Abstract
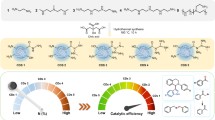
The present chain of five papers considers the concept of solar-to-chemical energy conversion using TiO2-based semiconductors. The series reports the effect of chromium on the key performance-related properties of polycrystalline TiO2 (rutile), including electronic structure, photocatalytic activity, intrinsic defect disorder, electrochemical coupling and surface versus bulk properties. In this work, we show that the effect of chromium on photocatalytic performance of TiO2 depends on its elemental content and the related defect disorder that is determined by oxygen activity in the oxide lattice. At high oxygen activity, chromium leads to enhanced photocatalytic performance only for dilute solid solutions (up to 0.04–0.043 at.% Cr). Higher chromium content results in a decrease of photocatalytic activity below that for pure TiO2, despite the observed substantial decrease of the band gap. The photocatalytic performance of Cr-doped TiO2 annealed in reducing conditions is low within the entire studied range of compositions. The obtained results led to derivation of a theoretical model representing the mechanism of the light-induced reactivity of TiO2 with water and the related charge transfer. The photocatalytic performance is considered in terms of a competitive effect of several key performance-related properties. The performance is predominantly influenced by the concentration of titanium vacancies acting as reactive surface sites related to anodic charge transfer.

ᅟ
















Similar content being viewed by others
Notes
The species in the solid phase are represented according to the Kroger-Vink notation [39]
References
Bak T, Li W, Nowotny J, Atanacio AJ, Davis J (2015) Photocatalytic properties of TiO2: evidence of the key role of surface active sites in water oxidation. JPhys Chem A 119(36):9465–9473. https://doi.org/10.1021/acs.jpca.5b05031
Rahman KA, Bak T, Atanacio A, Ionescu M, Nowotny J (2017) Toward sustainable energy: photocatalysis of Cr-doped TiO2: 2. effect of defect disorder, Ionics, Part 1, https://doi.org/10.1007/s11581-017-2370-9
Li W, Bak T, Atanacio A, Nowotny J (2016) Photocatalytic properties of TiO2: effect of niobium and oxygen activity on partial water oxidation. Appl Catal B 198:243–253
Alim MA, Bak T, Atanacio AJ, Ionescu M, Kennedy B, Price WS, Plessis JD, Pourmahdavi M, Zhou M, Torres A, Nowotny J (2017) Photocatalytic properties of Ta-doped TiO2. Int J Ionics 23(12):3517–3531. https://doi.org/10.1007/s11581-017-2162-2
Bak T, Nowotny J, Sucher NJ, Wachsman E (2011) Effect of crystal imperfections on reactivity and photoreactivity of TiO2(Rutile) with oxygen, water, and bacteria. J Phys Chem C 115(32):15711–15738. https://doi.org/10.1021/jp2027862
Nowotny J, Bak T, Nowotny M, Sheppard L (2007) Titanium dioxide for solar-hydrogen II. Defect chemistry. Int J Hydrogen Energ 32:2630–2643
Bechstein R, Kitta M, Schütte J, Kühnle A, Onishi H (2009) Evidence for vacancy creation by chromium doping of rutile titanium dioxide (110). J Phys Chem C 113(8):3277–3280. https://doi.org/10.1021/jp8095677
Jaimy KB, Ghosh S, Sankar S, Warrier K (2011) An aqueous sol–gel synthesis of chromium(III) doped mesoporous titanium dioxide for visible light photocatalysis. Mater Res Bull 46(6):914–921. https://doi.org/10.1016/j.materresbull.2011.02.030
Gong J, Pu W, Yang C, Zhang J (2012) A simple electrochemical oxidation method to prepare highly ordered Cr-doped titania nanotube arrays with promoted photoelectrochemical property. Electrochim Acta 68(2012):178–183. https://doi.org/10.1016/j.electacta.2012.02.049
Li X, Guo Z, He T (2013) The doping mechanism of Cr into TiO2 and its influence on the photocatalytic performance. Phys Chem Chem Phys 15(46):20037–20045. https://doi.org/10.1039/c3cp53531b
López R, Gómez R, Oros-Ruiz S (2011) Photophysical and photocatalytic properties of TiO2-Cr sol–gel prepared semiconductors. Catal Today 166(1):159–165. https://doi.org/10.1016/j.cattod.2011.01.010
Tian B, Li C, Zhang J (2012) One-step preparation, characterization and visible-light photocatalytic activity of Cr-doped TiO2 with anatase and rutile bicrystalline phases. Chem Eng J 191:402–440. https://doi.org/10.1016/j.cej.2012.03.038
Mardare D, Iacomi F, Cornei N, Girtan M, Luca D (2010) Undoped and Cr-doped TiO2 thin films obtained by spray pyrolysis. Thin Solid Films 518(16):4586–4589. https://doi.org/10.1016/j.tsf.2009.12.037
Diaz-Uribe C, Vallejo W, Ramos W (2014) Methylene blue photocatalytic mineralization under visible irradiation on TiO2 thin films doped with chromium. Appl Surf Sci 319:121–127. https://doi.org/10.1016/j.apsusc.2014.06.157
Zhu J, Deng Z, Chen F, Zhang J, Chen H, Anpo M, Huang J, Zhang L (2006) Hydrothermal doping method for preparation of Cr3+-TiO2 photocatalysts with concentration gradient distribution of Cr3+. Appl Catal B 62:329–335
Peng Y-H, Huang G-F, Huang W-Q (2012) Visible-light absorption and photocatalytic activity of Cr-doped TiO2 nanocrystal films. Adv Powder Technol 23:8–12
Choi J, Park H, Hoffmann MR (2010) Effects of single metal-ion doping on the visible-light photoreactivity of TiO2. J Phys Chem C 114(2):783–792. https://doi.org/10.1021/jp908088x
Wilke K, Breuer H (1999) The influence of transition metal doping on the physical and photocatalytic properties of titania. J Photoch Photobio A 121:49–53
Ghasemi S, Rahimnejad S, Setayesh SR, Rohani S, Gholami M (2009) Transition metal ions effect on the properties and photocatalytic activity of nanocrystalline TiO2 prepared in an ionic liquid. J Hazard Mater 172(2-3):1573–1578. https://doi.org/10.1016/j.jhazmat.2009.08.029
Jimmy CY, Li G, Wang X, Hu X, Leung CW, Zhang Z (2006) An ordered cubic Im3m mesoporous Cr–TiO2 visible light photocatalyst. Chem Commun 2717–2719. https://doi.org/10.1039/B603456J
Koh PW, Hatta MHM, Ong ST, Yuliati L, Lee SL (2017) Photocatalytic degradation of photosensitizing and non-photosensitizing dyes over chromium doped titania photocatalysts under visible light. J Photochem Photobiol A 332:215–223
Kohler K, Schlapfer CW, Vonzelewsky A, Nickl J, Engweiler J, Baiker A (1993) Chromia supported on titania. J Catal 143(1):201–214. https://doi.org/10.1006/jcat.1993.1266
Michalow KA, Otal EH, Burnat D, Fortunato G, Emerich H, Ferri D, Heel A, Graule T (2013) Flame-made visible light active TiO2: Cr photocatalysts: correlation between structural, optical and photocatalytic properties. Catal Today 209:47–53. https://doi.org/10.1016/j.cattod.2012.10.007
Trenczek-Zajac A, Radecka M, Jasinski M, Michalow K, Rekas M, Kusior E, Zakrzewska K, Heel A, Graule T, Kowalski K (2009) Influence of Cr on structural and optical properties of TiO2:Cr nanopowders prepared by flame spray synthesis. J Power Sources 194(1):104–111. https://doi.org/10.1016/j.jpowsour.2009.02.068
Hamadanian M, Reisi-Vanani A, Majedi A (2010) Synthesis, characterization and effect of calcination temperature on phase transformation and photocatalytic activity of Cu,S-codoped TiO2 nanoparticles. Appl Surf Sci 256(6):1837–1844. https://doi.org/10.1016/j.apsusc.2009.10.016
Vieira FTG, Melo DS, de Lima SJG, Longo E, Paskocimas CA, Júnior WS, de Souza AG, dos Santos IMG (2009) The influence of temperature on the color of TiO2:Cr pigments. Mater Res Bull 44(5):1086–1092. https://doi.org/10.1016/j.materresbull.2008.10.007
Li Y, Wlodarski W, Galatsis K, Moslih SH, Cole J, Russo S, Rockelmann N (2002) Gas sensing properties of p-type semiconducting Cr-doped TiO2 thin films. Sensor Actuat B-Chem 83:160–163
Escudero A, Langenhorst F (2012) Chromium incorporation into TiO2 at high pressure. J Solid State Chem 190:61–67
Belaya E, Viktorov V (2008) Formation of solid solutions in the TiO2-Cr2O3 system. Inorg Mater 44(1):62–66. https://doi.org/10.1134/S002016850801010X
Sōmiya S, Hirano S (1978) Kamiya S (1978) Phase relations of the Cr2O3-TiO2 system. J Solid State Chem 25(3):273–284. https://doi.org/10.1016/0022-4596(78)90112-3
Zakrzewska K, Radecka M, Rekas M (1997) Effect of Nb, Cr, Sn additions on gas sensing properties of TiO2 thin films. Thin Solid Films 310(1-2):161–166. https://doi.org/10.1016/S0040-6090(97)00401-X
Venezia AM, Palmisano L, Schiavello M (1995) Structural changes of titanium oxide induced by chromium addition as determined by an X-ray diffraction study. J Solid State Chem 114:364–368
Sikora I, Stolze F, Hirschwald W (1987) Segregation of chromium in CoO- Cr2O3 solid solutions and CoCr2O4 spinel phases studied by SIMS and ESCA. Surf Interface Anal 10:424–429
Zhang J, Zhou P, Liu J, Yu J (2014) New understanding of the difference of photocatalytic activity among anatase, rutile and brookite TiO2. Phys Chem Chem Phys 16(38):20382–20386. https://doi.org/10.1039/C4CP02201G
Xu M, Gao Y, Moreno EM, Kunst M, Muhler M, Wang Y, Idriss H, Wöll C (2011) Photocatalytic activity of bulk TiO2 anatase and rutile single crystals using infrared absorption spectroscopy. Phys Rev Lett 106(13):138302. https://doi.org/10.1103/PhysRevLett.106.138302
Umebayashi T, Yamaki T, Itoh H, Asai K (2002) Analysis of electronic structures of 3d transition metal-doped TiO 2 based on band calculations. J Phys Chem Solids 63(10):1909–1920. https://doi.org/10.1016/S0022-3697(02)00177-4
Shi J, Leng W, Zhu W, Zhang J, Cao C (2006) Electrochemically assisted photocatalytic oxidation of nitrite over Cr-doped TiO2 under visible light. Chem Eng Tech 29(1):146–154. https://doi.org/10.1002/ceat.200500236
Nowotny J, Macyk W, Wachsman E, Rahman KA (2016) Effect of oxygen activity on the n–p transition for pure and Cr-doped TiO2. J Phys Chem C 120(6):3221–3228. https://doi.org/10.1021/acs.jpcc.5b12101
Kröger F, Vink H (1956) Relations between the concentrations of imperfections in crystalline solids. Solid State Phys 3:307–435. https://doi.org/10.1016/S0081-1947(08)60135-6
Stoneham AM (1980) Theory of defect processes. Phys Today 33:34–37
Author information
Authors and Affiliations
Corresponding author
Additional information
New insight
1. Reduction of the band gap has little effect on photocatalytic performance of Cr-doped TiO2.
2. The photocatalytic activity of Cr-doped TiO2 formed in oxidising conditions changes with composition. The photocatalytic performance increases within dilute solid solutions, up to 0.04 at.%, and decreases at higher chromium content.
3. The effect of chromium on photocatalytic activity may be considered in terms of a competitive effect of the key performance-related properties.
Highlights
1. The incorporation of chromium into the TiO2 lattice results in an increase of photocatalytic activity only for dilute solid solutions (up to 0.04 at% Cr) annealed in oxidising conditions.
2. Chromium results in a decrease of photocatalytic activity when annealed in reducing conditions.
3. Photocatalytic activity of Cr-TiO2 depends on several defect-related properties that have competitive effect on performance.
Rights and permissions
About this article
Cite this article
Rahman, K.A., Bak, T., Atanacio, A. et al. Toward sustainable energy: photocatalysis of Cr-doped TiO2: 2. effect of defect disorder. Ionics 24, 327–341 (2018). https://doi.org/10.1007/s11581-017-2370-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-017-2370-9