Abstract
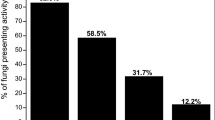
This research was designed to screen for strains that produce microbial oil by using straw as the substrate. One hundred and forty-one isolates of endophytic fungi were obtained from stems of seven oleaginous plant species. Sixty-nine isolates (48.9% of the total isolates) could be clearly seen having lipid bodies in their hyphae when examined with optical microscopy. Twenty-six isolates which had bigger and more oil bodies in their hyphae were selected for further research. These isolates belong to five genera includingMicrosphaeropsis, Phomopsis, Cephalosporium, Sclerocystis andNigrospora. Their oil contents ranged from 21.3 to 35.0% of dry cell weights when cultured in potato dextrose broth. When cultured on the solid-state medium composed of steam-exploded wheat straw (20% w/w), wheat bran (5%) and water (75%) they were able to produce cellulase and microbial oil with yields of 0.31≈0.69 filter paper unit and 19≈42 mg/g initial dry substrate, respectively. These results show that some endophytic fungi isolated from the oleaginous plants have the abilities of accumulating oil and producing cellulase simultaneously. They may be potential microbial oil producers by utilising straw as the substrate.
Similar content being viewed by others
References
Ballesteros M., Oliva J.M., Negro M.J., Manzanares P., Ballesteros I. (2004). Ethanol from lignocellulosic materials by a simultaneous saccharification and fermentation process (SSF) withKluyveromyces marxianus CECT 10875. Proc. Biochem., 39: 1843–1848.
Barnett H.L., Hunter B.B. (1998). Illustrated Genera of Imperfect Fungi. 4th edn., The American Phytopathological Society, Minnesota USA.
Considine P.J., O’Rorke A., Hackett T.J., Coughlan M.P. (1988). Hydrolysis of beet pulp polysaccharides by extracts if solid state cultures ofPenicillium capsulatum. Biotechnol. Bioeng., 31: 433–438.
Conti E., Stredansky M., Stredanska S., Zanetti F. (2001). ψ-Linolenic acid production by solid-state fermentation ofMucorales strains on cereals. Bioresour. Technol., 76: 283–286.
Dyal S.D., Bouzidi L., Narine S.S. (2005). Maximizing the production of ψ-linolenic acid inMortierella ramanniana var.ramanniana as a function of pH, temperature and carbon source, nitrogen source, metal ions and oil supplementation. Food Res. Int., 38: 815–829.
Dyal S.D., Narine S.S. (2005). Implications for the use ofMortierella fungi in the industrial production of essential fatty acids. Food Res. Int., 38: 445–467.
Gema H., Kavadia A., Dimou D., Tsagou V., Komaitis M., Aggelis G. (2002). Production of ψ-linolenic acid byCunninghamella echinulata cultivated on glucose and orange peels. Appl. Microbiol. Biotechnol., 58: 303–307.
Ghose T.K. (1987). Measurement of cellulase activities. Pure Appl. Chem., 59: 257–268.
Hannson L., Dostalek M. (1988). Effect of culture conditions on mycelian growth and production of ψ-linolenic acid by the fungusMortierella ramanniana. Appl. Microbiol. Biotechnol., 28: 240–246.
Kalogeris E., Fountoukides G., Kekos D., Macris B.J. (1999). Design of solid state bioreactor for thermophilic microorganisms. Biores. Technol., 67: 313–315.
Kang S.W., Park Y.S., Lee J.S., hong S.I., Kim S.W. (2004). Production of cellulases and hemicellulases byAspergillus niger KK2 from lignocellulosic biomass. Bioresour. Technol., 91: 153–156.
Lewis T., Nichols P.D., McMeekin T.A. (2000). Evaluation of extraction methods for recovery of fatty acids from lipid-producing microheterotrophs. J. Microbiol. Meth., 43: 107–116.
Li Z.F., Zhang L., Shen X.J., Lai B.S., Sun S.O. (2001). A comparative study on four methods of fungi lipid extraction. Chinese J. Microbiol., 28: 72–75.
Miao X.L., Wu Q.Y. (2006). Biodiesel production from heterotrophic microalgal oil. Bioresour. Technol., 97: 841–846.
Mo H.T., Zhang X.Y., Li Z.H. (2004). Control of gas phase for enhanced cellulase production byPenicillium decumbens in solid-state culture. Proc. Biochem., 39: 1293–1297.
Osono T., Takeda H. (1999). Decomposing ability of interior and surface fungi colonizers of beech leaves with reference to lignin decomposition. Eur. J. Soil Biol., 35: 51–56.
Papanikolaou S., Aggelis G. (2002). Lipid production byYarrowia lipolytica growing on industrial glycerol in a single-stage continuous culture. Bioresour. Technol., 82: 43–49.
Papanikolaou S., Chevalot I., Komaitis M., Marc I., Aggelis G. (2002). Single cell oil production byYarrowia lipolytica growing on an industrial derivative of animal fat in batch cultures. Appl. Microbiol. Biotechnol., 58: 308–312.
Papanikolaou S., Komaitis M., Aggelis G. (2004). Single cell oil (SCO) production byMortierella isabellina grown on high-sugar content media. Bioresour. Technol., 95: 287–291.
Ratledge C., Wynn J.P. (2002). The biochemistry and molecular biology of lipid accumulation in oleaginous microorganisms. Adv. Appl. Microbiol., 51: 1–51.
Ratledge C. (2004). Fatty acid biosynthesis in microorganisms being used for single cell oil production. Biochimie, 86: 807–815.
Shamala T.R., Sreekantiah K.R. (1986). Production of cellulases and D-xylanase by some selected fungal isolates. Enzyme Microb. Technol., 8: 178–182.
Stierle A., Strobel G., Stierle D. (1993). Taxol and taxane production byTaxomyces andreanae, an endophytic fungus of pacific yew. Science, 260: 214–216.
Strobel G., Yang X.S., Sears J., Kramer R., Sidhu R.S., Hess W.M. (1996). Taxol fromPestalotiopsis microspora, an endophytic fungus ofTaxus wallachiana. Microbiology, 142: 435–440.
Strobel G.A. (2003). Endophytes as sources of bioactive products. Microbes Infect., 5: 535–544.
Suto M., Takebayashi M., Saito K., Tanaka M., Yokota A., Tomita F. (2002). Endophytes as producers of xylanase. J. Biosci. Bioeng., 93: 88–90.
Sutton B.C. (1980). The Coelomycetes: Fungi Imperfecti with Pycnidia, Acervuli and Stromata. Commonwealth Agricultural Bureaux, Farnham Royal, Slough SL 23 BN, England.
Tengerdy R.P., Szakacs G. (2003). Bioconversion of lignocellulose in solid substrate fermentation. Biochem. Eng. J., 13: 169–79.
Tuohy M.G., Coughlan T.L., Coughlan M.P. (1990). Solid state versus liquid cultivation ofTalaromyces emersonii on straws and pulps: enzyme productivity. In: Coughlan M., Ed., Advances in Biological Treatments of Lignocellulosic Materials. Elsevier Applied Science. pp. 153–175.
Wang J.F., Li G.L., Lu H.Y., Zheng Z.H., Huang Y.J., Su W.J. (2000). Taxol fromTubercularia sp. strain TF5, an endophytic fungus ofTaxus mairei. FEMS Microbol. Lett., 193: 249–253.
Wang J.W., Wu J.H., Huang W.Y., Tan R.X. (2006). Laccase production byMonotospora sp., an endophytic fungus inCynodon dactylon. Bioresour. Technol., 97: 786–789.
Wei J.C. (1979). Manual of Determinative Fungi. Shanghai Scientific & Technical Publishers. Shanghai P.R. China.
Xu H., Miao X.L., Wu Q.Y. (2006). High quality biodiesel production from a microalgaChlorella protothecoides by heterotrophic growth in fermenters. J. Biotechnol., 126: 499–507.
Zhao R.J., Zheng Y.X. (1995). Studies on production of lipid in filamentous fungusMortierella sp. Acta Mycologica Sinica, 14: 130–135.
Zhu M., Zhou P.P., Yu L.J. (2002). Extraction of lipids fromMortierella alpina and enrichment of arachidonic acid from the fungal lipids. Bioresour. Technol., 84: 93–95.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peng, XW., Chen, HZ. Microbial oil accumulation and cellulase secretion of the endophytic fungi from oleaginous plants. Ann. Microbiol. 57, 239–242 (2007). https://doi.org/10.1007/BF03175213
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03175213