Abstract
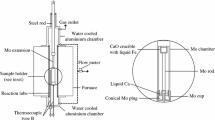
The equilibrium constants of Ce-S, La-S, and Nd-S in molten iron and the related interaction coefficients were determined experimentally and the following results obtained: KCeS = 2.80 × 10-6 eS Ce = −1.88 KLaS = 7.41 × 10-7 eS La= −1.51 KNdS = 2.57 × 10-6 eNd S= −1.51 In these experiments a radioactive isotope technique was used to determine the extremely low contents of the rare earth elements in the metal phase at equilibrium. In order to avoid errors caused by any partial inclusion of RE content of nonmetallic inclusions in the analytical results for the dissolved RE, the metal samples were electrolyzed in an organic electrolyte at low temperature, and the dissolved rare earth contents determined by measuring the radioactivity of the electrolyte. The dependence of the equilibrium values (concentration products) Ce-S on [pct C] in the system Fe-Ce-S-C was also determined. The interaction coefficient eCe C was determined to be −0.43. Earlier workers have obtained higher values for the Ce-S and La-S equilibrium constants than the present authors. Some explanation is offered for the difference.
Similar content being viewed by others
References
H. K. Lander and J. L. Mihelick: in “Molybdenum-ContainingSteels for Gas and Oil Industry Applications”, Climax Molybdenum Company (printed in U.S.A.), p. 2.
C.L. Briant and S.K. Banerji:Metall. Trans. A, 1981, vol. 12A, pp. 309–19.
F. C. Langenberg and J. Chipman:Trans. TMS-AIME, 1958, vol. 212, pp. 290–93.
R.H. Singleton:Trans. TMS-AIME, 1959, vol. 215, pp. 675–79.
DU Tin and Din Meizhi: Bau-tou, 1962, unpublished research.
K. Narita, A. Miymato, and E. Takajashi:Tetsu-to-Hagané, 1964, vol. 50, pp. 2011–13.
T. Kusakawa and T. Otani:Tetsu-to-Hagané, 1965, vol. 51, pp. 1987–90.
Z. Buzek, V. Schindlerova, and Hutn. Listy: 1966, vol. 3, pp. 169–75.
W. A. Fischer and H. Bertram:Arch. Eisenhüttenwes., 1973, vol. 44, pp. 97–109.
E. T. Turkdogan:Sulfide Inclusions in Steel, Proceedings of International Symposium, 7–8 November, 1974, Ohio, pp. 1–22.
Din Meizhi:Rare Earths and Niobium (in Chinese), 1975, no. 1.
A. Ejima, K. Suzuki, N. Marade, and K. Sanbongi:Trans, of the Iron and Steel Inst. of Japan, 1977, vol. 17, pp. 349–58;Tetsu-to-Hagané, 1975, vol. 61, pp. 2784-93.
E. Schurmann, J. Brauchwann, and H.J. Voss:Arch. Eisenhüttenwes., 1976, vol. 47, pp. c.l, 1-3.
A. Vahed and D.A.R. Kay:Metall. Trans. B, 1976, vol. 7B, pp. 375–83.
Wang Changzhen, Wang Fuzhen, DuYingmin, and Zhang Xiaopin:Acta Metallurgica Sinica, 1980, vol. 16, 1, pp. 83–89.
W. G. Wilson, D. A. R. Kay, and A. Vahed:Journal of Metals, 1974, vol. 26, 5, pp. 14–23.
Z. Buzek: inChemical Metallurgy of Iron and Steel, ISI, London, 1973, pp. 173–77.
R. J. Fruehan:Metall. Trans. B, 1979, vol. 10B, pp. 143–48.
A. McLean and W. K. Lu:Metals and Materials, 1974, vol. 8, pp. 452–57.
Han Qiyong, Liu Shiwei, Niu Hongbing, and Tan Zhiwei: First China-U.S.A. Bilateral Metallurgical Conference (Pre-Print) Nov. 1981, Beijing, pp. 192–207.
Han Qiyong, Wu Weijing, Fang Kerning, Huo Chengzhang, and Zhou Xiaolong: Sino-Japanese Symposium on Iron and Steel, First Symposium on Steelmaking, Oct. 1981, Beijing, China, pp. 25–43.
Han Qiyong, Liu Shiwei, Niu Hongbing, and Tan Zhiwei:Acta Metallurgical Sinica, 1982, vol. 18, 2, pp. 176–86.
G.K. Sigworth and J. F. Elliott:Metal Science, 1974, vol. 8, pp. 298–310.
E.T. Turkdogan:Physical Chemistry of High Temperature Technology, Academic Press, New York, NY, 1980, pp. 5–24.
F. KörberStahl u. Eisen, 1937, vol. 57, pp. 1349–55.
E. T. Turkdogan: “Clean Steel,” Proceedings in English of the Second International Conference on Clean Steel, 1–3 June, 1981, Balatonfured, Hungary, TMS, January 1983, pp. 75–121.
T. B. Reed:Free Energy of Formation of Binary Compounds, MIT, Cambridge, MA, 1971, p. 31.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Han, Q., Dong, Y., Feng, X. et al. Equilibria between rare earth elements and sulfur in molten iron. Metall Trans B 16, 785–792 (1985). https://doi.org/10.1007/BF02667514
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02667514