Abstract
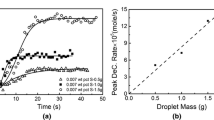
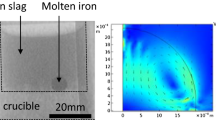
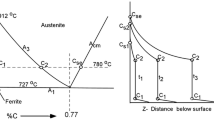
The chemical diffusion of oxygen in liquid iron oxide has been studied by the oxidation of a melt in a long capillary at 1615 °C. When pure oxygen was used as the oxidizing agent, the surface composition of the slag was found to be in close agreement with the expected gas-slag equilibrium, suggesting that diffusion is the controlling step. This was not the case when air, 5 pct oxygen in argon or pure CO2 was used to oxidize the slag. The deviation of the surface composition from the expected equilibrium was in accordance with a mechanism of mixed control by both the gas-slag reaction and diffusion in the bulk. The average value of the chemical diffusivity of oxygen (or iron) in liquid iron oxide with Fe2+/Fe T between 0.25 and 0.77 was established to be 3(±1) × 10-7 m2/s. This value is one to two orders of magnitude higher than those from earlier studies. There seems to be a reasonable correlation between the chemical and the ionic self-diffusivities through the Darken equation. A quantitative analysis in this respect and on the role of electron hole migration depends on the availability of data on the ionic conductivity and the tracer diffusivities.
Similar content being viewed by others
References
M. Sasabe and M. Jibiki:Can. Metall, Q., 1983, vol. 22, pp. 29–36.
P. Grieveson and E.T. Turkdogan:TMS-AIME, 1964, vol. 230, pp. 1609–14.
K. Mori and K. Suzuki:Trans. Iron Steel Inst. Jpn., 1969, vol. 9, pp. 409–12.
Y. Sasaki, S. Hara, D.R. Gaskell, and G.R. Belton:Metall. Trans. B, 1984, vol. 15B, pp. 563–71.
S.K. El-Rahaiby, Y. Sasaki, D.R. Gaskell, and G.R. Belton:Metall. Trans. B, 1986, vol. 17B, pp. 307–16.
S. Sun, Y. Sasaki, and G.R. Belton:Metall. Trans. B, 1988, vol. 19B, pp. 959–65.
F. Tsukihashi, K. Kato, K. Otsuka, and T. Soma:Trans. Iron Steel Inst. Jpn. 1982, vol. 22, pp. 688–95.
T. Nagasaka, Y. Iguchi, and S. Ban-ya:Tetsu-to-Hagané, 1985, vol. 71, pp. 204–11.
T. Nagasaka, Y. Iguchi, and S. Ban-ya:Process Technol. Proc, Iron Steel Soc, 1986, vol. 6, pp. 204–11.
T. Nagasaka and S. Ban-ya:Tetsu-to-Hagané, 1987, vol. 73, p. S773.
G.R. Belton:2nd Int. Symp. on Metallurgical Slags and Fluxes, TMS-AIME, Warrendale, PA, 1984, pp. 63–85.
A.M. Strachan: Ph.D. Thesis, University of Strathclyde, Glasgow, United Kingdom, 1974.
P. Grieveson:Physical Chemistry in Metallurgy, U.S. Steel Corp., Pittsburgh, PA, 1976, pp. 69–92.
E.T. Turkdogan and D.J. Goodwin:J. Iron Steel Inst., 1957, vol. 185, p. 104.
S. Hara, K. Irie, D.R. Gaskell, and K. Ogino:Trans. Jpn. Inst. Met., 1988, vol. 129, pp. 977–89.
L.S. Darken and R.W. Gurry:J. Am. Chem. Soc, 1946, vol. 68, pp. 798–816.
J. Crank:The Mathematics of Diffusion, Clarendon Press, Oxford, 1956, p. 30.
L.S. Darken:Physical Chemistry of Metals, McGraw-Hill, Inc., New York, NY, 1953, p. 442.
E.T. Turkdogan:Physicochemical Properties of Molten Slags and Glasses, Metals Society, London, 1983.
R.F. Johnston, R.A. Stark, and J. Taylor:Ironmaking and Steelmaking, 1974, pp. 220–27.
D.P. Agarwal and D.R. Gaskell:Metall. Trans. B, 1975, vol. 6B, pp. 263–67.
H. Keller and K. Schwerdtfeger:Metall. Trans. B, 1986, vol. 17B, pp. 497–501.
Y. Shiraishi, H. Nagahama, and H. Ohta:Can. Metall. g., 1983, vol. 22, pp. 37–43.
H. Keller, K. Schwerdtfeger, H. Petri, R. Holzle, and K. Hennesen:Metall. Trans. B, 1982, vol. 13B, pp. 237–40.
N. Nowak and K. Schwerdtfeger:Metal-slag-gas Reactions and Processes, Electrochemical Soc., Princeton, NJ, 1975, pp. 98–110.
H.-J. Engell and P. Vygen:Ber. Bunsengesell., 1968, vol. 72, pp. 5–12.
H. Inouye, J.W. Tomlinson, and J. Chipman:Trans. Faraday Soc, 1953, vol. 49, pp. 796–801.
M.T. Simnad, G. Derge, and I. George:Trans. AIME, J. Met., 1954, vol. 200, pp. 1386–90.
E.A. Dancy and G.J. Derge:TMS-AIME, 1966, vol. 236, pp. 1642–48.
P.J. Koros and T.B. King:TMS-AIME, 1962, vol. 224, pp. 299–306.
K.S. Goto, T. Kurahashi, and M. Sasabe:Metall. Trans. B, 1977, vol. 8B, pp. 523–28.
M. Sasabe and K.S. Goto:Metall. Trans., 1974, vol. 5, pp. 2225–33.
M. Sasabe and A. Asamura:2nd Int. Symp. on Metallurgical Slags and Fluxes, TMS-AIME, Warendale, PA, 1984, pp. 651–67.
M. Sasabe and S. Kitamura:Iron Steel Inst. Jpn. Int., 1993, vol. 33, pp. 133–39.
Author information
Authors and Affiliations
Additional information
Formerly Postgraduate Student
Rights and permissions
About this article
Cite this article
Sayadyaghoubi, Y., Sun, S. & Jahanshahi, S. Determination of the chemical diffusion of oxygen in liquid iron oxide at 1615 °c. Metall Mater Trans B 26, 795–802 (1995). https://doi.org/10.1007/BF02651726
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02651726