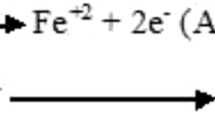Abstract
The effect of different temperatures and acid concentrations on the corrosion of mild steel in nitric acid are addressed in this work. The effect of temperature is explained by the application of the Arrhenius equation and transition state theory, while the acid concentration effect is explained using reaction kinetic equations. The combined effect of temperature and acid concentration is then modeled using a nonlinear regression method. Thermodynamic parameters of activation (E, ΔH* and ΔS*) and detailed kinetic studies for the corrosion reaction are carried out. Nonlinear corrosion rates as a function of temperature and acid concentration are estimated with a good prediction of corrosion rate values. The values of activation energy E and enthalpy of activation ΔH* decrease with an increase in acid concentration indicating the increasing reaction rate. Entropies of activation ΔS* tend to lower values with increasing acid concentration, which indicates that the activated complex is more orderly relative to the initial state. The corrosion reaction was approximately first order.





Similar content being viewed by others
References
Osarolube E, Owate IO, Oforka NC (2008) Corrosion behavior of mild and high carbon steels in various acidic media. Sci Res Essay 3:224–228
Eddy NO, Ekwungbo P, Odoemelam SA (2008) Inhibition of the corrosion of mild steel in H2SO4 by 5-amino-1-cyclopropyl-7-[(3R, 5S) 3,5-dimethylpiperazin-1-YL]-6,8-difluoro-4-oxo-uinoline-3-carboxylic acid (ACPDQC). Int J Phy Sci 3:275–280
Khadom AA, Yaro AS, AlTaie AS, Kadhum AA (2009) Electrochemical, activations and adsorption studies for the corrosion inhibition of low carbon steel in acidic media. Port Electrochim Acta 27:699–712
Khaled KF (2011) Studies of the corrosion inhibition of copper in sodium chloride solutions using chemical and electrochemical measurements. Mater Chem Phys 125:427–433
Eddy NO, Odoemelam SA, Odiongenyi AO (2009) J Appl Electrochem 39:849–857
Eddy NO, Odoemelam SA, Akpanudoh NW (2008) J Chem Tech 4:1–10
Khadom AA, Yaro AS, Kadhum AH (2010) J Chilean Chem Soc 55:150–152
Khadom AA, Yaro AS (2010) Int J Surf Sci Eng 4:429–438
Musa AY, Kadhum AH, Mohamad AB, Takriff MS, Daud AR, Kamarudin SK (2010) Corros Sci 52:526–532
Upadhyay SK (2006) Chemical kinetics and reaction dynamics. Anamaya Publishers, India
Grahame DC (1947) Chem Rev 41:441–448
Khadom AA (2013) Modeling of corrosion reaction data in inhibited acid environment using regressions and artificial neural networks. Korean J Chem Eng 30:2197–2204
Khadom AA (2013) Generalization of corrosion reaction kinetic models for steels in inhibited acidic media. Mater Res Innov 17:194–199
Khadom AA (2013) Effect of corrosive solution motion on copper–nickel alloy pipe in presence of naphthylamine as a corrosion inhibitor. J Mater Environ Sci 4:510–519
Evans UR (1944) Behavior of metals in nitric acid. Trans Faraday Soc 40:120–130
Uhlig HH (1948) Corrosion handbook. The Electrochemical Society, New York
Mathur PB, Vasudevan T (1982) Corrosion 38:150–160
Morad MS (1999) Mater Chem Phys 60:188–195
Khadom AA, Yaro AS (2011) Protection of low carbon steel in phosphoric acid by potassium iodide. Prot Met Phys Chem Surf J 47:662–669
Yaro AS, Wael RK, Khadom AA (2010) Reaction kinetics of corrosion of mild steel in phosphoric acid. J Univ Chem Technol Met 45:443–448
Popova A, Veleva S, Raicheva S (2005) React Kinet Catal Lett 85:99–105
Abiola OK (2005) J Chil Chem Soc 50:685–690
Musa AY, Khadom AA, Kadhum AH, Mohamad AB, Takriff MS (2010) Kinetic behavior of mild steel corrosion inhibition by 4-amino-5-phenyl-4H-1,2,4-trizole-3-thiol. J Taiwan Inst Chem Eng 41:126–128
Missen RW, Mims CA, Bradley AS (1999) Introduction to chemical reaction engineering and kinetics. Wiley, USA
Eddy NO, Odoemelam SA, Mbaba AJ (2008) Inhibition of the corrosion of mild steel in HCl by Sparfloxacin. Afr J Pure Appl Chem 2:132–138
Hill CG (1977) An introduction to chemical engineering kinetics and reactor design. Wiley, Canada
Yaro AS, Khadom AA, Lahmod SM (2013) Kinetics of the corrosion inhibition reaction of steel alloys in acidic media by potassium iodide. Reac Kinet Mech Cat 109:417–432
Acknowledgments
This work was supported by Kufa University, Materials Engineering Department, which is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khadom, A.A., Abdul-Hadi, A.A. Kinetic and mathematical approaches to the corrosion of mild steel in nitric acid. Reac Kinet Mech Cat 112, 15–26 (2014). https://doi.org/10.1007/s11144-014-0683-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-014-0683-5