Abstract
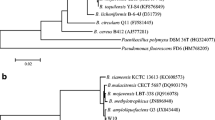
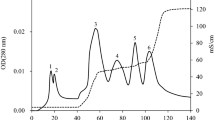
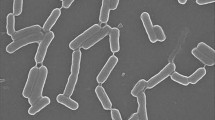
Bacillus amyloliquefaciens LBM 5006 produces antagonistic activity against pathogenic bacteria and phytopathogenic fungi, including Aspergillus spp., Fusarium spp., and Bipolaris sorokiniana. PCR analysis revealed the presence of ituD, but not sfp genes, coding for iturin and surfactin, respectively. The antimicrobial substance produced by this strain was isolated by ammonium sulfate precipitation, gel filtration chromatography and 1-butanol extraction. The ultraviolet spectrum was typical of a polypeptide and the infrared spectrum indicates the presence of peptide bonds and acyl group(s). The antimicrobial substance was resistant to proteolytic enzymes and heat treatment, and was reactive with ninhydrin. Mass spectroscopy analysis indicated that B. amyloliquefaciens LBM 5006 produces two antimicrobial peptides, with main peaks at m/z 1,058 Da and 1,464 Da, corresponding to iturin-like and fengycin-like peptides, respectively. B. amyloliquefaciens LBM 5006 showed significant activity against phytopatogenic fungi, showing potential for use as a biocontrol agent or production of antifungal preparations.
Similar content being viewed by others
References
Agrios, G.N. 2005. Plant pathology. Elsevier, London, UK.
Ajesh, K. and K. Sreejith. 2009. Peptide antibiotics: An alternative and effective antimicrobial strategy to circumvent fungal infections. Peptides 30, 999–1006.
Akpa, E., P. Jacques, B. Wathelet, M. Paquot, R. Fuchs, H. Budzikiewicz, and P. Thonart. 2001. Influence of culture conditions on lipopeptide production by Bacillus subtilis. Appl. Biochem. Biotechnol. 91, 551–561.
Bais, H.P., R. Fall, and J.M. Vivanco. 2004. Biocontrol of Bacillus subtilis against infection of Arabidopsis roots by Pseudomonas syringae is facilitated by biofilm formation and surfactin production. Plant Physiol. 134, 307–319.
Besson, F. and G. Michel. 1987. Isolation and characterization of new iturins: iturin D and iturin E. J. Antibiot. 40, 437–442.
Caldeira, A.T., S.S. Feio, J.M.S. Arteiro, A.V. Coelho, and J.C. Roseiro. 2008. Environmental dynamics of Bacillus amyloliquefaciens CCMI 1051 antifungal activity under different nitrogen patterns. J. Appl. Microbiol. 104, 808–816.
Chen, H., L. Wang, C.X. Su, G.H. Gong, P. Wang, and Z.L. Yu. 2008. Isolation and characterization of lipopeptide antibiotics produced by Bacillus subtilis. Lett. Appl. Microbiol. 47, 180–186.
Cladera-Olivera, F., G.R. Caron, and A. Brandelli. 2006. Bacteriocinlike substance inhibits potato soft rot caused by Erwinia carotovora. Can. J. Microbiol. 52, 533–539.
Duitman, E.H., L.W. Hamoen, M. Rembold, G. Venema, H. Seitz, W. Saenger, F. Bernhard, and et al. 1999. The mycosubtilin synthetase of Bacillus subtilis ATCC6633: a multifunctional hybrid between a peptide synthetase, an amino transferase, and a fatty acid synthetase. Proc. Natl. Acad. Sci. USA 96, 13294–13299.
Evans, F.F., A.S. Rosado, G.V. Sebastián, R. Casella, P.L.O.A. Machado, C. Holmstrom, S. Kjelleberg, J.D. van Elsas, and L. Seldin. 2004. Impact of oil contamination and biostimulation on the diversity of indigenous bacterial communities in soil microcosms. FEMS Microbiol. Ecol. 49, 295–305.
Fabian, H., C. Schultz, D. Naumann, O. Landt, U. Hahn, and W. Saenger. 1993. Secondary structure and temperature-induced unfolding and refolding of ribonuclease-T1 in aqueous solution — a Fourier transform infrared spectroscopy study. J. Mol. Biol. 232, 967–981.
Gaussier, H., M. Lavoie, and M. Subirade. 2003. Conformational changes of pediocin in an aqueous medium monitored by Fourier transform infrared spectroscopy: a biological implication. Int. J. Biol. Macromolec. 32, 1–9.
Hancock, R.E.W. and G. Diamond. 2000. The role of cationic antimicrobial peptides in innate host defences. Trends Microbiol. 8, 402–410.
Hiradate, S., S. Yoshida, H. Sugie, H. Yada, and Y. Fujii. 2002. Mulberry anthracnose antagonists (iturins) produced by Bacillus amyloliquefaciens RC-2. Phytochemistry 61, 693–698.
Hsieh, F.C., M.C. Li, T.C. Lin, and S.S. Kao. 2004. Rapid detection and characterization of surfactin-producing Bacillus subtilis and closely related species based on PCR. Curr. Microbiol. 49, 186–191.
Hsieh, F.C., T.C. Lin, M. Meng, and S.S. Kao. 2008. Comparing methods for identifying Bacillus strains capable of producing the antifungal lipopeptide iturin A. Curr. Microbiol. 56, 1–5.
Hu, L.B., Z.Q. Shi, T. Zhang, and Z.M. Yang. 2007. Fengycin antibiotics isolated from B-FS01 culture inhibiting the growth of Fusarium moniliforme Sheldon ATCC 38932. FEMS Microbiol. Lett. 272, 91–98.
Kurusu, K. and K. Ohba. 1987. New peptide antibiotics LI-FO3, FO4, FO5, FO7 and FO8, produced by Bacillus polymyxa. Isolation and characterization. J. Antibiot. 40, 1506–1514.
Le Marrec, C., B. Hyronimus, P. Bressollier, B. Verneuil, and M.C. Urdaci. 2000. Biochemical and genetic characterization of coagulin, a new antilisterial bacteriocin in the pediocin family of bacteriocins, produced by Bacillus coagulans I4. Appl. Environ. Microbiol. 66, 5213–5220.
Leifert, C., H. Li, S. Chidburee, S. Hampson, S. Workman, D. Sigee, H.A.S. Epton, and A. Harbour. 1995. Antibiotic production and biocontrol activity by Bacillus subtilis CL27 and Bacillus pumillus CL45. J. Appl. Bacteriol. 78, 97–108.
Lisboa, M.P., D. Bonatto, D. Bizani, J.A.P. Henriques, and A. Brandelli. 2006. Characterization of a bacteriocin-like substance produced by Bacillus amyloliquefaciens isolated from the Brazilian Atlantic Forest. Int. Microbiol. 9, 111–118.
Lowry, O.H., N.J. Rosebrough, A.L. Farr, and R.J. Randall. 1951. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193, 267–275.
Maget-Dana, R. and F. Peypoux. 1994. Iturins, a special class of poreforming lipopeptides: Biological and physicochemical properties. Toxicology 87, 151–174.
Maquelin, K., C. Kirschner, L.P. Choo-Smith, N. van den Braak, H.P. Endtz, D. Naumann, and G.J. Puppels. 2002. Identification of medically relevant microorganisms by vibrational spectroscopy. J. Microbiol. Methods 51, 255–271.
Matos, D.M.S. and M.L.A. Bovi. 2002. Understanding the threats to biological diversity in southeastern Brazil. Biodiv. Conserv. 11, 1747–1758.
Moore, S. and W.H. Stein. 1957. A modified ninhidrin reagent for the photometric determination of amino acids and related compounds. J. Biol. Chem. 211, 907–913.
Motta, A.S. and A. Brandelli. 2002. Characterization of an antibacterial peptide produced by Brevibacterium linens. J. Appl. Microbiol. 92, 63–71.
Motta, A.S. and A. Brandelli. 2009. Properties and antimicrobial activity of the smear surface cheese coryneform bacterium Brevibacterium linens. Eur. Food Res. Technol. 227, 1299–1306.
Motta, A.S., F.S. Cannavan, S.M. Tsai, and A. Brandelli. 2007. Characterization of a broad range antibacterial substance from a new Bacillus species isolated from Amazon basin. Arch. Microbiol. 188, 367–375.
Nascimento, A.M.A., L. Cursino, H. Gonąlves-Dornelas, A. Reis, E. Charton-Souza, and M.A. Marini. 2003. Antibiotic-resistant Gram-negative bacteria in birds from the Brazilian Atlantic forest. Condor 105, 358–361.
Oscáriz, J.C. and A.G. Pisabarro. 2000. Characterization and mechanism of action of cerein 7, a bacteriocin produced by Bacillus cereus Bc7. J. Appl. Microbiol. 89, 361–369.
O’sullivan, L., R.P. Ross, and C. Hill. 2002. Potential of bacteriocin-producing lactic acid bacteria for improvements in food safety and quality. Biochimie 84, 593–604.
Pabel, C., J. Vater, C. Wilde, P. Franke, J. Hofemeister, B. Adler, G. Bringmann, J. Hacker, and U. Hentschel. 2003. Antimicrobial activities and matrix-assisted laser desorption/ionization mass spectrometry of Bacillus isolates from the marine sponge Aplysina aerophoba. Mar. Biotechnol. 5, 424–434.
Riley, M.A. and J.E. Wertz. 2002. Bacteriocins: evolution, ecology, and application. Annu. Rev. Microbiol. 56, 117–137.
Romero, D., A. De Vicente, R.H. Rakotoaly, S.E. Dufour, J.W. Veening, E. Arrebola, F.M. Cazorla, O.P. Kuipers, M. Paquot, and A. Perez-Garcia. 2007. The iturin and fengycin families of lipopeptides are key factors in antagonism of Bacillus subtilis toward Podosphaera fusca. Mol. Plant Pathogen Interact. 20, 430–440.
Romero, D., A. Pérez-Garcia, M.E. Rivera, F.M. Cazoela, and A. Vicente. 2004. Isolation and evaluation of antagonistic bacteria towards the cucurbit powdery milder fungus Podospheara fusca. Appl. Microbiol. Biotechnol. 64, 263–269.
Schneider, A., T. Stachelhaus, and M.A. Marahiel. 1998. Targeted alteration of the substrate specificity of peptide synthetases by rational module swapping. Mol. Gen. Genet. 257, 308–318.
Souto, G.I., O.S. Correa, M.S. Montecchia, N.L. Kerber, N.L. Pucheu, M. Bachur, and A.F. Garcia. 2004. Genetic and functional characterization of a Bacillus sp. strain excreting surfactin and antifungal metabolites partially identified as iturin-like compounds. J. Appl. Microbiol. 97, 1247–1256.
Stein, T. 2005. Bacillus subtilis antibiotics: structures, syntheses and specific functions. Mol. Microbiol. 56, 845–857.
Stein, T. 2008. Whole-cell matrix-assisted laser desorption/ionization mass spectrometry for rapid identification of bacteriocin/lanthibiotic-producing bacteria. Rapid Commun. Mass Spectr. 22, 1146–1152.
Sutyak, K.E., R.E. Wirawan, A.A. Aroucheva, and M.L. Chikindas. 2008. Isolation of the Bacillus subtilis antimicrobial peptide subtilosin from the dairy product-derived Bacillus amyloliquefaciens. J. Appl. Microbiol. 104, 1067–1074.
Thompson, J.D., D.G. Higgins, and T.J. Gibson. 1994. Clustal W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22, 4673–4680.
Tsuge, K., T. Akiyama, and M. Shoda. 2001. Cloning, sequencing, and characterization of the iturin A operon. J. Bacteriol. 183, 6265–6273.
Valdés-Satuber, N. and S. Scherer. 1994. Isolation and characterization of linocin M18, a bacteriocin produced by Brevibacterium linens. Appl. Environ. Microbiol. 60, 3809–3814.
Vater, J., B. Kablitz, C. Wilde, P. Franke, N. Mehta, and S.S. Cameotra. 2002. Matrix-assisted laser desorption ionization-time of flight mass spectrometry of lipopeptide biosurfactants in whole cells and culture filtrates of Bacillus subtilis C-1 isolated from petroleum sludge. Appl. Environ. Microbiol. 68, 6210–6219.
Winteringham, F.P.W. 2008. Fate and significance of chemical pesticides: an appraisal in the context of integrated control. EPPO Bull. 5, 65–71.
Yakimov, M.M., K.N. Timmis, V. Wray, and H.L. Fredrickson. 1995. Characterization of a new lipopeptide surfactant produced by thermotolerant and halotolerant subsurface Bacillus licheniformis BAS50. Appl. Environ. Microbiol. 61, 1706–1713.
Yao, S., X. Gao, N. Fuchsbauer, W. Hillen, J. Vater, and J. Wang. 2003. Cloning, sequencing, and characterization of the genetic region relevant to biosynthesis of the lipopeptides iturin A and surfactin in Bacillus subtilis. Curr. Microbiol. 47, 272–277.
Ye, S.Y., H.X. Wang, and T.B. Ng. 1999. First chromatographic isolation from French bean legumes and demonstration of its antifungal activity. Biochem. Biophys. Res. Commun. 263, 130–134.
Yoshida, S., S. Hiradate, T. Tsukamoto, K. Hatakeda, and A. Shirata. 2001. Antimicrobial activity of culture filtrate of Bacillus amyloliquefaciens RC-2 isolated from mulberry leaves. Biol. Control 91, 181–187.
Yu, G.Y., J.B. Sinclair, G.L. Hartman, and B.L. Bertagnolli. 2002. Production of iturin A by Bacillus amyloliquefaciens suppressing Rhizoctonia solani. Soil Biol. Biochem. 34, 955–963.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Benitez, L.B., Velho, R.V., Lisboa, M.P. et al. Isolation and characterization of antifungal peptides produced by Bacillus amyloliquefaciens LBM5006. J Microbiol. 48, 791–797 (2010). https://doi.org/10.1007/s12275-010-0164-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-010-0164-0