Abstract
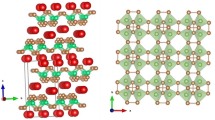
A phase transformation has been reported to occur at approximately 460 K in the compound Cu6Sn5, which has the NiAs (B81 structure. Single-crystal and polycrystalline specimens of this compound were heat treated to produce the high-temperature Ŋ-phase and the low-tem-perature Ŋ’-phase. X-ray diffraction determinations confirmed that the transformation was of the type: Simple Superlattice Ŋ Long-Period Superlattice Ŋ’ An analysis proposed by Pauling for phases with the NiAs structure indicated that the nature of bonding in Cu6Sn5 is predominantly metallic. The heat of formation and the energy effect associated with the Ŋ-Ŋ’ transformation were measured by liquid metal solution calorimetry with tin as solvent. The heat of formation of the Ŋ’-phase is approximately —1.68 kcal per g-at., which also suggests metallic bonding. The energy effect of the Ŋ -Ŋ ’ transformation is approximately -0.06 kcal per g-at. and is consistent with the nature of the transformation of a simple superlattice to a long-period superlattice.
Similar content being viewed by others
References
C. T. Heycock and F. H. Neville:Phil. Trans. Roy. Soc. London, Ser. A, 1904 vol. 202, p. 1.
T. Isihara:J. Inst. Metals, 1924, vol. 31, p. 315.
J. D. Bernai:Nature, 1928, vol. 122, p. 54.
M. Hamasumi:J. Inst. Metals, 1933, vol. 53, p. 550.
J. H. Hoke and V. D. Coombs:Trans. Amer. Foundrymen’s Soc., 1969, vol. 77, p. 7.
J. L. Haughton:J. Inst. Metals, 1921, vol. 25, p. 309.
G. V. Raynor:Annotated Equilibrium Diagram Series, No. 2, The Institute of Metals, London, 1944.
A. Westgren and G. Phragmen:Z. Anorg. Chem., 1928, vol. 175, p. 80.
O. Carlsson and G. Hägg:Z. Kristallog., 1932, vol. 83, p. 308.
B. W. Howlett, J. S. LI. Leach, L. B. Ticknor, and M. B. Bever:Rev. Sei Instrum., 1962, vol. 33, p. 619.
R. Hultgren, R. L. Orr, P. D. Anderson and K. K. Kelley: “Selected Values of Thermodynamic Properties of Metals and Alloys,” John Wiley, New York, 1963.
K. N. Tu:Acta Met, 1973, vol. 21, p. 347.
P. M. Robinson and M. B. Bever in “Intermetallic Compounds” (Ed. J. H. Westbrook), John Wiley, 1967, p. 38.
L. Pauling: “The Nature of the Chemical Bond”, Cornell U. Press, 1960.
R. Collonque in “The Physical Chemistry of Metallic Solutions and Inter-metallic Compounds-NPL Symposium”, H.M.S.O., 1969.
A. Gangulee and S. C. Moss:J. Appl. Crystallog., 1968, vol. 1, p. 61.
R. A. Oriani:Acta Met, 1954, vol. 2, p. 608.
P. L. Brooks and E. Gillam:Acta Met, 1970, vol. 18, p. 1181.
H. Sato and R. S. Toth in “Alloying Behavior and Effects in Concentrated Solid Solutions,” Gordon and Breach, New York, 1965.
R. W. Carpenter, R. L. Orr, and R. Hultgren:Trans. TMS-AIME, 1967, vol. 239, p. 107.
A. Gangulee and M. B. Bever:Trans. TMS-AIME, 1968, vol. 242, p. 278.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gangulee, A., Das, G.C. & Bever, M.B. An x-ray diffraction and calorimetric investigation of the compound Cu6Sn5 . Metall Trans 4, 2063–2066 (1973). https://doi.org/10.1007/BF02643268
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02643268