Abstract
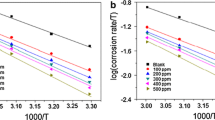
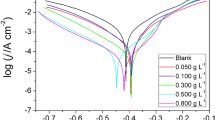
The alkaloid content of the leaves and stem bark of Xylopia ferruginea plant was isolated and tested for its anticorrosion potential on mild steel corrosion in a hydrochloric acid medium by using electrochemical impedance spectroscopy, potentiodynamic polarization measurement, scanning electron microscopy (SEM), and Fourier transform infra red (FTIR) analysis. The experimental results reveal the effective anticorrosion potential of the plant extract. The mixed mode of action exhibited by the plant extract is evidenced from the polarization study. SEM images proof the formation of a protective layer over the mild steel surface, and this is supported by the FTIR study. The possible mode of the corrosion inhibition mechanism has also been discussed.
Similar content being viewed by others
References
L. Jin, L. Wang, and D. Chen, Corrosion inhibition of a hygroscopic inorganic dust-depressor, J. Univ. Sci. Technol. Beijing, 13(2006), No.4, p.368.
P.B. Raja and M.G. Sethuraman, Atropine sulphate as corrosion inhibitor for mild steel in sulphuric acid media, Mater. Lett., 62(2008), No.10–11, p.1602.
E.E. Oguzie, Corrosion inhibition of aluminium in acidic and alkaline media by Sansevieria trifasciata extract, Corros. Sci., 49(2007), p.1527.
P.B. Raja and M.G. Sethuraman, Natural products as corrosion inhibitor for metals in corrosive media: a review, Mater. Lett., 62(2008), No.1, p.113.
P.B. Raja and M.G. Sethuraman, Inhibitive effect of black pepper extract on the sulphuric acid corrosion of mild steel, Mater. Lett., 62(2008), No.17–18, p.2977.
P.B. Raja and M.G. Sethuraman, Strychnos nux-vomica an eco-friendly corrosion inhibitor for mild steel in 1 M sulphuric acid medium, Mater. Corros., 22(2009), No.1, p.22.
A.A. Rahim, E. Rocca, J. Steinmetz, et al., Mangrove tannins and their flavanoid monomers as alternative steel corrosion inhibitors in acidic medium, Corros. Sci., 49(2007), No.2, p.402.
A.A. Rahim, E. Rocca, J. Steinmetz, and M.J. Kassim, Inhibitive action of mangrove tannins and phosphoric acid on pre-rusted steel via electrochemical methods, Corros. Sci., 50(2008), No.6, p.1546.
A.A. Rahim and J. Kassim, Recent development of vegetal tannins in corrosion protection of iron and steel, Recent Pat. Mater. Sci., 1(2008), No.3, p.223.
T.S. Kam, Alkaloids from Malaysian Flora, Alkaloids: Chem. Biol. Perspect., 14(1999), p.285.
R.H.F. Manske, The Alkaloids: Chemistry and Physiology, Academic Press, London, 1973, p.236.
M. Lebrini, M. Lagrenée, H. Vezin, et al., Experimental and theoretical study for corrosion inhibition of mild steel in normal hydrochloric acid solution by some new macrocyclic polyether compounds, Corros. Sci., 49(2007), No.5, p.2254.
M.A. Quraishi and J. Rawat, Influence of iodide ions on inhibitive performance of tetraphenyl-dithia-octaaza-cyclotet-radeca-hexaene (PTAT) during pickling of mild steel in hot sulfuric acid, Mater. Chem. Phys., 70(2001), No.1, p.95.
F. Bentiss, M. Lagrenee, M. Traisnel, and J.C. Hornez, The corrosion inhibition of mild steel in acidic media by a new triazole derivative, Corros. Sci., 41 (1999), p.789.
A.C. Vajpei, Uma, I.P. Saraswat, and K. Chandra, Characterization of brown ferric oxide hydrate xerogel, J. Mater. Sci., 19(1984), No.10, p.3341.
A.V. Ramesh Kumar, R.K. Nigam, S.S. Monga, and G.N. Mathur, Effect of inhibitors on the nature of corrosion products of mild steel by Mossbauer and FTIR spectroscopy, Anti Corros. Meth. Mater., 49(2002), No.2, p.111.
A.A. Rahim, Mangrove Tannin as A green Corrosion Inhibitor of Steel: A Comprehensive Study, VDM Verlag Dr. Müller, Germany, 2009, p.103.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was financially supported by the Foundation of University Sains Malaysia (RU-1001/229/PKIMIA/811143 and FRGS-203/PKMIA/671030).
Rights and permissions
About this article
Cite this article
Raja, P.B., Rahim, A.A., Osman, H. et al. Inhibitive effect of Xylopia ferruginea extract on the corrosion of mild steel in 1M HCl medium. Int J Miner Metall Mater 18, 413–418 (2011). https://doi.org/10.1007/s12613-011-0455-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12613-011-0455-4