Abstract
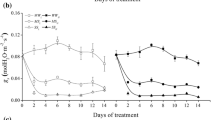
Cotton (Gossypium hirsutum L. cv. Acala SJ-2) seedlings were grown in modified Hoagland nutrient solution with or without 150 mM NaCl and supplemental 10 mM CaCl2. The spatial distribution of bulk-tissue osmotic potential (ΨS) and total osmotica, K, Na and Ca contents were determined in the growth zone of the primary root. This information was combined with the growth-velocity data from an earlier study (Zhong and Läuchli 1993) to estimate net deposition rates of osmoticum, water, K, Na and Ca by using the continuity equation. The ΨS was essentially uniform along the growth region for all treatments and considerably lowered by 150 mM NaCl in the medium. Total osmotica deposition was well synchronized with growth and deposition rates were enhanced by 150 mM NaCl. Osmoregulation in the treatments with 150 mM NaCl was indicated by an apparent solute accumulation which appeared to be due to the enhancement of osmoticum deposition rates. The presence of 150 mM NaCl greatly reduced the deposition rates of K and Ca throughout the growth zone; 10 mM Ca mitigated this effect only on K deposition in the apical 2.5-mm region. The deposition rate of Na was increased greatly by 150 mM NaCl; the increase was reduced by 10 mM Ca. At 150 mM NaCl, selectivity of K versus Na of the root was enhanced greatly in the apical 2mm region by the presence of 10 mM Ca; this mitigating effect by Ca declined rapidly with distance from the root tip. We conclude that one possible mechanism by which supplemental Ca alleviates the inhibitory effects of NaCl on cotton root growth is by maintaining plasma-membrane selectivity of K over Na.
Similar content being viewed by others
Abbreviations
- HCa, LCa:
-
high (10 mM) and low (1 mM) CaCl2
- HNa, LNa:
-
high (150 mM) and low (1 mM) NaCl
- ΨS :
-
osmotic potential
References
Campbell, N.A., Stika, K.M., Morrison, G.H. (1979) Calcium and potassium in the motor organ of the sensitive plant: Localization by ion microscopy. Science 204, 185–187
Chino, M. (1981) Species differences in calcium and potassium distributions within plant roots. Soil Sci. Plant Nutr. 27, 487–503
Cramer, G.R., Abdel-Basset, R, Seemann, J.R. (1990) Salinity-calcium interactions on root growth and osmotic adjustment of two corn cultivars differing in salt tolerance. J. Plant Nutr. 13, 1453–1462
Cramer, G.R., Läuchli, A., Polito, V.S. (1985) Displacementof Ca2+ by Na+ from the plasmalemma of root cells. Plant Physiol. 79, 207–211
Cramer, G.R., Lynch, J., Läuchli. A., Epstein, E. (1987) Influx of Na+, K+ and Ca2+ into roots of salt-stressed cotton seedlings. Effects of supplemental Ca2+. Plant Physiol. 83, 510–516
Epstein, E. (1961) The essential role of calcium in selective cation transport by plant cells. Plant Physiol. 36, 437–444
Erickson, R.O. (1976) Modelling of plant growth. Annu. Rev. Plant Physiol. 27, 407–434
Eshel, A., Waisel, Y. (1973) Variations in uptake of sodium and rubidium along barley roots. Physiol. Plant. 28, 557–560
Flowers, R.J., Troke, P.F., Yeo, A.R. (1977) The mechanism of salt tolerance in halophytes. Annu. Rev. Plant Physiol. 28, 89–121
Greenway, H., Munns, R. (1980) Mechanisms of salt tolerance in nonhalophytes. Annu. Rev. Plant Physiol. 31, 149–190
Hanson, J.B. (1984) The functions of calcium in plant nutrition. In: Advances in plant nutrition, vol. 1, 149–197, Tinker, P.B., Läuchli, A., eds. Praeger Publishers, New York
Henriksen, G.H., Raman, D.R., Walker, L.P., Spanswick, R.M. (1992) Measurement of net fluxes of ammonium and nitrate at the surface of barley root using ion-selective microelectrodes. II Patterns of uptake along the root axis and evaluation of the microelectrde estimation technique. Plant Physiol. 99, 734–747
Jeschke, W.D. (1979) Univalent cation selectivity and compartmentation in cereals. In: Recent advances in the biochemistry of cereals, pp. 37–61, Laidman, D.L., Wyn Jones, R.G., eds. Academic Press, New York
Jeschke, W.D. (1984) K+-Na- exchange at cellular membranes, intracellular compartmentation of cations, and salt tolerance. In: Salinity tolerance in plants. Strategies for crop improvement, pp. 37–66, Staples, R.C., Toenniessen, G.H., eds. John Wiley & Sons, New York
Jeschke, W.D., Stelter, W (1976) Measurement of longitudinal ion profiles in single roots of Hordeum and Atriplex by use of flameless atomic absorption spectroscopy. Planta 128, 107–112
Kent, L.M., Läuchli, A. (1985) Germination and seedling growth of cotton: salinity-calcium interactions. Plant, Cell Environ. 8, 155–159
Kochian, L.V., Shaff, J.E., Kühtreiber, W.M., Jaffe, L.F., Lucas, W.J. (1992) Use of an extracellular, ion-selective, vibrating microelectrode system for the quantification of K+, H+, and Ca2+ fluxes in maize roots and maize suspension cells. Planta 188, 601–610
Läuchli, A., Epstein, E. (1970) Transport of potassium and rubidium in plant roots. The significance of calcium. Plant Physiol. 45, 639–641
Lin, W, Wagner, J., Siegelman, H.W., Hind, G. (1977) Membranebound ATPase of intact vacuoles and tonoplasts isolated from mature plant tissue. Biochim. Biophys. Acta 465, 110–117
Lynch, J., Läuchli, A. (1984) Potassium transport in salt-stressed barley roots. Planta 161, 295–301
Miyasaka, S.C., Kochian, L.V., Shaff, J.E., Foy, C.D. (1989) Mecha nisms of aluminum tolerance in wheat: An investigation of genotypic differences in rhizosphere pH, K, and H transport, and root-cell membrane potentials. Plant Physiol. 91, 1188–1196
Nakamura, Y, Tanaka, K., Ohta, E., Sakata, M. (1990) Protective effect of external Ca2+ on elongation and the intracellular con centration of K+ in intact mung bean roots under high salt stress. Plant and Cell Physiol. 31, 815–821
Pitman, M.G., Saddler, H.D.W. (1967) Active sodium and potassium transport in cells of barley roots. Proc. Natl. Acad. Sci. USA 57, 44–49
Rains, D.W., Epstein, E. (1967) Sodium absorption by barley roots: role of the dual mechanisms of alkali cation transport. Plant Physiol. 42, 314–318
Sharp, R., Hsiao, T., Silk, W. (1990) Growth of the maize primary root at low water potentials. II. Role of growth and deposition of hexose and potassium in osmotic adjustment. Plant Physiol. 93, 1337–1346
Sharp, R.E., Silk, W.K., Hsiao, T.C. (1988) Growth of the maize primary root at low water potentials. I. Spatial distribution of expansive growth. Plant Physiol. 87, 50–57
Silk, W.K. (1984) Quantitative descriptions of development. Annu. Rev. Plant Physiol. 35, 479–518
Silk, W.K., Erickson, R.O. (1979) Kinematics of plant growth. J. Theor. Biol. 76, 481–501
Silk, W.K., Hsiao, T.C., Diedenhofen, U., Matson, C. (1986) Spatial distributions of potassium, solutes, and their deposition rates in the growth zone of the primary corn root. Plant Physiol. 82, 853–858
Silk, W.K, Walker, R.C., Labavitch, J. (1984) Uronide deposition rates in the primary root of Zea mays. Plant Physiol. 74, 721–726
Steward, F.C, Prevot, P., Harrison, J.A. (1942) Absorption and ac cumulation of rubidium bromide by barley plants. Localization in the root of cation accumulation and of transfer to the shoot. Plant Physiol. 17, 411–421
Zhong, H.L., Läuchli, A. (1993) Spatial and temporal aspects of growth in the primary root of cotton seedlings: Effects of NaCl and CaCl2. J. Exp. Bot. 44, 763–771
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors thank W.K. Silk for her advice and suggestions on this work, and her helpful discussion of the manuscript. J. Norlyn's technical assistance is also greatly appreciated. This work was partly supported by National Science Foundation grant DMB 84-04442.
Rights and permissions
About this article
Cite this article
Zhong, H., Läuchli, A. Spatial distribution of solutes, K, Na, Ca and their deposition rates in the growth zone of primary cotton roots: Effects of NaCl and CaCl2 . Planta 194, 34–41 (1994). https://doi.org/10.1007/BF00201032
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00201032