Abstract
Introduction
We evaluated the sub-chronic toxicity of the aqueous herbal extract prepared fromCassytha filiformis and administered daily for 28 days at dose levels (250, 500, and 1000 mg/kg bw) in male wistar albino rats. The LD50 of the aqueous extract was determined.
Methods
The effects on body weights, organ weights, and certain haematological and plasma biochemical parameters were measured as indices of organ toxicity.
Results
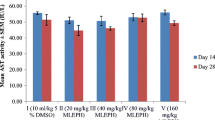
The aqueous extract did not affect plasma glutamate oxaloacetate transaminase (GOT) and glutamate pyruvate transaminase (GPT); however, a significant reduction in alkaline phosphatase (ALP) level occurred in all the treated groups. It also did not affect the electrolytes (Na+, Cl− and K+), total and direct bilirubin, creatinine, and glucose level. The aqueous extract elicited hypercholesterolaemic effects, but it did not affect the Hb, WBC, RBC, PVC, platelets, MCH, MCHC, MCV levels and differential counts (lympocytes, neutrophils, monocytes, eosinophils and basophils). It also reduced the body weight gain and absolute weight of the kidneys. The relative weights of the heart and lungs in some animal groups were equally reduced. The acute toxicological evaluation of the plant extract revealed an oral LD50 value greater than 500 mg/kg bw.
Conclusion
This study suggests that aqueous extract ofC. filiformis administered at normal therapeutic doses is not likely to produce severe toxic effects on some organs or haematological and biochemical indices in rats.
Article PDF
Similar content being viewed by others
References
Adedeji, AO. Rapid Interpretation of Routine Clinical Tests. Zaria, Nigeria: Asekome and Company,1992. 1981. pp. 16–31.
Abdullahi M, Mohammed G and Abdulkadir NU. Medicinal and Economic Plants of Nupeland. Bida, Nigeria: Jube — Evans Books and Publications;2003. pp. 140.
Akdogan M, Kilinc I, Oncu M, Karaoz E and Delibas N. Investigation of Biochemical and Histopathological Effects of Mentha piperita L and Mentha spicata L on Kidney Tissue in Rats.Human and Experimental Toxicology.2003;22:213–219.
Aniagu SO, Nwinyl FC, Akumka DD, Ajoku GA, Dzarma S, Izebe KS, et al. Toxicity Studies in Rats Fed Nature Cure Bitters.African Journal of Biotechnology.2005;4(1):72–78.
Dioka C, Orisakwe OE, Afonne OJ, Agbasi PU, Akumka DD, Okonkwo CJ and et al. Investigation into the haematologic and hepatoxic effects of rinbacin in rats.Journal of Health Science.2002;48(5):393–398.
Garba, (2000). Traditional Medical Practitioner. Zaria, Kaduna State, Nigeria.
Kallner A and Trydry N. IFCC Guidelines to the Evaluation of Drug Effects in Clinical Chemistry.Scandinanan Journal of Clinical and Laboratory Investigations 1989;195:1–29.
King DJ and Kelton JG. Heparin — Associated Thrombocytopaenia.Annuals of Internal Medicine. 1984;100:535–540.
Leclercq JQ, Hoet S, Block S, Wautier MC and Stevingny C. Studies on Cassytha filiformis from Benin: Isolation, Biological Activities and Quantification of Aporphines. Proceedings of Bioresources Towards Drug Discovery and Development2004; pp. 81–106.
Lin TJ, Su CC, Lan CK, Jiang DD, Tsai JL and Tsai MS. Acute Poisonings with Breynia officinalis — An Outbreak of Hepatoxicity.Journal of Clinical Toxicology.2003;41: 591–594.
Lorke D. A New Approach to Acute Toxicity Testing.Archives of Toxicology.1983;54:275–287.
Natural Institute of Environmental and Health Sciences. [webpage on the Internet]. Respect for life [updated 2002; cited2006 Nov 11]. Available from: http://www.nih.org/ factsheets.html
Neuwinger HD. African Traditional Medicine. A Dictionary of Plants’ Use and Applications.Medical Pharmacology.2000;99:1–12.
Stonard MD and Evans GO, (1995). Clinical Chemistry. In Ballantyne B, Marrs T, Turner P (eds). General and Applied Toxicology. Macmillan Press. London. pp. 247.
Synder R, Lea GW, Kocsis JJ and Witner, CM (1977). Bone marrow Depressant and Leuconogenic Actions of Benzene.Life Science. 21:1709–1722.
World Health Organization. Basic OECD Principles of Good Laboratory Practice. [updated 2004; cited2006 Nov 11] http://www.who.int/tdr/publications.
Zhu M, Low KT and Loung P. Protective effects of Plant formula on ethanolic—induced gastric lesions in rats.Phytotherapy Research.2002;16: 276–280.
Author information
Authors and Affiliations
Corresponding author
Additional information
There was no outside funding of any kind used for this study.
Rights and permissions
About this article
Cite this article
Babayi, H.M., Udeme, J.J.I., Abalaka, J.A. et al. Effect of oral administration of aqueous whole extract of Cassytha filiformis on haematograms and plasma biochemical parameters in rats. J. Med. Toxicol. 3, 146–151 (2007). https://doi.org/10.1007/BF03160930
Issue Date:
DOI: https://doi.org/10.1007/BF03160930