Abstract
Purpose
Deposition characteristics of MDI and DPI aerosols were compared throughout the conducting airways for the first time using a combination of in vitro experiments and a newly developed stochastic individual path (SIP) model for different inhalation profiles.
Methods
In vitro experiments were used to determine initial particle distribution profiles and to validate computational fluid dynamics (CFD) model results for a MDI and DPI delivering the same dose of drug in a geometry of the mouth-throat and tracheobronchial airways. The validated CFD model was then used to predict the transport and deposition of the drug using correct and incorrect inhalation profiles for each inhaler.
Results
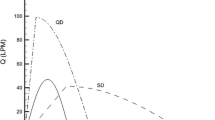
The MDI delivered approximately two times more drug to the tracheobronchial region compared with the DPI for both correct and incorrect inhalation profiles. Errors in inhalation reduced the deposited tracheobronchial dose by approximately 30% for both inhalers. The DPI delivered the largest dose to the mouth-throat (~70%) and the MDI delivered the largest dose to the alveolar airways (~50%).
Conclusions
The developed in silico model provides new insights into the lung delivery of pharmaceutical aerosols and can be applied in future studies in combination with pharmacokinetic analysis to establish bioequivalence between devices.











Similar content being viewed by others
Abbreviations
- B#:
-
bifurcation number (1 is trachea plus main bronchi)
- C/P:
-
central to peripheral lung deposition ratio
- CFC:
-
chlorofluorocarbon
- CFD:
-
computational fluid dynamics
- COPD:
-
chronic obstructive pulmonary disease
- CT:
-
computed tomography
- DE:
-
deposition efficiency
- DF:
-
deposition fraction
- DPI:
-
dry powder inhaler
- ECG:
-
enhanced condensational growth
- exp:
-
experiment
- FR:
-
fraction remaining
- HFA:
-
hydrofluoroalkane
- HPLC:
-
high-performance liquid chromatography
- LRN:
-
low Reynolds number
- MDI:
-
metered dose inhaler
- MMAD:
-
mass median aerodynamic diameter
- MT:
-
mouth-throat
- NGI:
-
next generation impactor
- PF:
-
penetration fraction
- QD:
-
quick and deep inhalation waveform
- SD:
-
slow and deep inhalation waveform
- SIP:
-
stochastic individual path
- Ta-mean :
-
time of mean accelerating flow
- TB:
-
tracheobronchial
- USP:
-
United States Pharmacopeial
References
Byron PR. Drug delivery devices: issues in drug development. Proc Am Thorac Soc. 2004;1:321–8.
Finlay WH. The mechanics of inhaled pharmaceutical aerosols. San Diego: Academic; 2001.
Newman SP. Principles of metered-dose inhaler design. Respir Care. 2005;50(9):1177–90.
Newman SP, Busse WW. Evolution of dry powder inhaler design, formulation, and performance. Respir Med. 2002;96:293–304.
Cochrane GM, Bala MV, Downs KE, Mauskopf J, Ben-Joseph RH. Inhaled corticosteroids for asthma therapy: patient compliance, devices, and inhalation technique. Chest. 2000;117:542–50.
Smith IJ, Bell J, Bowman N, Everard M, Stein S, Weers JG. Inhaler devices: what remains to be done? J Aerosol Med Pulm Drug Deliv. 2010;23:S25–37.
Ross DL, Schultz RK. Effect of inhalation flow rate on the dosing characteristics of dry powder inhaler (DPI) and metered dose inhaler (MDI) products. J Aerosol Med. 1996;9(2):215–26.
Tarsin WY, Pearson SB, Assi KH, Chrystyn H. Emitted dose estimates from Seretide Diskus and Symbicort Turbuhaler following inhalation by severe asthmatics. Int J Pharmaceut. 2006;316(1–2):131–7.
Scheuch G, Bennett W, Borgstrom L, Clark A, Dalby R, Dolovich M, et al. Deposition, Imaging, and Clearance: What Remains to be Done? J Aerosol Med Pulm Drug Deliv. 2010;23:S39–57.
Hindle M, Newton DAG, Chrystyn H. Dry powder inhalers are bioequivalent to metered dose inhalers: a study using a new urinary albuterol (salbutamol) assay technique. Chest. 1995;107:629–33.
Borgstrom L, Derom E, Stahl E, WahlinBoll E, Pauwels R. The inhalation device influences lung deposition and bronchodilating effect of terbutaline. Am J Respir Crit Care Med. 1996;153(5):1636–40.
Bondesson E, Friberg K, Soliman S, Lofdahl CG. Safety and efficacy of a high cumulative dose of salbutamol inhaled via Turbuhaler or via a pressurized metered-dose inhaler in patients with asthma. Respir Med. 1998;92(2):325–30.
Bollert FGE, Matusiewicz SP, Dewar MH, Brown GM, McLean A, Greening AP, et al. Comparative efficacy and potency of ipratropium via Turbuhaler and pressurized metered-dose inhaler in reversible airflow obstruction. Eur Respir J. 1997;10(8):1824–8.
Hirst PH, Bacon RE, Pitcairn GR, Silvasti M, Newman SP. A comparison of the lung deposition of budesonide from Easyhaler, Turbuhaler and pMDI plus spacer in asthmatic patients. Respir Med. 2001;95(9):720–7.
Fleming J, Conway J, Majoral C, Tossici-Bolt L, Katz I, Caillibotte G, et al. The use of combined single photon emission computed tomography and x-ray computed tomography to assess the fate of inhaled aerosol. J Aerosol Med Pulm Drug Deliv. 2011;24(1):49–60.
Longest PW, and Holbrook LT. In silico models of aerosol delivery to the respiratory tract—Development and applications. Advanced Drug Delivery Reviews 2011; doi:10.1016/j.addr.2011.05.009
Asgharian B, Hofmann W, Bergmann R. Particle deposition in a multiple-path model of the human lung. Aero Sci Tech. 2001;34:332–9.
Kim CS. Deposition of aerosol particles in human lungs: in vivo measurement and modeling. Biomarkers. 2009;14(S1):54–8.
Martonen TB, Musante CJ, Segal RA, Schroeter JD, Hwang D, Dolovich MA, et al. Lung models: strengths and limitations. Respir Care. 2000;45(6):712–36.
Ma B, Lutchen KR. CFD simulations of aerosol deposition in an anatomically based human large-medium airway model. Ann Biomed Eng. 2009;37(2):271–85.
Lambert AR, O’Shaughnessy PT, Tawhai MH, Hoffman EA, Lin C-L. Regional deposition of particles in an image-based airway model: large-eddy simulation and left-right lung ventilation asymmetry. Aero Sci Tech. 2011;45:11–25.
Longest PW, Xi J. Condensational growth may contribute to the enhanced deposition of cigarette smoke particles in the upper respiratory tract. Aero Sci Tech. 2008;42:579–602.
Zhang Z, Kleinstreuer C. Computational analysis of airflow and nanoparticle deposition in a combined nasal-oral-tracheobronchial airway model. J Aero Sci. 2011;42:174–94.
Kleinstreuer C, Zhang Z. An adjustable triple-bifurcation unit model for air-particle flow simulations in human tracheobronchial airways. J Biomech Eng. 2009;131:021007.
Zhang Z, Kleinstreuer C, Kim CS. Comparison of analytical and CFD models with regard to micron particle deposition in a human 16-generation tracheobronchial airway model. Aero Sci. 2009;40:16–28.
DeHaan WH, Finlay WH. Predicting extrathoracic deposition from dry powder inhalers. J Aero Sci. 2004;35:309–31.
Longest PW, Hindle M, Das Choudhuri S, Xi J. Comparison of ambient and spray aerosol deposition in a standard induction port and more realistic mouth-throat geometry. J Aero Sci. 2008;39:572–91.
Longest PW, Hindle M. Quantitative analysis and design of a spray aerosol inhaler. Part 1: Effects of dilution air inlets and flow paths. J Aerosol Med Pulm Drug Deliv. 2009;22(3):271–83.
Ilie M, Matida EA, Finlay WH. Asymmetrical aerosol deposition in an idealized mouth with a DPI mouthpiece inlet. Aero Sci Tech. 2008;42:10–7.
Longest PW, Hindle M, Das Choudhuri S. Effects of generation time on spray aerosol transport and deposition in models of the mouth-throat geometry. J Aerosol Med Pulm Drug Deliv. 2009;22(3):67–84.
Matida EA, DeHaan WH, Finlay WH, Lange CF. Simulation of particle deposition in an idealized mouth with different small diameter inlets. Aero Sci Tech. 2003;37:924–32.
Finlay WH, Martin AR. Modeling of aerosol deposition within interface devices. J Aerosol Med. 2007;20(S1):S19–28.
Tian G, Longest PW, Su G, Hindle M. Characterization of respiratory drug delivery with enhanced condensational growth (ECG) using an individual path model of the entire tracheobronchial airways. Ann Biomed Eng. 2011;39(3):1136–53.
Tian G, Longest PW, Su G, Walenga RL, Hindle M. Development of a stochastic individual path (SIP) model for predicting the tracheobronchial deposition of pharmaceutical aerosols: effects of transient inhalation and sampling the airways. J Aerosol Sci. 2011;42:781–99.
Hindle M, Longest PW. Evaluation of enhanced condensational growth (ECG) for controlled respiratory drug delivery in a mouth-throat and upper tracheobronchial model. Pharm Res. 2010;27:1800–11.
Cegla UH. Pressure and inspiratory flow characteristics of dry powder inhalers. Respir Med. 2004;98:S22–8.
Swift DL. The oral airway—A conduit or collector for pharmaceutical aerosols. Respiratory Drug Delivery IV 1994:187–195.
Fadl A, Wang J, Zhang Z. Metered-dose inhaler efficiency enhancement: A case study and novel design. Inhal Toxicol. 2010;22(7):601–9.
Finlay WH, Gehmlich MG. Inertial sizing of aerosol inhaled from two dry powder inhalers with realistic breath patterns versus constant flow rates. Int J Pharm. 2000;210:83–95.
Byron PR, Delvadia RR, Longest PW, Hindle M. Stepping into the trachea with realistic physical models: uncertainties in regional drug deposition from powder inhalers. Respiratory Drug Delivery. 2010;1:215–24.
Delvadia RR, Byron PR, Longest PW, Hindle M. In vitro prediction of regional drug deposition from dry powder inhalers. Respiratory Drug Delivery. 2010;2010:907–11.
Broeders MEAC, Molema J, Hop WCJ, Folgering HTM. Inhalation profiles in asthmatics and COPD patients: Reproducibility and effect of instruction. J Aerosol Med. 2003;16(2):131–41.
Xi J, Longest PW. Transport and deposition of micro-aerosols in realistic and simplified models of the oral airway. Ann Biomed Eng. 2007;35(4):560–81.
Cheng KH, Cheng YS, Yeh HC, Swift DL. Measurements of airway dimensions and calculation of mass transfer characteristics of the human oral passage. J Biomech Eng. 1997;119:476–82.
Yeh HC, Schum GM. Models of human lung airways and their application to inhaled particle deposition. Bull Math Biology. 1980;42:461–80.
ICRP. Human Respiratory Tract Model for Radiological Protection. New York: Elsevier Science Ltd.; 1994.
Heistracher T, Hofmann W. Physiologically realistic models of bronchial airway bifurcations. J Aerosol Sci. 1995;26(3):497–509.
Horsfield K, Dart G, Olson DE, Cumming G. Models of the human bronchial tree. J Appl Physiol. 1971;31:207–17.
Hammersley JR, Olson DE. Physical models of the smaller pulmonary airways. J Appl Physiol. 1992;72:2402–14.
Russo J, Robinson R, Oldham MJ. Effects of cartilage rings on airflow and particle deposition in the trachea and main bronchi. Med Eng Phys. 2008;30:581–9.
Vinchurkar S, Longest PW. Evaluation of hexahedral, prismatic and hybrid mesh styles for simulating respiratory aerosol dynamics. Computers and Fluids. 2008;37:317–31.
Phalen RF, Yeh HC, Schum GM, Raabe OG. Application of an idealized model to morphometry of the mammalian tracheobronchial tree. Anat Rec. 1978;190:167–76.
Olsson B, Berg E, Svensson M. Comparing aerosol size distributions that penetrate mouth-throat models under realistic inhalation conditions. Respiratory Drug Delivery. 2010;2010:225–34.
van der Palen J. Peak inspiratory flow through Diskus and Turbuhaler, measured by means of a peak inspiratory flow meter (In-Check Dial). Respir Med. 2003;97:285–98.
Horsfield K, Dart G, Olson DE, Filley GF, Cumming G. Models of the human bronchial tree. J Appl Physiol. 1971;31:207–17.
Asgharian B, Price OT. Airflow distribution in the human lung and its influence on particle deposition. Inhal Toxicol. 2006;18:795–801.
Yin Y, Choi J, Hoffman EA, Tawhai MH, Lin C-L. Simulation of pulmonary air flow with a subject-specific boundary condition. J Biomech. 2010;43:2159–63.
Hochrainer D, Holz H, Kreher C, Scaffidi L, Spallek M, Wachtel H. Comparison of the aerosol velocity and spray duration of Respimat soft mist inhaler and pressurized metered dose inhalers. J Aerosol Med. 2005;18(3):273–82.
Longest PW, Vinchurkar S. Validating CFD predictions of respiratory aerosol deposition: effects of upstream transition and turbulence. J Biomech. 2007;40:305–16.
Xi J, Longest PW, Martonen TB. Effects of the laryngeal jet on nano- and microparticle transport and deposition in an approximate model of the upper tracheobronchial airways. J Appl Physiol. 2008;104:1761–77.
Longest PW, Hindle M. Das Choudhuri S, and Byron PR. Numerical simulations of capillary aerosol generation: CFD model development and comparisons with experimental data. Aero Sci Tech. 2007;41:952–73.
Longest PW, Hindle M. Evaluation of the Respimat Soft Mist inhaler using a concurrent CFD and in vitro approach. J Aerosol Med Pulm Drug Deliv. 2009;22(2):99–112.
Longest PW, Hindle M, Das Choudhuri S, and Byron PR. Developing a better understanding of spray system design using a combination of CFD modeling and experiment. In: Dalby RN, Byron PR, Peart J, Suman JD, Farr SJ, Young PM editors. Proceedings of Respiratory Drug Delivery 2008. Illinois: Davis Healthcare International Publishing; 2008. p. 151–163.
Matida EA, Finlay WH, Grgic LB. Improved numerical simulation of aerosol deposition in an idealized mouth-throat. J Aerosol Sci. 2004;35:1–19.
Longest PW, Xi J. Effectiveness of direct Lagrangian tracking models for simulating nanoparticle deposition in the upper airways. Aero Sci Tech. 2007;41:380–97.
Stein SW, Myrdal PB. The relative influence of atomization and evaporation on metered dose inhaler drug delivery efficiency. Aero Sci Tech. 2006;40:335–47.
Li Z, Kleinstreuer C, Zhang Z. Particle deposition in the human tracheobronchial airways due to transient inspiratory flow patterns. Aerosol Science. 2007;38:625–44.
Longest PW, Vinchurkar S. Effects of mesh style and grid convergence on particle deposition in bifurcating airway models with comparisons to experimental data. Med Engr Phys. 2007;29(3):350–66.
Stein SW. Aiming for a moving target: challenges with impactor measurements of MDI aerosols. Int J Pharm. 2008;355(1–2):53–61.
Patton JS, Brain JD, Davies LA, Fiegel J, Gumbleton M, Kim KJ, et al. The particle has landed-characterizing the fate of inhaled pharmaceuticals. J Aerosol Med Pulm Drug Deliv. 2010;23:S71–87.
Patton JS, Byron PR. Inhaling medicines: delivering drugs to the body through the lungs. Nat Rev Drug Discov. 2007;6:67–74.
Warren SJ, Taylor G. Effect of inhalation flow profiles on the deposition of radiolabelled BDP from a novel dry powder inhaler, a conventional metered dose inhaler and MDI plus spacer. In: Dalby RN, Byron PR, Farr SJ, editors. Respiratory Drug Delviery VI. Buffalo Grove: Interpharm Press; 1998. p. 453–5.
Anafi RC, Wilson TA. Airway stability and heterogeneity in the constricted lung. J Appl Physiol. 2001;91(3):1185–92.
Kotaru C, Coreno A, Skowronski M, Muswick G, Gilkeson RC, McFadden ER. Morphometric changes after thermal and methacholine bronchoprovocations. J Appl Physiol. 2005;98(3):1028–36.
Tgavalekos NT, Tawhai M, Harris RS, Mush G, Vidal-Melo M, Venegas JG, et al. Identifying airways responsible for heterogeneous ventilation and mechanical dysfunction in asthma: an image functional modeling approach. J Appl Physiol. 2005;99(6):2388–97.
O’Callaghan C, Ehtezazi T, Horsfield MA, and Barry PW. A voyage into the respiratory tract. Respiratory Drug Delivery VIII 2002:165–170.
O’Callaghan C, Wright P, Shrubb I, and Barry PW. Oral configuration during inhalation from a pressurised metered dose inhaler. Respiratory Drug Delivery VII 2000:589–591.
Acknowledgments & Disclosures
This study was supported in part by a contract from the United States Food and Drug Administration (Number HHSF223201000093C). The content is solely the responsibility of the authors and does not necessarily represent the official views of the US Food and Drug Administration.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Longest, P.W., Tian, G., Walenga, R.L. et al. Comparing MDI and DPI Aerosol Deposition Using In Vitro Experiments and a New Stochastic Individual Path (SIP) Model of the Conducting Airways. Pharm Res 29, 1670–1688 (2012). https://doi.org/10.1007/s11095-012-0691-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-012-0691-y