Summary
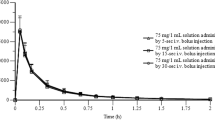
The pharmacokinetics of diclofenac were examined following single rapid intravenous injection and also following single oral doses to healthy female volunteers. After intravenous injection plasma levels of diclofenac fell rapidly and were below the limits of detection at 5.5 h postdosing. Individual drug profiles were described by a triexponential function and mean half-lives of the three exponential phases were 0.05, 0.26 and 1.1 h. After oral doses of enteric-coated tablets, the lag time between dosing and the appearance of drug in plasma varied between 1.0 and 4.5 h. However once drug absorption had commenced similar plasma drug profiles were obtained in different individuals. Peak plasma diclofenac levels ranged from 1.4 to 3.0 µg · ml−1. The mean terminal drug half-life in plasma was 1.8 h after oral doses. This value was not significantly greater than the value of 1.1 h following intravenous doses. Fifty percent of orally dosed diclofenac did not reach the systemic circulation due, predominantly, to first-pass metabolism.
Similar content being viewed by others
References
Boxenbaum, H. G., Riegelman, S., Elashoff, R.: Statistical estimations in pharmacokinetics. J. Pharmacokinet. Biopharm.2, 123–48 (1974)
Geiger, U. P., Degen, P. H., Sioufli, A.: Quantitative assay of diclofenac in biological material by gas-liquid chromatography. J. Chromatogr.111, 293–298 (1975)
Gibaldi, M., Perrier, D.: Pharmacokinetics. Vol. 1. Drugs and the pharmaceutical sciences. New York: Marcel Dekker 1975
Kendall, M. J., Thornhill, D. P., Willis, J. V.: Factors affecting the pharmacokinetics of diclofenac sodium. Rheumatol. Rehabil. (in press)
Krupp, P. J., Exer, B., Menassé, R., Ziel, R.: Neue Aspekte der Entzündungshemmung durch nicht-steroide Antiphlogistika: Wirkung von Voltaren. Schweiz. Med. Wochenschr.105, 646 (1975)
Krupp, P. J., Menassé-Gdynia, R., Sallmann, A., Wilhelmi, G., Ziel, R., Jaques, R.: Sodium (0-((2,6-dichlorophenyl)-amino)-phenyl)-acetate (GP 45,840) a new non-steroidal anti-inflammatory agent. Experientia29, 450 (1978)
Kornguth, M. L., Monson, R. A., and Kunin, C. M.: The binding of antibiotics to a soluble protein from rat liver. J. Infect. Dis.129, 552–558 (1974)
Kornguth, M. L., Monson, R. A., and Kunin, C. M.: The binding of penicillin antibiotics to a human liver protein. Arch. Biochem. Biophys.174, 339–343 (1976)
Menassé, R., Hedwall, P. R., Kraetz, J., Pericin, C., Riesterer, L., Sallmann, A., Ziel, R., Jaques, R.: Pharmacological properties of diclofenac sodium and its metabolites. Scand. J. Rheumatol. Suppl.22, 5–16 (1978)
Metzler, C. M.: A user's manual for NONLIN Technical Report 7292/69/7292/005, Upjohn Company, Kalamazoo, MI, November 25, 1969
Riess, W., Stierlin, H., Faigle, J. W., Geiger, U. P., Gerardin, A., Schmid, K., Sulc, M., Wagner, J., Theobald, W. P.: Kinetik und Biotransformation von Diclofenac in Tier und Mensch. Therapiewoche26, 18 (1976)
Riess, W., Stierlin, H., Degen, P., Faigle, J. W., Gerardin, A., Moppert, J., Sallmann, A., Schmid, K., Schweizer, A., Sulc, M., Theobald, W., Wagner, J.: Pharmacokinetics and metabolism of the anti-inflammatory agent Voltaren. Scand. J. Rheumatol. Suppl.22, 17–29 (1978)
Stierlin, H., Faigle, J. W. and Columbi, A.: Pharmacokinetics of diclofenac sodium (Voltaren) and metabolites in patients with impaired renal function. Scand. J. Rheumatol. Suppl.22, 30–35 (1978)
Wagner, J. G.: Do you need a pharmacokinetic model, and if so, which one? J. Pharmacokinet. Biopharm.3, 457–478 (1955)
Willis, J. V., Kendall, M. J.: Pharmacokinetic studies on diclofenac sodium in young and old volunteers. Scand. J. Rheumatol. Suppl.22, 36–41 (1978)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Willis, J.V., Kendall, M.J., Flinn, R.M. et al. The pharmacokinetics of diclofenac sodium following intravenous and oral administration. Eur J Clin Pharmacol 16, 405–410 (1979). https://doi.org/10.1007/BF00568201
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00568201