Summary
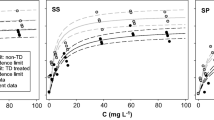
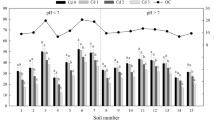
The kinetics of zinc desorption by DTPA were investigated in several soils. The rate of desorption of soil zinc and adsorbed zinc was rapid initially and gradually declined with time. The desorption reaction can be described by a two constant rate equation, C=AtB. The rate of zinc desorption in soil containing freshly adsorbed zinc was considerably higher than soil zinc, possibly due to the higher solubility and zinc concentration on the surfaces of soil particulate matter. The amount of zinc desorbed by DTPA, however, continued to decrease with increasing aging time. Elevated temperature further enhanced zinc aging and reduced zinc extractability. Recrystallization of adsorbed zinc in soil which subsequently increased the bonding strength of adsorbed zinc, could be responsible for the reduction of zinc desorption by DTPA.
Similar content being viewed by others
Literature Cited
Bingham, F. T., Page, A. L. and Sims, J. R. 1964 Retention of Cu and Zn by H-Montmorillonite, Soil Sci. Soc. Am. Proc.28, 351–354.
Boawn, L. C. 1974 Residue availability of fertilizer zinc. Soil Sci. Soc. Am. Proc.38, 800–804.
Elgawhary, S. M., Lindsay, W. L. and Kemper, W. D. 1970 Effect of EDTA on the self-diffusion of zinc in aqueous solution and in soil. Soil Sci. Soc. Am. Proc.34, 66–70.
Evans, R. L. and Jurinak, J. J. 1976 Kinetics of phosphate release from a desert soil. Soil Sci.121, 205–211.
Jackson, M. L. 1956 Soil chemical analysis-Advanced course. Published by the author. Dept. of Soil Sci., Univ. of Wisconsin, Madison.
Jurinak, J. J., Whitmore, J. C. and Wagenet, R. J. 1977 Kinetics of salt release from a saline soil. Soil Sci. Soc. Am. J.41, 721–724.
Kuo, S. and Mikkelsen, D. S. 1979 Zinc adsorption by two alkaline soils. Soil Sci.128, 274–279.
Kuo, S. and Lotse, E. G. 1979 Kinetics of phosphate adsorption and desorption by lake sediments. Soil Sci. Soc. Am. Proc.38, 50–54.
Lindsay, W. L. and Norvell, W. A. 1969 Development of a DTPA micronutrient soil test. Agron. Abstr. p. 84.
Mikkelsen, D. S. and Kuo, S. 1977 Zinc fertilization and behavior in flooded soils. Commonwealth Bureau of Soils. Special publication No.5. Commonwealth Agricultural Bureaux, Farnham Roya, England.
Nelson, J. L. and Melsted, S. W. 1955 The chemistry of zinc added to soils and clays. Soil Sci. Soc. Am. Proc.19, 439–443.
Olsen, R. A. 1975 Rate of dissolution of phosphate from minerals and soils. Soil Sci. Soc. Am. Proc.39, 634–639.
Randhawa, N. S. and Broadbent, F. E. 1965 Soil organic matter-metal complexes: Reaction of zinc with model compounds and humic acid. Soil Sci.99, 295–300.
Saeed, M. and Fox, R. L. 1977 Relation between suspension pH and zinc solubility in acid and calcareous soils. Soil Sci124, 199–204.
Shuman, L. M. 1975 The effect of soil properties on zinc adsorption by soil. Soil Sci. Soc. Am. J.39, 454–458.
Walkley, A. and Black, I. A. 1934 An examination of the Degtjaref method for determining soil organic matter and a proposed modification of chromic and titration method. Soil Sci.37, 29–38.
Udo, E. J., Bohn, H. L. and Tucker, T. C. 1970 Zinc adsorption by calcareous soils. Soil Sci. Soc. Am. Proc.34, 405–407.
Author information
Authors and Affiliations
Additional information
Contribution from the Department of Agronomy and Range Science, University of California, Davis, Ca.
Rights and permissions
About this article
Cite this article
Kuo, S., Mikkelsen, D.S. Kinetics of zinc desorption from soils. Plant Soil 56, 355–364 (1980). https://doi.org/10.1007/BF02143030
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02143030