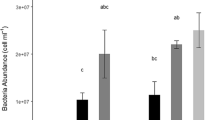
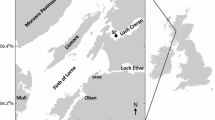
Abstract
Numerous investigations have been directed at verifying and calibrating methods for measuring bacterioplankton production, particularly methods based on the incorporation of thymidine (TdR) into DNA. Careful examination of these data can provide insights into other aspects of the ecology of aerobic heterotrophic microbial communities. Once method-specific biases are eliminated, these measurements indicate that there are broad-scale patterns in the metabolic fate of TdR, differences that seem to reflect broad differences in community metabolic capabilities. Based on work conducted primarily in San Francisco and Tomales Bays, California, I suggest that the metabolic fate of TdR in a given environment may reflect the relative importance to bacterioplankton nutrition of detritus versus fresh phytoplankton carbon. This is probably due to differences in community composition that result from growth on qualitatively different carbon sources. If true, the metabolic fate of TdR may provide a broadly applicable, but simple, index that can be used to assess the relative importance of these general sources of organic matter. Such an index could be very useful in characterizing lacustrine, estuarine, and nearshore environments.
Similar content being viewed by others
References
Alpine AE, Cloern JE (1993) Trophic interactions and direct physical effects control phytoplankton biomass and production in an estuary. Limnol Oceanog 37:946–955
Austin B, Allen DA, Mills AL, Colwell RR (1977) Numerical taxonomy of heavy metal tolerant bacteria isolated from an estuary. Can J Microbiol 23:1433–1447
Austin B, Garges S, Conrad B, Harding EE, Colwell RR, Simidu U, Taga N (1979) Comparative study of the aerobic heterotrophic bacterial flora of Chesapeake Bay and Tokyo Bay. Appl Environ Microbiol 37:704–714
Bell RT (1990) An explanation for the variability in the conversion factor deriving bacterial cell production from incorporation of [3H]thymidine. Limnol Oceanogr 35:910–915
Bell WH (1983) Bacterial utilization of algal extracellular products. 3. The specificity of the algal-bacterial interaction. Limnol Oceanogr 28:369–371
Bell WH, Sakshaug E (1980) Bacterial utilization of algal extracellular products. 2. A kinetic study of natural populations. Limnol Oceanogr 25:1021–1033
Cho BC, Azam F (1988) Heterotrophic bacterioplankton production measured by tritiated thymidine incorporation method. Arch Hydrobiol Beih Ergebn Limnol 31:153–162
Chróst R, Overbeck J, Wcislo R (1988) Evaluation of the [3H]thymidine method for estimating bacterial growth rates and production in lake water: reexamination and methodological comments. Acta Microbiol Pol 37:95–112
Cloern JE, Nichols FH (1985) Temporal dynamics of an estuary: San Francisco Bay. Hydrobiologia 129:1–237
Cloern JE, Cole BE, Wong RJL Alpine AE (1985) Temporal dynamics of estuarine phytoplankton: a case study of San Francisco Bay. Hydrobiologia 129:153–176
Cole BE (1989) Temporal and spatial patterns of phytoplankton production in Tomales Bay, California, USA. Estuar Coastal Shelf Sci 28:103–115
Cole JJ, Caracao NF, Strayer DL, Ochs C (1989) Bacterial production in freshwaters: ecosystem level constraints on conversion factors. Limnol Oceanogr 34:286–296
Colwell RR, Mills AL, Walker JD, Garcia-Tello P, Campos V (1978) Microbial ecology studies of the Metula oil spill in the Straits of Magellan. J Fish Res Bd Can 35:573–580
Conomos TJ (1979) San Francisco Bay: the urbanized estuary. Pacific Division, AAAS, San Francisco
Dahle AB, Laake M, (1982) Diversity dynamics of marine bacteria studied by immunofluorescent staining on membrane filters. Appl Environ Microbiol 43:169–176
Davis CL (1988) Uptake and incorporation of thymidine by bacterial isolates from an upwelling environment. Appl Environ Microbiol 55:1267–1272
DeLong EF, Franks DG, Alldredge AL (1993) Phylogenetic diversity of aggregate-attached vs free-living marine bacterial assemblages. Limnol Oceanogr 38:924–934
Ducklow HW, Kirchman DL, Quinby HL (1993) Bacterioplankton cell growth and macromolecular synthesis in seawater cultures during the North Atlantic Spring Bloom, May, 1989. Microb Ecol 24:125–144
Fuhrman JA, Azam F (1980) Bacterioplankton secondary production estimates for coastal waters of British Columbia, Antarctica and California. Appl Environ Microbiol 39:1085–1095
Fuhrman JA, Azam F (1982) Thymidine incorporation as a measure of heterotrophic bacterioplankton production in marine surface water: evaluation and field results. Mar Biol 66:109–120.
Fuhrman JA, McCallum K, Davis AL (1993) Phylogenetic diversity of marine microbial communities from the Atlantic and Pacific oceans. Appl Environ Microbiol 59:1294–1302
Garland JL, Mills AL (1991) Classification and characterization of heterotrophic microbial populations on the basis of patterns of community-level sole-carbon-source utilization. Appl Environ Microbiol 57:2351–2359
Hollibaugh JT (1979) Metabolic adaptation in natural bacterial populations supplemented with selected amino acids. Estuar Coastal Mar Sci 9:215–230
Hollibaugh JT (1988) Limitations of the [3H]thymidine method for estimating bacterial productivity due to thymidine metabolism. Mar Ecol Prog Ser 43:19–30
Hollibaugh JT, Wong PS (1992) Ethanol-extractable pools and the incorporation of thymidine, L-leucine and other substrates by bacterioplankton. Can J Microbiol 38:605–613
Hollibaugh JT, Fuhrman JA, Azam F (1980) Radioactively labeling of natural assemblages of bacterioplankton for use in trophic studies. Limnol Oceanogr 25:172–181
Hollibaugh JT, Wong PS, Azam F, Smith DC, Steward GF, Cole BE (1992) Measurement of bacterioplankton production in Antarctic coastal waters: comparison of thymidine and L-leucine methods and verification of labeling patterns. Antarctic J US 27:127–128
Jassby AD, Cloern JE, Powell TM (1993) Organic carbon sources and sinks in San Francisco Bay: variability induced by riverflow. Mar Ecol Prog Ser 95:39–54
Karl DM (1979) Measurements of microbial activity and growth in the ocean by rates of stable ribonucleic acid synthesis. Appl Environ Microbiol 38:850–860
Karl DM (1982) Selected nucleic acid precursors in studies of aquatic microbial ecology. Appl Environ Microbiol 44:891–902
Karl DM, Holm-Hansen O, Taylor GT, Tien G, Bird DF (1991) Microbial biomass and productivity in the western Bransfield Strait, Antarctica during the 1986–1987 austral summer. Deep-Sea Res 38:1029–1055
Kirchman DL, Hoch MP (1988) Bacterial production in the Delaware Bay estuary estimated from thymidine and leucine incorporation rates. Mar Ecol Prog Ser 45:169–178
Kirchman DL, K'Nees E, Hodson RE (1985) Leucine incorporation and its potential as a measure of protein synthesis by bacteria in natural aquatic systems. Appl Environ Microbiol 49:599–607
Kirchman DL, Suzuki Y, Garside C, Ducklow H (1991) High turnover rates of dissolved organic carbon during a spring phytoplankton bloom. Nature 352:612–614
Laake M, Dahle AB, Hertzschel G (1983) Productivity and population diversity of marine organotrophic bacteria in enclosed planktonic ecosystems. Mar Ecol Prog Ser 14:59–69
Lee S-H, Fuhrman JA (1990) DNA hybridization to compare species compositions of natural bacterioplankton assemblages. Appl Environ Microbiol 56:739–746
McDonough RJ, Sanders RW, Porter KG, Kirchamn DL (1986) Depth distribution of bacterial production in a stratified lake with an anoxic hypolimnion. Appl Environ Microbiol 52:992–1000
Moriarty DJW (1986) Measurement of bacterial growth rates in aquatic systems from rates of nucleic acid synthesis. Adv Microb Ecol 9:246–292
O'Donovan GA, Neuhard J (1970) Pyrimidine metabolism in microorganisms. Bacteriol Rev 34:278–343
Pomeroy LR (1974) The ocean's foodweb: a changing paradigm. BioScience 9:499–504
Pomeroy LR (1979) Secondary production mechanisms of continental shelf communities. In Livingston RJ (ed) Ecological processes in coastal marine systems. Plenum Publishing Co, New York, pp 163–186
Pomeroy LR (1980) Microbial roles in aquatic foodwebs. In Colwell RR, Foster J (eds) Aquatic microbial ecology. University of Maryland SeaGrant, College Park, Md, pp 85–109
Rehnstom A-S, Backman S, Smith DC, Azam F, Hagstrom A (1993) Blooms of sequence-specific culturable bacteria in the sea. FEMS Microbiol Ecol 102:161–166
Riemann B, Bell RT (1990) Advances in estimating bacterial biomass and growth in aquatic systems. Arch Hydrobiol 118:385–402
Riemann B, Bell RT, Jorgensen NOG (1990) Incorporation of thymidine, adenine, and leucine into natural bacterial assemblages. Mar Ecol Prog Ser 65:87–94
Robarts RD (1987) Effect of rainstorms on heterotrophic bacterial activity in a hypertrophic African lake. Hydrobiologia 148:281–286
Robarts RD, Wicks RJ (1989) [methyl-3H]thymidine uptake, macromolecular incorporation, and lipid labeling: their significance to DNA labeling during aquatic bacterial growth rate measurements. Limnol Oceanogr 34:213–222
Robarts RD, Zohary T (1993) Fact or fiction—bacterial growth rates and production as determined by [3H-methyl]thymidine? Adv Microb Ecol 13:371–425
Robarts RD, Wicks RJ, Sephton LM (1986) Spatial and temporal variations in bacterial macromolecule labeling with [methyl-3H]thymidine. Appl Environ Microbiol 52:1368–1373
Ryther JH (1969) Relationship of photosynthesis to fish production in the sea. Science 166:72–76
Servais PJ, Martinez J, Billen G, Vives-Rego J (1987) Determining [3H]thymidine incorporation into bacterioplankton DNA: improvement of the method by DNase treatment. Appl Environ Microbiol 53:1977–1979
Simon M, Azam F (1989) Protein content and protein synthesis rates of planktonic marine bacteria. Mar Ecol Prog Ser 51:201–213
Smith SV, Hollibaugh JT (1993) Coastal metabolism and the oceanic organic carbon balance. Rev Geophys 31:75–89
Smith SV, Hollibaugh JT, Dollar SJ, Vink S (1989) Tomales Bay, California: a case for carbon-controlled nitrogen cycling. Limnol Oceanogr 34:37–52
Smith SV, Hollibaugh JT, Dollar SJ, Vink S (1991) Tomales Bay metabolism: C-N-P stoichiometry and ecosystem heterotrophy at the land-sea interface. Estuar Coast Shelf Sci 33:223–257
Steele JDH (1974) The structure of marine ecosystems. Harvard University Press, Cambridge, Mass.
Torréton JP, Bouvy M (1991) Estimating bacterial DNA synthesis from [3H]thymidine incorporation: discrepancies among macromolecular extraction procedures. Limnol Oceanogr 36:299–306
Walker JD, Colwell RR (1976) Enumeration of petroleum degrading microorganisms. Appl Environ Microbiol 31:198–207
White DC, Bobbie RJ, Morrison SJ, Oosterhof DK, Taylor CW, Meeter DA (1977) Determination of microbial activity of estuarine detritus by relative rates of lipid biosynthesis. Limnol Oceanogr 22:1089–1099
Wicks RJ, Robarts RD (1987) The extraction and purification of DNA labeled with [methyl-3H]thymidine in aquatic bacterial population studies. J Plankton Res 9:1159–1166
Witzel K-P, Graf G (1984) On the use of different nucleic acid precursors for the measurement of microbial nucleic acid turnover. Arch Hydrobiol Beih Ergebn Limnol 19:59–65
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hollibaugh, J.T. Relationship between thymidine metabolism, bacterioplankton community metabolic capabilities, and sources of organic matter. Microb Ecol 28, 117–131 (1994). https://doi.org/10.1007/BF00166800
Issue Date:
DOI: https://doi.org/10.1007/BF00166800