Abstract
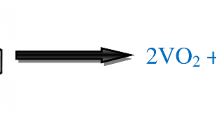
Molybdenum (Mo)-doped vanadium dioxide (\(\hbox {VO}_{2}\)(B)) nanobelts were successfully synthesized using commercial vanadium pentoxide (\(\hbox {V}_{2}\hbox {O}_{5}\)) as the starting material and ammonium molybdate as the dopant by a simple hydrothermal route. Then, Mo-doped \(\hbox {VO}_{2}\)(B) nanobelts were transformed to Mo-doped \(\hbox {V}_{2}\hbox {O}_{5}\) nanobelts by calcination at \(400{^{\circ }}\hbox {C}\) under an air atmosphere. The samples were characterized by X-ray powder diffraction, energy-dispersive X-ray spectrometer, elemental mapping, X-ray photoelectron spectroscopy, X-ray fluorescence and transmission electron microscopy techniques. The results showed that Mo-doped \(\hbox {VO}_{2}\)(B) and \(\hbox {V}_{2}\hbox {O}_{5}\) solid solution with high purity were obtained. The electrochemical properties of Mo-doped \(\hbox {VO}_{2}\)(B) and \(\hbox {V}_{2}\hbox {O}_{5}\) nanobelts as supercapacitor electrodes were measured using cyclic voltammetry (CV) and galvanostatic charge–discharge (GCD). The specific capacitance of \(\hbox {VO}_{2}\)(B) nanobelts slightly declines with Mo doping, however, the specific capacitance of \(\hbox {V}_{2}\hbox {O}_{5}\) nanobelts greatly improves with Mo doping. Mo-doped \(\hbox {V}_{2}\hbox {O}_{5}\) nanobelts exhibit the specific capacitance as high as 526 F \(\hbox {g}^{-1}\) at the current density of 1 A \(\hbox {g}^{-1}\). Both CV and GCD curves show that they have good rate capability and retain 464, 380, 324 and 273 F \(\hbox {g}^{-1}\) even at a high-current density of 2, 5, 10 and 20 A \(\hbox {g}^{-1}\), respectively. It turns out that Mo-doped \(\hbox {V}_{2}\hbox {O}_{5}\) nanobelts are ideal materials for supercapacitor electrodes in the present work.










Similar content being viewed by others
References
Yu Z, Tetard L, Zhai L and Thomas J 2015 Energy Environ. Sci. 8 702
Zhong C, Deng Y, Hu W, Qiao J, Zhang L and Zhang J 2015 Chem. Soc. Rev. 44 7484
Zhang Y and Meng C 2015 Mater. Lett. 160 404
Yu M, Qiu W, Wang F, Zhai T, Fang P, Lu X et al 2015 J. Mater. Chem. A 3 15792
Zhang Y, Zheng J, Wang Q, Hu T and Meng C 2016 RSC Adv. 6 93741
Jing X, Wang C, Feng W, Xing N, Jiang H, Lu X et al 2018 R. Soc. Open Sci. 5 171768
Zhang Y 2018 Chem. Sel. 3 1577
Xia X, Zhang Y, Chao D, Guan C, Zhang Y, Li L et al 2014 Nanoscale 6 5008
Zhang L L and Zhao X S 2009 Chem. Soc. Rev. 38 2520
Zheng J, Zhang Y, Wang N, Zhao Y, Tian F and Meng C 2016 Mater. Lett. 171 240
Singh A and Chandra A 2015 Sci. Rep. 5 15551
Trasatti S and Kurzweil I 1994 Platinum Met. Rev. 38 46
Perera S D, Rudolph M, Mariano R G, Nijem N, Ferraris J P, Chabal Y J et al 2013 Nano Energy 2 966
Wang Q, Zhang Y, Zheng J, Wang Y, Hu T and Meng C 2017 Dalton Trans. 46 4303
Umeshbabu E and Ranga Rao G 2016 J. Colloid Interface Sci. 472 210
Mu J, Wang J, Hao J, Cao P, Zhao S, Zeng W et al 2015 Ceram. Int. 41 12626
Peng T, Wang J, Liu Q, Liu J and Wang P 2015 CrystEngComm 17 1673
Guo Y, Li J, Chen M and Gao G 2015 J. Power Sources 273 804
Wang N, Zhang Y, Hu T, Zhao Y and Meng C 2015 Curr. Appl. Phys. 15 493
Zhang Y and Huang Y 2016 Mater. Lett. 182 285
Liang L, Liu H and Yang W 2013 J. Alloys Compd. 559 167
Xu L, Zhang Y, Deng Y, Zhong Y, Mo S, Cheng G et al 2013 Mater. Res. Bull. 48 3620
Zhang Y, Huang Y, Zhang J, Wu W, Niu F, Zhong Y et al 2012 Mater. Res. Bull. 47 1978
Zhang Y, Fan M, Zhou M, Huang C, Chen C, Cao Y et al 2012 Bull. Mater. Sci. 35 369
Xu L, Zhang Y, Zhang X, Huang Y, Tan X, Huang C et al 2014 Bull. Mater. Sci. 37 1397
Zhang Y, Zheng J, Wang Q, Zhang S, Hu T and Meng C 2017 Appl. Surf. Sci. 423 728
Zhang Y, Jing X, Wang Q, Zheng J, Jiang H and Meng C 2017 Dalton Trans. 46 15048
Zheng J, Zhang Y, Wang Q, Jiang H, Liu Y, Lv T et al 2018 Dalton Trans. 47 452
Hu T, Liu Y, Zhang Y, Nie Y, Zheng J, Wang Q et al 2018 Microporous Mesoporous Mater. 262 199
Zhu J, Cao L, Wu Y, Gong Y, Liu Z, Hoster H E et al 2013 Nano Lett. 13 5408
Zhang Y, Zheng J, Zhao Y, Hu T, Gao Z and Meng C 2016 Appl. Surf. Sci. 377 385
Saravanakumar B, Purushothaman K K and Muralidharan G 2012 ACS Appl. Mater. Interfaces 4 4484
Zhang Y, Zheng J, Hu T, Tian F and Meng C 2016 Appl. Surf. Sci. 371 189
Yang J, Lan T, Liu J, Song Y and Wei M 2013 Electrochim. Acta 105 489
Wee G, Soh H Z, Cheah Y L, Mhaisalkar S G and Srinivasan M 2010 J. Mater. Chem. 20 6720
Zhang Y, Zheng J, Wang Q, Hu T, Tian F and Meng C 2017 Appl. Surf. Sci. 399 151
Zhi M, Xiang C, Li J, Li M and Wu N 2013 Nanoscale 5 72
Qian T, Xu N, Zhou J, Yang T, Liu X, Shen X et al 2015 J. Mater. Chem. A 3 488
Wu Y, Gao G and Wu G 2015 J. Mater. Chem. A 3 1828
Jeyalakshmi K, Vijayakumar S, Purushothaman K K and Muralidharan G 2013 Mater. Res. Bull. 48 2578
Zheng J, Zhang Y, Jing X, Wang Q, Hu T, Xing N et al 2017 Mater. Chem. Phys. 166 5
Zhang Y, Chen C, Wu W, Niu F, Liu X, Zhong Y et al 2013 Ceram. Int. 39 129
Theobald F, Cabala R and Bernard J 1976 J. Solid State Chem. 17 431
Zhang Y, Li W, Fan M, Zhang F, Zhang J, Liu X et al 2012 J. Alloys Compd. 544 30
Enjalbert R and Galy J 1986 Acta Crystallogr. C 42 1467
Zhang Y, Zhang J, Zhang X, Mo S, Wu W, Niu F et al 2013 J. Alloys Compd. 570 104
Silversmit G, Depla D, Poelman H, Marin G B and Gryse R D 2004 J. Electron Spectrosc. 135 167
Wagner C D, Riggs W M, Davis L E and Moulder J F 1979 Handbook of X-ray photoelectrom spectroscopy (Minnesota: Perkin-Elmer Corporation)
Tang C, Georgopoulos P, Fine M E, Cohen J B, Nygren M, Knapp G S et al 1985 Phys. Rev. B 31 1000
Manning T D, Parkin I P, Blackman C and Qureshi U 2005 J. Mater. Chem. 15 4560
Zhang Y, Zhang J, Zhang X, Deng Y, Zhong Y, Huang C et al 2013 Ceram. Int. 39 8363
Qi J L, Wang X, Lin J H, Zhang F, Feng J C and Fei W D 2015 J. Mater. Chem. A 3 12396
Delmas C, Cognac-Auradou H, Cocciantelli J M, Menetrier M and Doumerc J P 1994 Solid State Ion. 69 257
Deng L, Zhang G, Kang L, Lei Z, Liu C and Liu Z-H 2013 Electrochim. Acta 112 448
Lao Z J, Konstantinov K, Tournaire Y, Ng S H, Wang G X and Liu H K 2006 J. Power Sources 162 1451
Lin Z, Yan X, Lang J, Wang R and Kong L-B 2015 J. Power Sources 279 358
Chen Z, Qin Y, Weng D, Xiao Q, Peng Y, Wang X et al 2009 Adv. Funct. Mater. 19 3420
Reddy R N and Reddy R G 2006 J. Power Sources 156 700
Lala N L, Jose R, Yusoff M M and Ramakrishna S 2012 J. Nanopart. Res. 14 1201
Cao L, Zhu J, Li Y, Xiao P, Zhang Y, Zhang S et al 2014 J. Mater. Chem. A 2 13136
Jeyalakshmi K, Vijayakumar S, Nagamuthu S and Muralidharan G 2013 Mater. Res. Bull. 48 760
Acknowledgements
This work was partially supported by the National Natural Science Foundation of China (Grant Nos. 21601026 and 21771030) and Doctoral Research Foundation of Liaoning Province (201601035).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, Y., Zhang, Y. Influence of the electrochemical properties of vanadium oxides on specific capacitance by molybdenum doping. Bull Mater Sci 42, 37 (2019). https://doi.org/10.1007/s12034-018-1693-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-018-1693-0