Summary
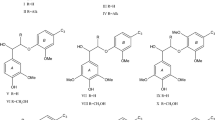
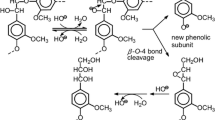
Delignification processes comprising various pulping and bleaching reactions are reviewed in the light of a general concept dividing these reactions into nucleophilic and electrophilic categories. It is shown by numerous examples that pulping is due to the action of external nucleophilic species attacking electron-deficient centres in carbonyl and conjugated carbonyl (=enone) structures and to the presence of neighbouring nucleophilic groups attacking the β-carbon atom of side chains. On the other hand, bleaching is initiated by electrophilic species attacking electron-rich centres in aromatic nuclei and unsaturated, ring-conjugated side chains. These initial bleaching reactions are followed by nucleophilic reactions taking place during the same or during subsequent bleaching steps.
The efficiency of a particular delignification procedure is determined by the extent of cooperation between nucleophilic and electrophilic reactions.
During delignification processes carbohydrate constituents of wood are subject to reactions which follow mechanisms analogous to those of lignins. This provides an explanation for the fact that the selectivity of pulping and bleaching processes is limited.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gierer, J. Chemistry of delignification. Wood Sci. Technol. 19, 289–312 (1985). https://doi.org/10.1007/BF00350807
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00350807