Abstract
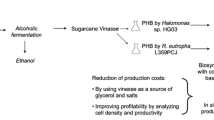
Vinasse, a recalcitrant waste of the ethanol industry was employed for the production of polyhydroxyalkanoate (PHA) by the extremely halophilic archaeon, Haloarcula marismortui in shake flasks. The PHA was recovered by osmotic lysis of the cells and subsequent purification by sodium hypochlorite and organic solvents. Through UV–vis spectroscopy, differential scanning calorimetry, Fourier transform infrared, and nuclear magnetic resonance spectroscopy, the PHA was found to have characteristics very similar to that of the standard polyhydroxybutyrate (PHB) from Sigma. Inhibitory effect of polyphenols contained in vinasse was assessed by a quick and reliable cup-plate agar-diffusion method. Raw vinasse (10%) was utilized leading to accumulation of 23% PHA (of cell dry weight) and following an efficacious pre-treatment process through adsorption on activated carbon, 100% pre-treated vinasse could be utilized leading to 30% accumulation of PHB by H. marismortui. Maximum specific growth rate, specific production rate, and volumetric productivity attained using 10% raw vinasse were comparable to that obtained using a previously reported nutrient deficient medium (NDM), while the values with 100% pre-treated vinasse were higher than that determined using NDM medium. This is the first report of polyhydroxybutyrate production by a halophilic microorganism utilizing vinasse.


Similar content being viewed by others
References
American Public Health Association (1998) Standard methods for the examination of water and wastewater, 20th edn. APHA, Washington
Bräsen C, Schönheit P (2004) Regulation of acetate and acetyl-CoA converting enzymes during growth on acetate and/or glucose in the halophilic archaeon Haloarcula marismortui. FEMS Microbiol Lett 241:21–26
Caqueret V, Bostyn S, Cagnon B, Fauduet H (2008) Purification of sugar beet vinasse—adsorption of polyphenolic and dark colored compounds on different commercial activated carbons. Bioresour Technol 99:5814–5821
Caqueret V, Cagnon B, Bostyn S, Fauduet H (2011) Removal of dark coloured and polyphenolic compounds of sugar beet vinasse by adsorption onto activated carbons: application to a crosscurrent adsorption process. Can J Chem Eng 9999:1–9
Dąbrowski A, Podkościelny P, Hubicki Z, Barczak M (2005) Adsorption of phenolic compounds by activated carbon—a critical review. Chemosphere 58:1049–1070
Fernandez-Castillo R, Rodriguez-Valera F, Gonzalez-Ramos J, Ruiz-Berraquero F (1986) Accumulation of poly (β-hydroxybutyrate) by Halobacteria. Appl Environ Microbiol 51:214–216
Flieger M, Kantorova M, Prell A, Řezanka T, Votruba J (2003) Biodegradable plastics from renewable sources. Folia Microbiol 48:27–44
Franzetti B, Schoehn G, Garcia D, Ruigrok RWH, Zaccai G (2002) Characterization of the proteasome from the extremely halophilic archaeon Haloarcula marismortui. Archaea 1:53–61
García García I, Bonilla Venceslada JL, Jiménez Peña PR, Ramos Gómez E (1997) Biodegradation of phenol compounds in vinasse using Aspergillus terreus and Geotrichum candidum. Water Res 31:2005–2011
Garcia Lillo J, Rodriguez-Valera F (1990) Effects of culture conditions on poly(β-hydroxybutyrate acid) production by Haloferax mediterranei. Appl Environ Microbiol 56:517–2521
Han J, Lu Q, Zhou L, Zhou J, Xiang H (2007) Molecular characterization of the phaECHm genes, required for biosynthesis of poly(3-hydroxybutyrate) in the extremely halophilic archaeon Haloarcula marismortui. Appl Environ Microbiol 73:6058–6065
Han J, Hou J, Liu H, Cai S, Feng B, Zhou J, Xiang H (2010) Wide distribution among halophilic archaea of a novel polyhydroxyalkanoate synthase subtype with homology to bacterial type III synthases. Appl Environ Microbiol 76:7811–7819
Hedge JE, Hofreiter BT (1962) Estimation of carbohydrates. In: Whistler RL, BeMiller JN (eds) Carbohydrate chemistry. Academic Press, New York, pp 17–22
Jacquel N, Lo C-W, Wei Y-H, Wu H-S, Wang SS (2008) Isolation and purification of bacterial poly(3-hydroxyalkanoates). Biochem Eng J 39:15–27
Law JH, Slepecky RA (1961) Assay of poly-β-hydroxybutyric acid. J Bacteriol 82:33–36
López-López A, Benlloch S, Bonfá M, Rodríguez-Valera F, Mira A (2007) Intragenomic 16S rDNA divergence in Haloarcula marismortui is an adaptation to different temperatures. J Mol Evol 65:687–696
Martín Santos MA, Fernández Bocanegra JL, Martín Martín A, García García I (2003) Ozonation of vinasse in acid and alkaline media. J Chem Technol Biotechnol 78:1121–1127
Nawaz H, Shi J, Mittal GS, Kakuda Y (2006) Extraction of polyphenols from grape seeds and concentration by ultrafiltration. Sep Purif Technol 48:176–181
Omar MNA, Mostafa AT, Ahmed AS (2002) Concentrated vinasse as a novel diazotrophs growth medium (biovinasse inoculant) and soil conditioner to improve faba bean yield under dripping irrigation system. Symposium No. 3, Paper No. 137, 17th WCSS, 14–21 August, Thailand
Oren A (2002) Halophilic microorganisms and their environments. Kluwer Academic Publishers, Dordrecht, p 136
Oren A, Ginzburg M, Ginzburg BZ, Hochstein LI, Volcani BE (1990) Haloarcula marismortui (Volcani) sp. nov., nom. rev., an extremely halophilic bacterium from the Dead Sea. Int J Syst Bacteriol 40:209–210
Ostle AG, Holt JG (1982) Nile blue A as a fluorescent stain for poly-β-hydroxybutyrate. Appl Environ Microbiol 44:238–241
Parnaudeau V, Condom N, Oliver R, Cazevieille P, Recous S (2008) Vinasse organic matter quality and mineralization potential, as influenced by raw material, fermentation and concentration processes. Bioresour Technol 99:1553–1562
Peres CS, Figueiredo MG, Vitoratto E, Perege L Jr (1986) Growth of anaerobic photosynthetic bacteria in vinasse medium. Rev Microbiol 17:1–9
Qi J, Li Z, Guo Y, Xu H (2004) Adsorption of phenolic compounds on micro- and mesoporous rice husk-based active carbons. Mater Chem Phys 87:96–101
Quillaguamán J, Guzmán H, Van-Thuoc D, Hatti-Kaul R (2010) Synthesis and production of polyhydroxyalkanoates by halophiles: current potential and future prospects. Appl Microbiol Biotechnol 85:1687–1696
Santos M, Diánez F, de Cara M, Tello JC (2008) Possibilities of the use of vinasses in the control of fungi phytopathogens. Bioresour Technol 99:9040–9043
Shrivastav A, Mishra SK, Mishra S (2010) Polyhydroxyalkanoate (PHA) synthesis by Spirulina subsalsa from Gujarat coast of India. Int J Biol Macromol 46:255–260
Swinnen IAM, Bernaerts K, Dens EJJ, Geeraerd AH, Van Impe JF (2004) Predictive modelling of the microbial lag phase: a review. Int J Food Microbiol 94:137–159
Tamboli DP, Kagalkar AN, Jadhav MU, Jadhav JP, Govindwar SP (2010) Production of polyhydroxyhexadecanoic acid by using waste biomass of Sphingobacterium sp. ATM generated after degradation of textile dye Direct Red 5B. Bioresour Technol 101:2421–2427
Tu D, Blaha G, Moore PB, Steitz TA (2005) Gene replacement in Haloarcula marismortui: construction of a strain with two of its three chromosomal rRNA operons deleted. Extremophiles 9:427–435
Van-Thuoc D, Quillaguamán J, Mamo G, Mattiasson B (2008) Utilization of agricultural residues for poly(3-hydroxybutyrate) production by Halomonas boliviensis LC1. J Appl Microbiol 104:420–428
Walkley A, Black IA (1934) An examination of the Degtjareff method for determining soil organic matter and a proposed modification of the chromic acid titration method. Soil Sci 37:29–38
Acknowledgements
Authors wish to thank Mr. Swapan K Bayen, Mr. Santanu Ghose, and Mr. Anindya Roy of IFB Agro Industries for the gift of the vinasse. Assistance from Mr. Biswarup Saha during fluorescence microscopy is thankfully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Figure 1
UV–vis scanning spectroscopy following hydrolysis and dehydrogenation of standard PHB from Sigma (red line) and PHA from H. marismortui (blue line) (JPEG 88 kb)
Supplementary Figure 2
DSC thermograms of a PHA from H. marismortui (JPEG 74 kb)
Supplementary Figure 2
DSC thermograms of b standard PHB from Sigma (JPEG 71 kb)
Supplementary Figure 3
FTIR spectra of PHA from H. marismortui (lower trace), compared with standard PHB from Sigma (upper trace) (JPEG 265 kb)
Supplementary Figure 4
1H NMR spectra of a PHA from H. marismortui (JPEG 204 kb)
Supplementary Figure 4
1H NMR spectra of b standard PHB from Sigma (JPEG 134 kb)
Rights and permissions
About this article
Cite this article
Pramanik, A., Mitra, A., Arumugam, M. et al. Utilization of vinasse for the production of polyhydroxybutyrate by Haloarcula marismortui . Folia Microbiol 57, 71–79 (2012). https://doi.org/10.1007/s12223-011-0092-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-011-0092-3