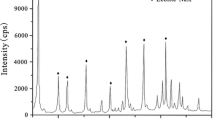
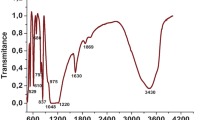
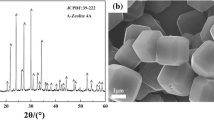
Abstract
Zeolite can remove the heavy metals in wastewater, but the removal efficiency was determined by the types of zeolites and the treatment conditions. In this study, a kind of zeolite NaX synthesized from the coal gangue, a by-product of coal production, was used and the removal efficiency of Cd2+ and Cu2+and the kinetic models were studied. The effects of its dosage, initial pH value of wastewater, and adsorption temperature on its adsorption of heavy metals Cd2+ and Cu2+ in the simulated wastewater were studied through the indoor experiments in laboratory, and the adsorption mechanism was analyzed by the adsorption kinetic model based on its adsorption efficiency and its structures. The results show that the zeolite NaX synthesized from coal gangue has a good adsorption effect on Cd2+ and Cu2+. The adsorption reaches the best effect when the dosage of zeolite is 2 g/L, the initial pH of simulated wastewater is 5, the adsorption temperature is room temperature (25 ℃), and the removal efficiency of Cd2+ and Cu2+ (100 mg/L) is more than 90%. Additionally, the Langmuir, Freundlich, and Temkin isothermal adsorption models were used to compare and analyze the adsorption effects. The equilibrium data was better fitted by the Langmuir model with the maximum adsorption capacities of 100.11 mg/g (Cd2+) and 95.29 mg/g (Cu2+), and both separation coefficients were 0–1, which shows that the process was the favorable adsorption. Weber Morris diffusion model shows that the adsorption process at 120 min was more consistent with the pseudo-second-order kinetics model, and the adsorption efficiency was simultaneously controlled by the liquid diffusion step and intraparticle diffusion step. Moreover, the removal mechanism of Cd2+ and Cu2+ mainly includes physical adsorption and ion exchange. Therefore, the adsorption effect of zeolite synthesized from coal gangue is remarkable, which will provide a feasible and potential alternative for its resource application.







Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Ansari M, Raisi A, Aroujalian A, Dabir B, Irani M (2015) Synthesis of nano-NaX zeolite by microwave heating method for removal of lead, copper, and cobalt ions from aqueous solution. J Environ Eng 141(5):04014088. https://doi.org/10.1061/(ASCE)EE.1943-7870.0000919
Bessaha G, Bessaha F, Bendenia S, Khelifa A (2022) Exchanged zeolite adsorbent for removing Cr (VI): Kinetics, thermodynamics and adsorption mechanism. Int J Environ Anal Chem 1-19.https://doi.org/10.1080/03067319.2021.2006193
Bu N, Liu X, Song S, Liu J, Yang Q, Li R, Zheng F, Yan L, Zhen Q, Zhang J (2020) Synthesis of NaY zeolite from coal gangue and its characterization for lead removal from aqueous solution. Adv Powder Technol 31(7):2699–2710. https://doi.org/10.1016/j.apt.2020.04.035
Chen J, Lu X (2018) Equilibrium and kinetics studies of Cd (II) sorption on zeolite NaX synthesized from coal gangue. J Water Reuse Desalination 8(1):94–101. https://doi.org/10.2166/wrd.2016.137
Chen J, Lu L, Zhang M, Xu X, Zhao N (2014) Removal of Co2+, Cu2+, Cd2+ and Cr3+ from aqueous solution by zeolite X prepared from coal gangue. Chin J Environ Eng 8(9):3625–3632 (in Chinese) CNKI:SUN:HJJZ.0.2014-09-017
Cheng T, Chen C, Tang R, Han C, Tian Y (2018) Competitive adsorption of Cu, Ni, Pb, and Cd from aqueous solution onto fly ash-based linde F (K) Zeolite. Iranian J Chem Chem Eng (IJCCE) 37(1):61–72. https://doi.org/10.30492/IJCCE.2018.31971
Collins F, Rozhkovskaya A, Outram J, Millar G (2020) A critical review of waste resources, synthesis, and applications for Zeolite LTA. Microporous Mesoporous Mater 291:109667. https://doi.org/10.1016/j.micromeso.2019.109667
Cong X, Lu S, Yao Y, Wang Z (2016) Fabrication and characterization of self-ignition coal gangue autoclaved aerated concrete. Mater Des 97:155–162. https://doi.org/10.1016/j.matdes.2016.02.068
Du H, Ma L, Liu X, Zhang F, Yang X, Wu Y, Zhang J (2018) A novel mesoporous SiO2 material with MCM-41 structure from coal gangue: preparation, ethylenediamine modification, and adsorption properties for CO2 capture. Energy Fuels 32(4):5374–5385. https://doi.org/10.1021/acs.energyfuels.8b00318
Gao Y, Zhang J (2020) Chitosan modified zeolite molecular sieve particles as a filter for ammonium nitrogen removal from water. Int J Mol Sci 21(7):2383. https://doi.org/10.3390/ijms21072383
Ge Q, Moeen M, Tian Q, Xu J, Feng K (2020) Highly effective removal of Pb2+ in aqueous solution by Na-X zeolite derived from coal gangue. Environ Sci Pollut Res 27(7):7398–7408. https://doi.org/10.1007/s11356-019-07412-z
Guo Y, Zhang Y, Cheng F (2014) Industrial development and prospect about comprehensive utilization of coal gangue. CIESC J 65(7):2443–2453. https://doi.org/10.3969/j.issn.0438-1157.2014.07.006 (in Chinese)
Hameed B, Rahman A (2008) Removal of phenol from aqueous solutions by adsorption onto activated carbon prepared from biomass material. J Hazard Mater 160(2–3):576–581. https://doi.org/10.1016/j.jhazmat.2008.03.028
Jin Y, Li L, Liu Z, Zhu S, Wang D (2021) Synthesis and characterization of low-cost zeolite NaA from coal gangue by hydrothermal method. Adv Powder Technol 32(3):791–801. https://doi.org/10.1016/j.apt.2021.01.024
Li H, Zheng F, Wang J, Zhou J, Huang X, Chen L, Hu P, Gao J, Zhen Q, Bashir S, Liu J (2020) Facile preparation of zeolite-activated carbon composite from coal gangue with enhanced adsorption performance. Chem Eng J 390:124513. https://doi.org/10.1016/j.cej.2020.124513
Li H, Cheng R, Liu Z, Du C (2019) Waste control by waste: Fenton–like oxidation of phenol over Cu modified ZSM-5 from coal gangue. Sci Total Environ 683:638–647. https://doi.org/10.1016/j.scitotenv.2019.05.242
Liang Z, Gao Q, Liu H, Gao H (2020) Synthesis of NaX zeolite from coal gangue and its adsorption capability for Cd2+. J Southeast Univ (Natural Science Edition) 50(4):741–747. https://doi.org/10.3969/j.issn.1001-0505.2020.04.019 (in Chinese)
Liu H, Liu Z (2010) Recycling utilization patterns of coal mining waste in China. Resour Conserv Recycl 54(12):1331–1340. https://doi.org/10.1016/j.resconrec.2010.05.005
Lu X, Shi D, Chen J (2017) Sorption of Cu2+ and Co2+ using zeolite synthesized from coal gangue: isotherm and kinetic studies. Environ Earth Sci 76(17):591. https://doi.org/10.1007/s12665-017-6923-z
Qian T, Li J (2015) Synthesis of Na-A zeolite from coal gangue with the in-situ crystallization technique. Adv Powder Technol 26(1):98–104. https://doi.org/10.1016/j.apt.2014.08.010
Rad LR, Anbia M (2021) Zeolite-based composites for the adsorption of toxic matters from water: a review. J Environ Chem Eng 9(5):106088. https://doi.org/10.1016/j.jece.2021.106088
Sethia G, Somani R, Bajaj H (2014) Sorption of methane and nitrogen on cesium exchanged zeolite-X: structure, cation position and adsorption relationship. Ind Eng Chem Res 53(16):6807–6814. https://doi.org/10.1021/ie5002839
Tasić ŽZ, Bogdanović GD, Antonijević MM (2019) Application of natural zeolite in wastewater treatment: a review. J Min Metall: Mining 55(1):67–79. https://doi.org/10.5937/JMMA1901067T
Tseng R, Wu F (2008) Inferring the favorable adsorption level and the concurrent multi-stage process with the Freundlich constant. J Hazard Mater 155(1–2):277–287. https://doi.org/10.1016/j.jhazmat.2007.11.061
Wen J, Dong H, Zeng G (2018) Application of zeolite in removing salinity/sodicity from wastewater: a review of mechanisms, challenges and opportunities. J Clean Prod 197:1435–1446. https://doi.org/10.1016/j.jclepro.2018.06.270
Xu W, Li L, Grace J (2010) Zinc removal from acid rock drainage by clinoptilolite in a slurry bubble column. Appl Clay Sci 50(1):158–163. https://doi.org/10.1016/j.clay.2010.07.005
Zhang X, Li C, Zheng S, Di Y, Sun Z (2022) A review of the synthesis and application of zeolites from coal-based solid wastes. Int J Miner Metall Mater 29(1):1–21. https://doi.org/10.1007/s12613-021-2256-8
Zhang X, Pan Y (2022) Preparation, properties and application of gel materials for coal gangue control. Energies 15(2):557. https://doi.org/10.3390/en15020557
Zhang Y, Ling TC (2020) Reactivity activation of waste coal gangue and its impact on the properties of cement-based materials–a review. Constr Build Mater 234:117424. https://doi.org/10.1016/j.conbuildmat.2019.117424
Zhao Y (2016) Review of the natural, modified, and synthetic zeolites for heavy metals removal from wastewater. Environ Eng Sci 33(7):443–454. https://doi.org/10.1089/ees.2015.0166
Zhou J, Zheng F, Li H, Wang J, Bu N, Hu P, Gao J, Zhen Q, Bashir S, Liu J (2020) Optimization of post-treatment variables to produce hierarchical porous zeolites from coal gangue to enhance adsorption performance. Chem Eng J 381:122698. https://doi.org/10.1016/j.cej.2019.122698
Acknowledgements
We gratefully acknowledge the National and Local Unified Engineering Research Center for Basalt Fiber Production and Application Technology, International Institute for Urban Systems Engineering, Southeast University.
Funding
This research was funded by the National Natural Science Foundation of Jiangsu Province in China (Grant number BK20193048), and the National Key Research and Development Program of China (Grant number 2017YFC0504505).
Author information
Authors and Affiliations
Contributions
Methodology, Q. G.; experiment, Z. L. and Q. G.; formal analysis, Z. L. and H. G.; data curation, Z. L. and Q.G.; writing—original draft preparation, Z. L. and Q. G.; writing—review and editing, H. G. and Z. W.; supervision, H. G. and Z. W.; project administration, H. G. and Z. W.; funding acquisition, Z. L. and H. G. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not application.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liang, Z., Gao, Q., Wu, Z. et al. Removal and kinetics of cadmium and copper ion adsorption in aqueous solution by zeolite NaX synthesized from coal gangue. Environ Sci Pollut Res 29, 84651–84660 (2022). https://doi.org/10.1007/s11356-022-21700-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-21700-1