheng GD, Li JX, Wang XK, Nagatsu M (2010) J Phys Chem B 114:6779–6785
Shao DD, Hu J, Sheng GD, Ren XM, Chen CL, Wang XK (2010) J Phys Chem C 114:21524–21530
El-Dessouky SI, El-Sofany EA, Daoud JA (2007) J Hazard Mater 143:17–23
Xu D, Shao DD, Chen CL, Ren AP, Wang XK (2006) Radiochim Acta 94:97–102
Chen CL, Hu J, Shao DD, Li JX, Wang XK (2009) J Hazard Mater 164:923–928
Prabhu PVSS, Narayana Swamy MS, Narasa Raju TSS (1981) IAWPC Tech Annu 8:46–52
Yang ST, Li JX, Shao DD, Hu J, Wang XK (2009) J Hazard Mater 166:109–116
Yang ST, Zhao DL, Zhang H, Lu SS, Chen L, Yu XJ (2010) J Hazard Mater 183:632–640
Zhao GX, Zhang HX, Fan QH, Ren XM, Li JX, Chen YX, Wang XK (2010) J Hazard Mater 173:661–668
Sheng GD, Li YM, Dong HP, Shao DD (2012) J Radioanal Nucl Chem. doi:10.1007/s10967-012-1735-9
Viseras MT, Aguzzi C, Cerezo P, Viseras C, Valenzuela C (2008) Microporous Mesoporous Mater 108:112–116
Liu RC, Zhang B, Mei DD, Zhang HQ, Liu JD (2011) Desalination 268:111–116
Luo P, Zhao YF, Zhang B, Liu JD, Yang Y, Liu JF (2010) Water Res 44:1489–1497
Wang JH, Zhang X, Zhang B, Zhao YF, Zhao R, Liu JD, Chen RF (2010) Desalination 259:22–28
Dong YH, Liu ZJ, Chen L (2012) J Radioanal Nucl Chem 292:435–443
Chen CL, Wang XK, Nagatsu M (2009) Environ Sci Technol 43:2362–2367
Ren XM, Shao DD, Yang ST, Hu J, Sheng GD, Tan XL, Wang XK (2011) Chem Eng J 170:170–177
Ho YS (2006) J Hazard Mater 136:681–689
Yang SB, Hu J, Chen CL, Shao DD, Wang XK (2011) Environ Sci Technol 45:3621–3627
Ren XM, Wang SW, Yang ST, Li JX (2010) J Radioanal Nucl Chem 283:253–259
Zhao GX, Ren XM, Gao X, Tan XL, Li JX, Chen CL, Huang YY, Wang XK (2011) Dalton Trans 40:10945–40952
Yüzer H, Kara M, Sabah E, Celik MS (2008) J Hazard Mater 151:33–37
Tan XL, Wang XK, Geckeis H, Rabung T (2008) Environ Sci Technol 42:6532–6537
Yang ST, Li JX, Lu Y, Chen YX, Wang XK (2009) Appl Radiat Isot 67:1600–1608
Baeyens B, Bradbury MH (1997) J Contam Hydrol 27:199–222
Shao DD, Jiang ZQ, Wang XK, Li JX, Meng YD (2009) J Phys Chem B 113:860–864
Yang ST, Zhao DL, Sheng GD, Sun YB (2011) J Radioanal Nucl Chem 289:467–477
Tan XL, Hu J, Montavon G, Wang XK (2011) Dalton Trans 40:10953–10960
Zuo LM, Yu SM, Zhou H, Tian X, Jiang J (2011) J Radioanal Nucl Chem 288:379–387
Cotton F, Wilkinson G (1980) Advance inorganic chemistry. Wiley, New York
Esmadi F, Simm J (1995) Colloid Surf A 1042:65–270
Shao DD, Xu D, Wang SW, Fan QH, Wu WS, Dong YH, Wang XK (2009) Sci China Chem 52:362–371
Liu MC, Chen CL, Hu J, Wu XL, Wang XK (2011) J Phys Chem C 115:25234–25240
Yang ST, Guo ZQ, Sheng GD, Wang XK (2012) Sci Total Environ 420:214–221
Xu D, Tan XL, Chen CL, Wang XK (2008) Appl Clay Sci 41:37–46
Jiang MQ, Jin XY, Lu XQ, Chen ZL (2010) Desalination 252:33–39
Zhou YT, Nie HL, Branford-White C, He ZY, Zhu LM (2009) J Colloid Interface Sci 330:29–37
Sheng GD, Li JX, Shao DD, Hu J, Chen CL, Chen YX, Wang XK (2010) J Hazard Mater 178:333–340
Bartell FE, Thomas TL, Fu Y (1951) J Phys Chem 55:1456–1462
Tan XL, Fang M, Chen CL, Yu SM, Wang XK (2008) Carbon 46:1741–1750
Zhao GX, Li JX, Ren XM, Chen CL, Wang XK (2011) Environ Sci Technol 45:10454–10462
Ajmal M, Rao RAK, Anwar S, Ahmad J, Ahmad R (2003) Bioresour Technol 86:147–149
Chen CL, Li XL, Zhao DL, Tan XL, Wang XK (2007) Colloids Surf A 302:449–454
Wong KK, Lee CK, Low KS, Haron MJ (2003) Chemosphere 50:23–28
Acknowledgments
Financial support from the National Science Foundation of China (21071147, 21107115, 21077107, 20971126 and 51062002)) is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, J., Wen, F., Pan, L. et al. Removal of radiocobalt ions from aqueous solutions by natural halloysite nanotubes. J Radioanal Nucl Chem 295, 431–438 (2013). https://doi.org/10.1007/s10967-012-1823-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-012-1823-x