Abstract
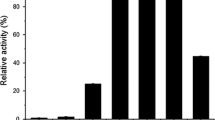
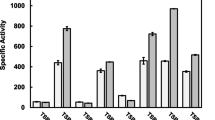
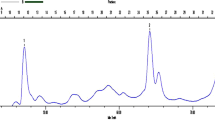
β-d-glucosidase purified from commercial preparations of clarified culture broth ofAspergillus niger (Novo SP188) was shown to elute as two distinct species during analytical anion-exchange chromatography (AEC). However, the two enzyme forms behaved identically on sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis (PAGE), high-performance size-exclusion chromatography (HPSEC), and isoelectric focusing. Also, the N-terminal amino acid sequence, amino acid composition, fingerprint of tryptic-digest peptides, circular dichroism spectra, and reaction kinetics appear identical for these forms. This feature of theA. niger enzyme is distinctly different from β-d-glucosidase isozymes reported from other sources, where multiple forms tend to differ in molecular weight and/or isoelectric pH. Michaelis-Menten kinetic analysis also gave comparable results for the two forms. The distinct behavior on AEC was explained by considering the differences in N-linked carbohydrates liberated from both species following treatment with endoglycosidase H or F.
Similar content being viewed by others
References
Wright, J. D., Power, A. J., and Douglas, L. J. (1986),Biotechnol. Bioeng. Symp. 17, 285–302.
Wright, J. D., Wyman, C. E., and Grohmann, K. (1988),Applied Biochem. Biotechnol. 17, 75–90.
Wilke, C. R., Yan, R. D., and von Stockar, U. (1976),Biotechnol. Bioeng. Symp. 6, 55.
King, K. W. and Vessal, M. I. (1976), inCellulases and Their Applications, vol. 95, Gould, R. F., ed., American Chemical Society, Washington, D.C., pp. 7–25.
Wood, T. M. and McCrae, S. I. (1981), inHydrolysis of Cellulose: Mechanisms of Enzymatic and Acid Catalysis, vol. 181. Brown, R. D. and Jurasek, L., eds., American Chemical Society, Washington, D.C., pp. 181–209.
Gritzali, M. and Brown, R. D. (1979), inHydrolysis of Cellulose: Mechanisms of Enzymatic and Acidic Catalysis, vol. 181. Brown, R. D. and Jurasek, L., eds., American Chemical Society, Washington, D.C., pp. 237–260.
Ryu, D. and Mandels, M. (1980),Enzyme Microb. Technol. 2, 91–102.
Murti, C. R. K. and Stone, B. A. (1961),Biochem. J. 78, 715.
Adikane, H. V. and Patil, M. B. (1985),Ind. J. Biochem. Biophys. 22, 97.
Witte, K. and Wartenberg, A. (1989),Acta Biotechnol. 9, 179.
Baker, J. O., Oh, K. K., Grohmann, K., and Himmel, M. E. (1988),Biotech. Lett. 10, 325–330.
Wood, T. M. (1981),Biochem. J. 121, 353–362.
Polacheck, I. and Cabib, E. (1981),Anal. Biochem. 117, 311–314.
Yphantis, D. A. (1964),Biochemistry 3, 297–317.
Wales, M., Adler, F. T., and Van Holde, K. E. (1951),J. Phys. Colloid Chem. 55, 145–161.
Cohn, E. J. and Edsall, J. R. (1943), inProteins, Amino Acids, and Peptides as Ions and Dipolar Ions. Reinhold, NY, pp. 370–381.
Knecht, R. and Chang, J.-Y. (1986),Anal. Chem. 58, 2375–2379.
Pastuszyn, A., Noland, B. J., Bazan, J. F., Fletterick, R. J., and Scallen, T. J. (1987),J. Biol. Chem. 262, 13,219–13,227.
Shoseyov, O., Bravdo, B.-A., Ikan, R., and Chet, I. (1988),Phytochemistry 27, 1973–1976.
Woodward, J. and Wiseman, A. (1982),Enzyme Microb. Technol. 4, 73–79.
Dekker, R. F. H. (1989),Appl. Biochem. Biotechnol. 23, 25–39.
Tanford, C. (1961),Physical Chemistry of Macromolecules, Wiley, NY.
Kopaciewicz, W., Rounds, M. A., Fausnaugh, J., and Regnier, F. E. (1983),J. Chromatogr. 266, 3.
Park, J. M., Muhoberac, B. B., Dubin, P. L., and Xia, J. (1992),Macromol. 25, 290.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Himmel, M.E., Adney, W.S., Fox, J.W. et al. Isolation and characterization of two forms of β-d-glucosidase fromAspergillus niger . Appl Biochem Biotechnol 39, 213–225 (1993). https://doi.org/10.1007/BF02918991
Issue Date:
DOI: https://doi.org/10.1007/BF02918991