Abstract
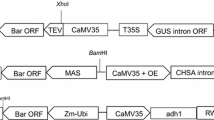
Genetic engineering provides new opportunities for improving economically important traits in sugarcane cultivars. In this study, an efficient Agrobacterium-mediated transformation system that uses the bar gene (a herbicide resistance gene that is used in conjunction with the herbicide Basta) as a selection marker was developed. Using this transformation selection system, all of the resistant plants after selection were nearly 100% polymerase chain reaction (PCR) detection positive and showed herbicide resistance. Each gram of sugarcane calli used for transformation produced approximately 12 transgenic lines. It took approximately 4 months to generate transgenic plants that measured 10 cm in height for greenhouse transplantation.









Similar content being viewed by others
Abbreviations
- MS:
-
Murashige and Skoog medium
- GFP:
-
Green fluorescent protein
- 2,4-D:
-
2,4-Dichlorophenoxyacetic acid
- 6-BA:
-
6-Benzylaminopurine
- NAA:
-
1-Naphthaleneacetic acid
- AD2:
-
Synthetic Medicago truncatula Defensins4
- AD3:
-
Native Medicago truncatula Defensins4 with KDEL
- AD4:
-
Nynthetic Medicago truncatula Defensins4 with KDEL
- Ppase:
-
Inorganic Pyrophosphatase Gene
- SST:
-
Sucrose:sucrose 1-fructosyl transferase gene
References
Arencibia A, Molina P, Dela Riva G, Selman-Housein G (1995) Production of transgenic sugarcane (Saccharum officinarum L.) plants by intact cell electroporation. Plant Cell Rep 14(5):305–309
Arvinth S, Arun S, Selvakesavan RK, Srikanth J, Mukunthan N, Ananda Kumar P, Premachandran MN, Subramonian N (2010) Genetic transformation and pyramiding of aprotinin-expressing sugarcane with cry1Ab for shoot borer(Chilo infuscatellus) resistance. Plant Cell Rep 29(4):383–395
Basnayake SWV, Moyle R, Brich RG (2011) Embryogenic callus proliferation and regeneration conditions for genetic transformation of diverse sugarcane cultivars. Plant Cell Rep 30(3):439–448
Bower R, Brich RG (1992) Transgenic sugarcane plants via microprojectile bombardment. Plant J 2(3):409–416
Darbani B, Eimanifar A, Stewart CN, Camargo WN (2007) Methods to produce marker free transgenic plants. Biotechnol J 2(1):83–90
Dong SJ, Delucca P, Geijskes RJ, Ke J, Mayo K, Mai P, Sainz M, Caffall K, Moser T, Yarnall M, Setliff K, Jain R, Rawls E, Jones MS, Dunder E (2014) Advances in agrobacterium-mediated sugarcane transformation and stable transgene expression. Sugar Tech 16(4):366–371
Enríquez-Obregón GA, Vázquez-Padrón RI, Prieto-Samsonov DL, De la Riva GA, Selman-Housein G (1998) Herbicide resistance sugarcane (Saccharum officinarum L.) plants by agrobacterium-mediated transformation. Planta 206(1):20–27
Falco MC, Neto AT, Ulian EC (2000) Transformation and expression of a gene for herbicide resistance in a Brazilian sugarcane. Plant Cell Rep 19(12):1188–1194
Fromm ME, Morrish F, Armstrong C, Williams R, Thomas J, Klein TM (1990) Inheritance and expression of chimeric genes in the progeny of transgenic maize plants. Bio/Technology 8:833–838
Ingelbrecht IL, Irvine JE, Mirkov TE (1999) Posttranscriptional gene silencing in transgenic sugarcane: dissection of homology-dependent virus resistance in a monocot that has complex poly-ploid genome. Plant Physiol 119(4):1187–1197
Joyce P, Kuwahata M, Turner N, Lakshmanan P (2010) Selection system and co-cultivation medium are important determinants of Agrobacterium-mediated transformation of sugarcane. Plant Cell Rep 29(2):173–183
Kalunke RM, Kolge AM, Babu KH, Prasad DT (2009) Agrobacterium mediated transformation of sugarcane for borer resistance using Cry1Aa3 gene and one-step regeneration of transgenic plants. Sugar Tech 11(4):355–359
Manickavasagam M, Ganapathi A, Anbazhagan VR, Sudhakar B, Selvaraj N, Vasudevan A, Kasthurirengan S (2004) Agrobacterium-mediated genetic transformation and development of herbicide-resistant sugarcane (Saccharum species hybrids) using axillary buds. Plant Cell Rep 23(3):134–143
Mayavan S, Subramanyam K, Arun M, Rajesh M, Kapil Dev G, Sivanandhan G, Jaganath B, Manickavasagam M, Selvaraj N, Ganapathi A (2013) Agrobacterium tumefaciens-mediated in planta seed transformation strategy in sugarcane. Plant Cell Rep 32(10):1557–1574
Mudge RS, Osabe K, Casu RE, Bonnett GD, Manners J, Brich RG (2009) Efficient silencing of reporter transgenes coupled to known functional promoters in sugarcane, a highly polyploid crop species. Planta 229(3):549–558
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Plant Physiol 15(3):473–497
Priji PJ, Hemaprabha G (2015) Sugarcane specific drought responsive candidate genes belonging to ABA dependent pathway indentified from basic species clones of Saccharum sp. and Erianthus sp. Sugar Tech 17(2):130–137
Rani K, Sandhu SK, Gosal SS (2012) Genetic augmentation of sugarcane through direct gene transformation with OsglyII gene construct. Sugar Tech 14(3):229–236
Santosa DA, Hendroko R, Farouk A, Greiner R (2004) A rapid and highly efficient method for transformation of sugarcane callus. Mol Biotechnol 28(2):113–119
Snyman SJ (2004) Sugarcane transformation. Transgenic Crops World 2004:103–114
Srikanth K, Subramonian N, Premachandran MN (2011) Advances in transgenic research for insect resistance in sugarcane. Trop Plant Biol 4(1):52–61
Suprasanna P, Patade VY, Desai NS, Devarumath RM, Kawar PG, Pagariya MC, Canapathi A, Manickavasagam M, Badu KH (2011) Biotechnological developments in sugarcane improvement: an overview. Sugar Tech 13(4):322–335
Viswanathan R, Rao GP (2011) Disease scenario and Management of Major Sugarcane Diseases in India. Sugar Tech 13(4):336–353
Wei HR, Wang ML, Moore PH, Albert HH (2003) Comparative expression analysis of two sugarcane polyubiquitin promotersand flanking sequences in transgenic plants. J Plant Physiol 160:1241–1251
Xiong Y, Jung JH, Zeng QC, Gallo M, Altpeter F (2012) Comparison of procedures for DNA coating of micro-carriers in the transient and stable biolistic transformation of sugarcane. Plant Cell 112(1):95–99
Zhangsun DT, Luo SL, Chen RK, Tang KX (2007) Improved agrobacterium-mediated genetic transformation of GNA transgenic sugarcane. Biologia 62(4):386–393
Zhu YJ, McCafferty H, Osterman G, Lim S, Agbayani R, Lehrer A, Schenck S, Komor E (2011) Genetic transformation with untranslatable coat protein gene of sugarcane yellow leaf virus reduces virus titers in sugarcane. Transgenic Res 20(3):503–512
Acknowledgements
Mr. Wenzhi Wang received financial support from Hainan Natural Science Foundation of Hainan Province of China (20163122). Ms. Shuzhen Zhang received financial support from National Natural Science Foundation of China (31371687).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Hugo A. Campos
Rights and permissions
About this article
Cite this article
Wang, W.Z., Yang, B.P., Feng, C.L. et al. Efficient Sugarcane Transformation via bar Gene Selection. Tropical Plant Biol. 10, 77–85 (2017). https://doi.org/10.1007/s12042-017-9186-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12042-017-9186-7