Abstract
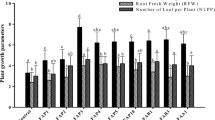
Drought ranks among the most severe environmental stressors that adversely affect crop productivity. Use of stress-tolerant plant growth-promoting rhizobacteria (PGPR) is expected to ameliorate the drought stress in plants. Therefore, in this study, a novel strain, Pseudomonas azotoformans FAP5 with multifunctional traits was obtained from screening of 50 indigenous isolates. The criteria for selection of most promising strain were based on tolerance to water stress, production of PGP traits and biofilm-forming ability in vitro. Further, inoculation response of FAP5 on wheat growth parameters and physiological attributes under different drought conditions was executed under pot experiment. The drought-tolerant FAP5 strain revealed production of exopolysaccharides (EPS), indole 3 acetic acid (IAA), solubilized tricalcium phosphate and exhibited 1-aminpcyclopropane-1-carboxylate (ACC) deaminase (41.58 ± 2.10 μmol α-KB/mg Pr h) activity. ACC deaminase (acdS) and biofilm-associated (AdnA and FliC) genes were confirmed by PCR-based method. Production of PGP traits and biofilm development by FAP5 was exhibited even at water stress condition in vitro. The inoculated wheat plants with FAP5 strain were grown under drought stress showed significant (P < 0.05) improvement in growth attributes, photosynthetic pigment efficiency and other physiological attributes. Similarly, a significant (P < 0.05) antioxidative enzymatic activities in FAP5-inoculated plant were observed compared to uninoculated plants. Under drought stress condition, biofilm development by FAP5 was enhanced significantly and root colonization by inoculant strain was confirmed by viable count and scanning electron microscopy (SEM). The findings indicated that FAP5 strain is effective to drought stress alleviation in wheat plants through various biochemical mechanisms.





Similar content being viewed by others
References
Adler J (1966) Chemotaxis in bacteria. Science 153:708–716. https://doi.org/10.1126/SCIENCE.153.3737.708
Ahmad F, Ahmad I, Khan MS (2008) Screening of free-living rhizospheric bacteria for their multiple plant growth promoting activities. Microbiol Res 163:173–181. https://doi.org/10.1016/j.micres.2006.04.001
Alexander DB, Zuberer DA (1991) Use of chrome azurol S reagents to evaluate siderophore production by rhizosphere bacteria. Biol Fertil Soils 12:39–45. https://doi.org/10.1007/BF00369386
Angus AA, Hirsch AM (2013) Biofilm formation in the rhizosphere: multispecies interactions and implications for plant growth. Mol Microb Rhizosphere 1:701–12
Ansari FA, Ahmad I (2018) Biofilm development, plant growth promoting traits and rhizosphere colonization by Pseudomonas entomophila FAP1: a Promising PGPR. Adv Microbiol 08:235–251. https://doi.org/10.4236/aim.2018.83016
Ansari FA, Ahmad I (2019) Fluorescent Pseudomonas -FAP2 and Bacillus licheniformis interact positively in biofilm mode enhancing plant growth and photosynthetic attributes. Sci Rep 9:4547. https://doi.org/10.1038/s41598-019-40864-4
Ansari FA, Jafri H, Ahmad I, Abulreesh HH (2017) Factors affecting biofilm formation in in vitro and in the rhizosphere. Biofilms in plant and soil health. John Wiley & Sons Ltd, Chichester, pp 275–290
Ansari FA, Ahmad I, Pichtel J (2019) Growth stimulation and alleviation of salinity stress to wheat by the biofilm forming Bacillus pumilus strain FAB10. Appl Soil Ecol 143:45–54. https://doi.org/10.1016/j.apsoil.2019.05.023
Bakker AW, Schippers B (1987) Microbial cyanide production in the rhizosphere in relation to potato yield reduction and Pseudomonas Spp-mediated plant growth-stimulation. Soil Biol Biochem 19:451–457. https://doi.org/10.1016/0038-0717(87)90037-X
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Beer RF Jr, Sizer IW (1952) A spectrophotometric method for measuring the breakdown 585 of hydrogen peroxide by catalase. J Biol Chem 195:133–140
Bric JM, Bostock RM, Silverstone SE (1991) Rapid in situ assay for indoleacetic acid production by bacteria immobilized on a nitrocellulose membrane. Appl Environ Microbiol 57:535–538
Bogino PC, Oliva MD, Sorroche FG, Giordano W (2013) The role of bacterial biofilms and surface components in plant-bacterial associations. Int J Mole Sci 14(8):15838–59
Cruz de Carvalho MH (2008) Drought stress and reactive oxygen species: production, scavenging and signaling. Plant Signal Behav 3:156–165. https://doi.org/10.4161/psb.3.3.5536
Dworkin M, Foster JW (1958) Experiments with some microorganisms which utilize ethane and hydrogen. J Bacteriol 75(5):592
Duo LA, Liu CX, Zhao SL (2018) Alleviation of drought stress in turfgrass by the combined application of nano-compost and microbes from compost. Russ J Plant Physiol 65:419–426. https://doi.org/10.1134/S102144371803010X
Dye D (1962) The inadequacy of the usual determinative tests for the identification of Xanthomonas spp. N Z J Sci 5:393–416
Egamberdieva D, Wirth S, Bellingrath-Kimura SD, Mishra J, Arora NK (2019) Salt-tolerant plant growth promoting rhizobacteria for enhancing crop productivity of saline soils. Front Microbiol 10:2791. https://doi.org/10.3389/fmicb.2019.02791
Fahad S, Bajwa AA, Nazir U et al (2017) Crop production under drought and heat stress: plant responses and management options. Front Plant Sci. https://doi.org/10.3389/fpls.2017.01147
Gontia-Mishra I, Sasidharan S, Tiwari S (2014) Recent developments in use of 1-aminocyclopropane-1-carboxylate (ACC) deaminase for conferring tolerance to biotic and abiotic stress. Biotechnol Lett 36:889–898. https://doi.org/10.1007/s10529-014-1458-9
Gontia-Mishra I, Sapre S, Sharma A, Tiwari S (2016) Amelioration of drought tolerance in wheat by the interaction of plant growth-promoting rhizobacteria. Plant Biol 18:992–1000. https://doi.org/10.1111/plb.12505
Grover M, Madhubala R, Ali SZ et al (2014) Influence of Bacillus spp. strains on seedling growth and physiological parameters of sorghum under moisture stress conditions. J Basic Microbiol 54:951–961. https://doi.org/10.1002/jobm.201300250
Hare PD, Cress WA, Van Staden J (1998) Dissecting the roles of osmolyte accumulation during stress. Plant Cell Environ 21:535–553. https://doi.org/10.1046/j.1365-3040.1998.00309.x
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts. Arch Biochem Biophys 125:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Holt JG, Krieg NR, Sneath PHA, Staley JT, Williams ST (1994) Bergey’s manual of determinative bacteriology, 9 edn
Iwase T, Tajima A, Sugimoto S et al (2013) A simple assay for measuring catalase activity: a visual approach. Sci Rep 3:3081. https://doi.org/10.1038/srep03081
Jablonski PP, Anderson JW (1978) Light-dependent reduction of oxidized glutathione by ruptured chloroplasts. Plant Physiol 61:221–225. https://doi.org/10.1104/pp.61.2.221
Jha B, Gontia I, Hartmann A (2012) The roots of the halophyte Salicornia brachiata are a source of new halotolerant diazotrophic bacteria with plant growth-promoting potential. Plant Soil 356:265–277. https://doi.org/10.1007/s11104-011-0877-9
Kasim WA, Osman ME, Omar MN et al (2013) Control of drought stress in wheat using plant-growth-promoting bacteria. J Plant Growth Regul 32:122–130. https://doi.org/10.1007/s00344-012-9283-7
Kaushal M, Wani SP (2016) Plant-growth-promoting rhizobacteria: drought stress alleviators to ameliorate crop production in drylands. Ann Microbiol 66:35–42. https://doi.org/10.1007/s13213-015-1112-3
Kechid M, Desbrosses G, Rokhsi W et al (2013) The NRT2.5 and NRT2.6 genes are involved in growth promotion of Arabidopsis by the plant growth-promoting rhizobacterium (PGPR) strain Phyllobacterium brassicacearum STM196. New Phytol 198:514–524. https://doi.org/10.1111/nph.12158
Lata C, Jha S, Dixit V et al (2011) Differential antioxidative responses to dehydration-induced oxidative stress in core set of foxtail millet cultivars [Setaria italica (L.)]. Protoplasma 248:817–828. https://doi.org/10.1007/s00709-010-0257-y
Lowry OH, Rosenbrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Martins SJ, Rocha GA, de Melo HC et al (2018) Plant-associated bacteria mitigate drought stress in soybean. Environ Sci Pollut Res 25:13676–13686. https://doi.org/10.1007/s11356-018-1610-5
Mayak S, Tirosh T, Glick BR (2004) Plant growth-promoting bacteria that confer resistance to water stress in tomatoes and peppers. Plant Sci 166:525–530. https://doi.org/10.1016/j.plantsci.2003.10.025
Mody B, Bindra M, Modi V (1989) Extracellular polysaccharides of cowpea rhizobia: compositional and functional studies. Arch Microbiol 153:38–42. https://doi.org/10.1007/BF00277538
Nautiyal CS, Srivastava S, Chauhan PS et al (2013) Plant growth-promoting bacteria Bacillus amyloliquefaciens NBRISN13 modulates gene expression profile of leaf and rhizosphere community in rice during salt stress. Plant Physiol Biochem 66:1–9. https://doi.org/10.1016/j.plaphy.2013.01.020
Naveed M, Hussain MB, Zahir ZA et al (2014) Drought stress amelioration in wheat through inoculation with Burkholderia phytofirmans strain PsJN. Plant Growth Regul 73:121–131. https://doi.org/10.1007/s10725-013-9874-8
Nadeem SM, Zahir ZA, Naveed M, Nawaz S (2013) Mitigation of salinity-induced negative impact on the growth and yield of wheat by plant growth promoting rhizobacteria in naturally saline conditions. Annal Microbiol 63(1):225–32
Niu G, Rodriguez DS, Cabrera R, Jifon J, Leskovar D, Crosby K (2010) Salinity and soil type effects on emergence and growth of pepper seedlings. Hort Sci. 45(8):1265–9
Niu M, Xie J, Chen C, Cao H, Sun J, Kong Q, Shabala S, Shabala L, Huang Y, Bie Z (2018) An early ABA-induced stomatal closure, Na+ sequestration in leaf vein and K+ retention in mesophyll confer salt tissue tolerance in Cucurbita species. J Exper Bot 69(20):4945–60
O’Toole GA (2011) Microtiter dish biofilm formation assay. J Vis Exp. https://doi.org/10.3791/2437
Olanrewaju OS, Glick BR, Babalola OO (2017) Mechanisms of action of plant growth promoting bacteria. World J Microbiol Biotechnol 33:197. https://doi.org/10.1007/s11274-017-2364-9
Penrose DM, Glick BR (2003) Methods for isolating and characterizing ACC deaminase-containing plant growth-promoting rhizobacteria. Physiol Plant 118:10–15. https://doi.org/10.1034/j.1399-3054.2003.00086.x
Pikovskaya RI (1948) Mobilization of phosphorus in soil connection with the vital activity of some microbial species. Microbiologiya 17:362–370
Ray S, Singh S, Sarma BK, Singh HB (2016) Endophytic alcaligenes isolated from horticultural and medicinal crops promotes growth in okra (Abelmoschus esculentus). J Plant Growth Regul 35:401–412. https://doi.org/10.1007/s00344-015-9548-z
Rosenberg M, Gutnick D, Rosenberg E (1980) Adherence of bacteria to hydrocarbons: a simple method for measuring cell-surface hydrophobicity. FEMS Microbiol Lett 9:29–33. https://doi.org/10.1111/j.1574-6968.1980.tb05599.x
Saikia J, Sarma RK, Dhandia R et al (2018) Alleviation of drought stress in pulse crops with ACC deaminase producing rhizobacteria isolated from acidic soil of Northeast India. Sci Rep 8:3560. https://doi.org/10.1038/s41598-018-21921-w
Sandhya V, Ali SKZ, Grover M et al (2009) Alleviation of drought stress effects in sunflower seedlings by the exopolysaccharides producing Pseudomonas putida strain GAP-P45. Biol Fertil Soils 46:17–26. https://doi.org/10.1007/s00374-009-0401-z
Sandhya V, Ali SZ, Grover M et al (2010) Effect of plant growth promoting Pseudomonas spp. on compatible solutes, antioxidant status and plant growth of maize under drought stress. Plant Growth Regul 62:21–30. https://doi.org/10.1007/s10725-010-9479-4
Sarma RK, Saikia R (2014) Alleviation of drought stress in mung bean by strain Pseudomonas aeruginosa GGRJ21. Plant Soil 377:111–126. https://doi.org/10.1007/s11104-013-1981-9
Simons M, van der Bij AJ, Brand I et al (1996) Gnotobiotic system for studying rhizosphere colonization by plant growth-promoting Pseudomonas bacteria. Mol Plant Microbe Interact 9:600–607
Singh A, Jain A, Sarma BK et al (2014) Rhizosphere competent microbial consortium mediates rapid changes in phenolic profiles in chickpea during Sclerotium rolfsii infection. Microbiol Res 169:353–360. https://doi.org/10.1016/j.micres.2013.09.014
Smyth DR, Bowman JL, Meyerowitz EM (1990) Early flower development in Arabidopsis. Plant Cell 2:755–767
Srivastava S, Yadav A, Seem K et al (2008) Effect of high temperature on Pseudomonas putida NBRI0987 biofilm formation and expression of stress sigma factor RpoS. Curr Microbiol 56:453–457. https://doi.org/10.1007/s00284-008-9105-0
Upadhyay SK, Singh DP, Saikia R (2009) Genetic diversity of plant growth promoting rhizobacteria isolated from rhizospheric soil of wheat under saline condition. Curr Microbiol 59:489–496. https://doi.org/10.1007/s00284-009-9464-1
Vurukonda SSKP, Vardharajula S, Shrivastava M, SkZ A (2016) Enhancement of drought stress tolerance in crops by plant growth promoting rhizobacteria. Microbiol Res 184:13–24. https://doi.org/10.1016/j.micres.2015.12.003
Wu Z, Peng Y, Guo L, Li C (2014) Root colonization of encapsulated Klebsiella oxytoca Rs-5 on cotton plants and its promoting growth performance under salinity stress. Eur J Soil Biol 60:81–87
Wozniak DJ, Wyckoff TJO, Starkey M et al (2003) Alginate is not a significant component of the extracellular polysaccharide matrix of PA14 and PAO1 Pseudomonas aeruginosa biofilms. Proc Natl Acad Sci 100:7907–7912. https://doi.org/10.1073/pnas.1231792100
Yogendra SG, Singh US, Sharma AK (2015) Bacterial mediated amelioration of drought stress in drought tolerant and susceptible cultivars of rice (Oryza sativa L.). African J Biotechnol 14:764–773. https://doi.org/10.5897/AJB2015.14405
Acknowledgements
The author F. A. A. gratefully acknowledges to University Grant Commission (UGC-MAN-SRF), New Delhi, for financial support in the form of research fellowship. We are also appreciatively acknowledged the support of University Sophisticated Instrumentation Facility (USIF), Aligarh Muslim University, Aligarh and Macrogen Sequencing Team, Seoul, South Korea, for 16S rRNA gene sequencing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that there is no competing interest in this research manuscript.
Additional information
Editorial responsibility: BV Thomas.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ansari, F.A., Jabeen, M. & Ahmad, I. Pseudomonas azotoformans FAP5, a novel biofilm-forming PGPR strain, alleviates drought stress in wheat plant. Int. J. Environ. Sci. Technol. 18, 3855–3870 (2021). https://doi.org/10.1007/s13762-020-03045-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-020-03045-9