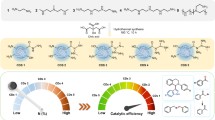
Abstract
In this work, titanium dioxide nanoparticles (TiO2 NPs) and modified TiO2 NPs with silver (Ag) or platinum (Pt) dopant were developed through photodeposition method for the NOx conversion into nitric acid (HNO3) under visible light irradiation. The formed photocatalysts TiO2, Ag/TiO2, and Pt/TiO2 nanocomposites were characterized by utilizing TEM, SEM, energy-dispersive X-ray analysis (EDX), XRD, UV/visible diffuse reflectance spectroscopy (UV-Vis DRS), and FT-IR. It had been investigated that an enhancement within the conversion of NOx into HNO3 was increased from 34.3 to 78.3% for Ag/TiO2 and from 35.2 to 78.5% for Pt/TiO2 under visible light irradiation conditions at room temperature for less than 2 h. The photodegradation rate order of NOx under visible light irradiation is Pt/TiO2 ~ Ag/TiO2 > TiO2. A possible mechanism for the catalytic conversion of NOx gases has been proposed, which depends on the photogeneration of electrons and holes after the excitation of nanocatalysts under visible radiation that promoted superoxide and hydroxyl ions, which can depredate NOx gases. This approach of NOx photocatalytic conversion is characterized by its chemical stability, low cost, high efficiency, simple operation, and strong durability than traditional methods.









Similar content being viewed by others
References
Abou Saoud W, Assadi AA, Guiza M, Bouzaza A, Aboussaoud W, Ouederni A, Soutrel I, Wolbert D, Rtimi S (2017) Study of synergetic effect, catalytic poisoning and regeneration using dielectric barrier discharge and photocatalysis in a continuous reactor: abatement of pollutants in air mixture system. Appl Catal B Environ 213:53–61. https://doi.org/10.1016/j.apcatb.2017.05.012
Amer MS, Ghanem MA, Al-Mayouf AM, Arunachalam P, Khdary NH (2020) Low-loading of oxidized platinum nanoparticles into mesoporous titanium dioxide for effective and durable hydrogen evolution in acidic media. Arab J Chem 13(1):2257–2270. https://doi.org/10.1016/j.arabjc.2018.04.010
Ao CH, Lee SC, Mak CL, Chan LY (2003) Photodegradation of volatile organic compounds (VOCs) and NO for indoor air purification using TiO2: promotion versus inhibition effect of NO. Appl Catal B Environ 42(2):119–129. https://doi.org/10.1016/S0926-3373(02)00219-9
Arana J, Dona-Rodriguez JM, Melian JAH, Rendon ET, Diaz OG (2005) Role of Pd and Cu in gas-phase alcohols photocatalytic degradation with doped TiO2. J Photochem Photobiol A Chem 174:7–14. https://doi.org/10.1016/j.jphotochem.2005.03.003
Attia YA (2016) Ag/ZnO/graphene-TBSCl hybrid nanocomposite as highly efficient catalyst for hydrogen production. Mater Express 6(3):211–219. https://doi.org/10.1166/mex.2016.1297
Attia YA, Abdel-Hafez SH (2020) Reusable photoresponsive Ag/AgCl nanocube-catalyzed one-pot synthesis of seleno [2, 3-b] pyridine derivatives. Res Chem Intermed 46(6):3165–3177. https://doi.org/10.1007/s11164-020-04143-6
Attia YA, Altalhi TA (2017) Low-cost synthesis of titanium dioxide anatase nanoclusters as advanced materials for hydrogen photoproduction. Res Chem Intermed 43(7):4051–4062. https://doi.org/10.1007/s11164-017-2862-2
Attia YA, Mohamed YMA (2019) Silicon-grafted Ag/AgX/rGO nanomaterials (X= Cl or Br) as dip-photocatalysts for highly efficient p-nitrophenol reduction and paracetamol production. Appl Organomet Chem 33(3):e4757. https://doi.org/10.1002/aoc.4757
Attia YA, Vázquez- Vázquez C, YMA M (2017) Facile production of vitamin B3 and other heterocyclic carboxylic acids using an efficient Ag/ZnO/graphene-Si hybrid nanocatalyst. Res Chem Intermed 43(1):203–218. https://doi.org/10.1007/s11164-016-2615-7
Attia YA, Mohamed YMA, Awad MM, Alexeree S (2020) Ag doped ZnO nanorods catalyzed photo-triggered synthesis of some novel (1H-tetrazol-5-yl)-coumarin hybrids. J Organomet Chem 919:121320. https://doi.org/10.1016/j.jorganchem.2020.121320
Ballari MM, Hunger M, Hüsken G, Brouwers HJH (2010) NOx photocatalytic degradation employing concrete pavement containing titanium dioxide. Appl Catal B Environ 95:245–254. https://doi.org/10.1016/j.apcatb.2010.01.002
Ballari MM, Yu QL, Brouwers HJH (2011) Experimental study of the NO and NO2 degradation by photocatalytically active concrete. Catal Today 161:175–180. https://doi.org/10.1016/j.cattod.2010.09.028
Bellardita M, El Nazer HA, Loddo V, Parrino F, Venezia AM, Palmisano L (2017) Photoactivity under visible light of metal loaded TiO2 catalysts prepared by low frequency ultrasound treatment. Catal Today 284:92–99. https://doi.org/10.1016/j.cattod.2016.11.026
Bindu P, Thomas S (2014) Estimation of lattice strain in ZnO nanoparticles: X-ray peak profile analysis. J Theor Appl Phys 8:123–134. https://doi.org/10.1007/s40094-014-0141-9
Dalton JS, Janes PA, Jones NG, Nicholson JA, Hallam KR, Allen GC (2002) Photocatalytic oxidation of NOx gases using TiO2: a surface spectroscopic approach. Environ Pollut 120:415–422. https://doi.org/10.1016/s0269-7491(02)00107-0
Devahasdin S, Fan C, Li KY, Chen DH (2003) TiO2 photocatalytic oxidation of nitric oxide: transient behavior and reaction kinetics. J Photochem Photobiol A Chem 156(1–3):161–170. https://doi.org/10.1016/S1010-6030(03)00005-4
Ding X, Song X, Li P, Ai Z, Zhang L (2011) Efficient visible light driven photocatalytic removal of NO with aerosol flow synthesized B, N-codoped TiO2 hollow spheres. J Hazard Mater 190:604–612. https://doi.org/10.1016/j.jhazmat.2011.03.099
Gaidi M, Hajjaji A, El Khakani MA, Chenevier B, Labeau M, Bessaïs B (2009) Optical properties tuning of SnO2 films by metal incorporation (Pt,Pd): correlation with microstructure change. Jpn J Appl Phys 48:072501. https://doi.org/10.1143/JJAP.48.072501
Guo GF, Hu Y, Jiang SM, Wei CH (2012) Photocatalytic oxidation of NOx over TiO2/HZSM-5 catalysts in the presence of water vapor: effect of hydrophobicity of zeolites. J Hazard Mater 223:39–45. https://doi.org/10.1016/j.jhazmat.2012.04.043
Hajjaji A, Gaidi M, Bessais B, El Khakani MA (2011a) Effect of Cr incorporation on the structural and optoelectronic properties of TiO2:Cr deposited by means of a magnetron co-sputtering process. Appl Surf Sci 257(24):10351–10357. https://doi.org/10.1016/j.apsusc.2011.06.072
Hajjaji A, Labidi A, Gaidi M, Ben Rabha M, Smirani R, Bejaoui A, Bessais B, El Khakani MA (2011b) Structural, optical and sensing properties of Cr-doped TiO2 thin films. J Sens Lett 9:1697–1703. https://doi.org/10.1166/sl.2011.1736
Hashimoto K, Wasada K, Osaki M, Shono E, Adachi K, Toukai N, Kominami H, Kera Y (2001) Photocatalytic oxidation of nitrogen oxide over titania–zeolite composite catalyst to remove nitrogen oxides in the atmosphere. Appl Catal B 30:429–436. https://doi.org/10.1016/S0926-3373(00)00258-7
Hu Y, Song X, Jiang S, Wei C (2015) Enhanced photocatalytic activity of Pt-doped TiO2 for NOx oxidation both under UV and visible light irradiation: a synergistic effect of lattice Pt4+ and surface PtO. Chem Eng J 274:102–112. https://doi.org/10.1016/j.cej.2015.03.135
Ichiura H, Kitaota T, Tanaka H (2003) Photocatalytic oxidation of NOx using composite sheets containing TiO2 and a metal compound. Chemosphere 51:855–860. https://doi.org/10.1016/S0045-6535(03)00049-3
Janus M, Zając K, Zatorska J, Kusiak-Nejman E, Czyżewski A, Morawski AWA (2015) Cementitious plates containing TiO2-N, C photocatalysts for NOx degradation. J Adv Oxid Technol 18:227–232. https://doi.org/10.1515/jaots-2015-0207
Karthik L, Kumar G, Kirthi AV, Rahuman AA, Bhaskara RKV (2014) Streptomyces sp. LK3 mediated synthesis of silver nanoparticles and its biomedical application. Bioprocess Biosyst Eng 37:261–267. https://doi.org/10.1007/s00449-013-0994-3
Kim BM, Yadav HM, Kim JS (2016) Photocatalytic degradation of gaseous benzene on photodeposited Ag–TiO2 nanoparticles. J Nanosci Nanotechnol 16(10):10991–10997. https://doi.org/10.1166/jnn.2016.13276
Lasek J, Yu YH, Wu JCS (2013) Removal of NOx by photocatalytic processes. J Photochem Photobiol C: Photochem Rev 14(1):29–52. https://doi.org/10.1016/j.jphotochemrev.2012.08.002
Li X, Zou X, Qu Z, Zhao Q, Wang L (2011) Photocatalytic degradation of gaseous toluene over Ag-doping TiO2 nanotube powder prepared by anodization coupled with impregnation method. Chemosphere 83(5):674–679. https://doi.org/10.1016/j.chemosphere.2011.02.043
Lin L, Chai Y, Zhao B, Wei W, He D, He B, Tang Q (2013) Photocatalytic oxidation for degradation of VOCs. Open J Inorg Chem 3:14–25. https://doi.org/10.4236/ojic.2013.31003
Liu H, Zhang H, Yang H (2014) Photocatalytic removal of nitric oxide by multi-walled carbon nanotubess-upported TiO2. Chin J Catal 35:66–77. https://doi.org/10.1016/S1872-2067(12)60705-0
Martinez T, Bertron A, Ringot E, Escadeillas G (2011) Degradation of NO using photocatalytic coatings applied to different substrates. Build Environ 46(9):1808–1816. https://doi.org/10.1016/j.buildenv.2011.03.001
Mei GM, Chai SP, Xu BQ, Mohamed AR (2014) Enhanced visible light responsive MWCNT/TiO2 core–shell nanocomposites as the potential photocatalyst for reduction of CO2 into methane. Sol Energy Mater Sol Cells 122:183–189. https://doi.org/10.1016/j.solmat.2013.11.034
Meng T, Wang M, Jiang F, Yu J, Hu Y, Wu K (2014) Photocatalytic property of Ag modified nano-TiO2/carbon nanotube composites for NO2 degradation under visible light. Mater Res Innov 18:691–695. https://doi.org/10.1179/1432891714Z.000000000576
Mohamed YMA, Attia YA (2020) The influence of ultrasonic irradiation on catalytic performance of ZnO nanoparticles toward the synthesis of chiral 1-substituted-1H -tetrazolederivatives from α -amino acid ethyl esters. Appl Organomet Chem. https://doi.org/10.1002/aoc.5758
O’Regan B, Gratzel M (1991) A low cost, high-efficiency solar cell based on dyesensitized colloidal TiO2 films. Nature 353:737–739. https://doi.org/10.1038/353737a0
Peerakiatkhajorn P, Chawengkijwanich C, Onreabroy W, Chiarakorn S (2012) Novel photocatalytic Ag/TiO2 thin film on polyvinyl chloride for gaseous BTEX treatment. Mater Sci Forum 712:133–145. https://doi.org/10.4028/www.scientific.net/MSF.712.133
Reddy KM (2018) Photocatalytic degradation of organic dyes using Ag-TiO2 nanomaterials. J Chem Pharm Res 10(2):128–133
Sheng Z, Wu Z, Liu Y, Wang H (2008) Gas-phase photocatalytic oxidation of NO over palladium modified TiO2 catalysts. Catal Commun 9:1941–1944. https://doi.org/10.1016/j.catcom.2008.03.022
Trabelsi K, Hajjaji A, Ka I, Gaidi M, Bessais B, El Khakani MA (2017) Optoelectronic and photocatalytic properties of in situ platinum-doped TiO2 films deposited by means of pulsed laser ablation technique. J Mater Sci Mater Electron 28:3317–3324. https://doi.org/10.1007/s10854-016-5925-z
Tseng YH, Huang BK (2012) Photocatalytic degradation of NOx using Ni-containing TiO2. Int J Photoenergy 2012:1–7 (Article ID 832180. https://doi.org/10.1155/2012/832180
Wu JCS, Cheng YTJ (2006) In situ FTIR study of photocatalytic NO reaction on photocatalysts under UV irradiation. J Catal 237(2):393–404. https://doi.org/10.1016/j.jcat.2005.11.023
Wu Z, Sheng Z, Liu Y, Wang H, Tang N, Wang J (2009) Characterization and activity of Pd-modified TiO2 catalysts for photocatalytic oxidation of NO in gas phase. J Hazard Mater 164:542–548. https://doi.org/10.1016/j.jhazmat.2008.08.028
Xing M, Zhang J, Chen F (2009) New approaches to prepare nitrogen-doped TiO2 photocatalytsts and study on their photocatalytic activities in visible light. Appl Catal B 89:563–569. https://doi.org/10.1016/j.apcatb.2009.01.016
Yan W, Jiwei Z, Zhenshen J, Shunli Z (2007) Visible light photocatalytic decoloration of methylene blue on novel N doped TiO2. Chin Sci Bull 52:2157–2160. https://doi.org/10.1007/s11434-007-0306-x
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Sami Rtimi
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Abdelsalam, E.M., Mohamed, Y.M.A., Abdelkhalik, S. et al. Photocatalytic oxidation of nitrogen oxides (NOx) using Ag- and Pt-doped TiO2 nanoparticles under visible light irradiation. Environ Sci Pollut Res 27, 35828–35836 (2020). https://doi.org/10.1007/s11356-020-09649-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-09649-5