Abstract
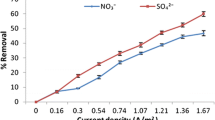
Water supply for consumption is one of the crucial objectives of water supply systems. Using of excessive fertilizer is a main source of nitrate content in water. The high amounts of nitrate in water have a determinable effect on the environment which must be removed due to drinking and industrial water standards. The purpose of this study is nitrate removal from aqueous solution by Electrocoagulation process. The applied pilot was comprised of a reservoir, electrode and power supply. In this study pH, electrical potential difference, nitrate initial concentration, total dissolved solid, kind of electrode, electrode connection methods and number of electrode were studied. Moreover, obtained optimum conditions were tested on Kerman water. The results showed that the electrocoagulation process can reach nitrate to less than standard limit. pH, electrical potential difference, total dissolved solids and number of electrodes have direct effect and initial concentration of nitrate has reverse effect on nitrate removal. This study also showed that under optimum condition, nitrate removal from Kerman water distribution system was 89.7 %. According to the results, Electrocoagulation process is suggested as an effective technique in nitrate removal.
Similar content being viewed by others
References
Abdel-Ghani, N. T.; Elchaghaby, G. A., (2007). Influence of operating conditions on the removal of Cu, Zn, Cd and Pb ions from wastewater by adsorption. Int. J. Environ. Sci. Tech., 4(4), 451–456 (6 pages).
Abdel-Ghani, N. T.; Hegazy, A. K.; El-Chaghaby, G. A., (2009). Typha domingensis leaf powder for decontamination of aluminium, iron, zinc and lead: Biosorption kinetics and equilibrium modeling. Int. J. Environ. Sci. Tech., 6(2), 243–248 (6 pages).
APHA, (1998). Standard methods for the examination of water and wastewater. 20th. Ed., American Public Health Association. American Water Works Associations. Wter Environment Federation, Washington Dc, USA.
Aslan, S.; Turkman, A., (2006). Nitrate and pesticides removal from contaminated water using biodenitrification reactor. Process Biochem., 41, 882–886 (4 pages).
Atafar, Z.; Mesdaghinia, A.; Nouri, J.; Homaee, M.; Yunesian, M.; Ahmadimoghaddam, M.; Mahvi, A. H., (2010). Effect of fertilizer application on soil heavy metal concentration. Environ. Monit. Assess., 160(1-4), 83–89 (7 pages).
Ayyasamy, P.M.; Shanthi, K.; Lakshmanaperumalsamy, P.; Lee, S.J.; Choi, N.; Kim, D., (2007). Two-Stage Removal of nitrate from groundwater using biological and chemical treatments. J. Biosci. Bioeng., 104, 129–134 (5 pages).
Babel, S.; Sae-Tang, J.; Pecharaply, A., (2009). Anaerobic co-digestion of sewage and brewery sludge for biogas production and land application. Int. J. Environ. Sci. Tech., 6(1), 131–140 (10 Pages).
Bae, B.; Junga, Y.; Hana, W.; Shin, H., (2002). Improved brine recycling during nitrate removal using ion exchange. Water Res., 36, 3330–3340 (10 pages).
Bandyopadhyay, G.; Chattopadhyay, S., (2007). Single hidden layer artificial neural network models versus multiple linear regression model in forecasting the time series of total ozone. Int. J. Environ. Sci. Tech., 4(1), 141–150 (10 pages).
Bazrafshan, E.; Mahvi, A. H.; Nasseri, S.; Shaieghi, M., (2007). Performance evaluation of electrocoagulation process for diazinon removal from aqueous environments by using iron electrodes. Iran J. Environ. Health Sci. Eng., 4(2), 127–132 (6 pages)
Drouiche, N.; Ghaffour, N.; Lounici, H.; Mameri, N.; Maallemi, A.; Mahmoudi, H., (2008). Electrocoagulation treatment of chemical mechanical polishing wastewater: removal of fluoride — sludge characteristics — operation cost. Desalination., 223, 134–142 (8 pages).
Emamjomeh, M. M.; Sivakumar, M., (2009). Review of pollutants removed by electrocoagulation and electrocoagulation/flotation processes. J. Environ. Manage., 90, 1663–1679 (16 pages).
Ghafari, S.; Hasan, M.; Aroua, M. K., (2008). Bio-electrochemical removal of nitrate from water and wastewater. A review: Bioresource Tech., 99, 3965–3974 (9 pages).
Ghernaout, D.; Badis, A.; Kellil, A.; Ghernaout, B., (2008). Application of electro coagulation in Escherichia coli culture and two surface waters. Desalination., 219, 118–125 (7 pages).
Hell, F.; Lahnsteiner, J.; Frischherz, H.; Baumgartner, G, (1998). Experience with full-scale electrodialysis for nitrate and hardness removal. Desalination., 117, 173–180 (7 pages).
Horold, S.; Vorlop, K.; Tacke, T.; Sell, M., (1993). Development of catalysts for a selective nitrate and nitrite removal from drinking water. Catalysis Today., 17, 21–30 (9 pages).
Hu, C. Y.; Lo, S. L.; Kuan, W. H., (2003). Effects of coexisting anions on fluoride removal in electrocoagulation (EC) process using aluminum electrodes. Water Res., 37, 4513–4523 (10 pages).
Igbinosa, E. O.; Okoh, A. I., (2009). Impact of discharge wastewater effluents on the physico-chemical qualities of a receiving watershed in a typical rural community. Int. J. Environ. Sci. Tech., 6(2), 175–182 (8 pages).
Igwe, J. C.; Abia, A. A.; Ibeh, C. A., (2008). Adsorption kinetics and intraparticulate diffusivities of Hg, As and Pb ions on unmodified and thiolated coconut fiber. Int. J. Environ. Sci. Tech., 5(1), 83–92 (10 Pages).
Jiang, J. Q.; Graham, N.; Andre, C.; Kelsall, G. H.; Brandon, N., (2002). Laboratory study of electro-coagulation-flotation for water treatment. Water Res., 36, 4064–4078 (14 pages).
Kashefialasl,M.; Khosravi, M.; Marandi, R.; Seyyedi, K., (2006). Treatment of dye solution containing colored index acid yellow 36 by electrocoagulation using iron electrodes. Int. J. Environ. Sci. Tech., 2(4), 365–371 (7 pages).
Kawamura, S. (2000). Integrated design and operation of water treatment facilities, New York, John Wiley and sons.
Kim, T.; Park, S.; Shin, E.; Kim, S., (2002). Decholorization of disperse and reactive dyes by continuous electro coagulation process. Desalination., 150, 165–175 (10 pages).
Kobya, M.; Hiz, H.; Senturk, E.; Aydiner, C.; Demirbas, E., (2006). Treatment of potato chips manufacturing wastewater by electro coagulation. Desalination., 190, 201–211 (10 pages).
Koparal, A..S., Ogutveren, U.B., (2002). Removal of nitrate from water by electroreduction and electrocoagulation, J. Hazard. Mater. 89, 83–94 (12 pages).
Lin, S.; Wu, C., (1996). Electrochemical removal of nitrite and ammonia for aquaculture. Water Res., 30, 715–721 (6 pages).
Luk, G.; Au-Yeung, W., (2002). Experimental investigation on the chemical reduction of nitrate from groundwater. Adv. Environ. Res., 6, 441–453 (12 pages).
Mahvi, A. H.; Nouri, J.; Babaei. A. A.; Nabizadeh, R., (2005). Agricultural activities impact on groundwater nitrate pollution. Int. J. Environ. Sci. Tech., 2(1), 41–47 (7 pages).
Malakootian, M.; Dowlatshahi, S., (2007). Variation of chemical quality for drinking water sources in Zarand plain. Iran. J. Environ. Health. Sci. Eng., 4(4), 257–262 (6 pages).
Malakootian, M.; Yousefi, N., (2009). The efficiency of electrocoagulation process using aluminum electrodes in removal of hardness from water. Iran. J. Environ. Health. Sci. Eng., 6, 131–136 (5 pages).
Mavrov, V.; Stamenov, S.; Todorova, E.; Chniel, H.; Erwe, T., (2006). New hybrid electro coagulation membrane process for removing selenium from industrial wastewater. Desalination., 20, 290–294 (4 pages).
Nabi Bidhendi, Gh.; Nasrabadi, T.; Sharif Vaghefi, H. R.; Hoveidi, H., (2006). Biological nitrate removal from water resources, Int. J. Environ. Sci. Tech., 3(3), 281–287 (7 pages).
Nouri, J.; Mahvi, A. H.; Jahed, G. R.; Babaei, A. A., (2008). Regional distribution pattern of groundwater heavy metals resulting from agricultural activities. Environ. Geol., 55(6), 1337–1343 (7 pages).
Nouri, J.; Mahvi, A. H.; Babaei, A. A.; Ahmadpour, E., (2006). Regional pattern distribution of groundwater fluoride in the Shush aquifer of Khuzestan County, Iran. Fluoride, 39(4), 321–325 (5 pages).
Paidar, M.; Bouzek, K.; Bergmann, H., (2002). Influence of cell construction on the electrochemical reduction of nitrate. Chem. Eng. J., 85, 99–109 (10 pages).
Paidar, M.; Rousar, I.; Bouzek, K., (1999). Electrochemical removal of nitrate ions in waste solutions after regeneration of ion exchange columns. J. Appl. Electrochem., 29, 611–617 (6 pages).
Pintar, A.; Batista, J.; Levec, J., (2001). Integrated ion exchange/catalytic process for efficient removal of nitrates from drinking water. Chem. Eng. Sci., 56, 1551–1559 (9 pages).
Rahmani, A., (2008). Removal of Water Turbidity by the Electroco-agulation Method. J Res Health Sci., 8(1), 18–24 (6 pages).
Ramesh, R.; Bhadrinarayana, N.; Meera, S.; Anantharaman, N., (2007). Treatment of tannery Waste water by electrocoagulation. J. Univ. Chem. Tech. Metall., 42, 201–206 (5 pages).
Ratna Kumar, P.; Chaudhari, S.; Khilar, K. C.; Mahajan, S. P., (2004). Removal of arsenic from water by electrocoagulation. Chemosphere., 55, 1245–1252 (7 pages).
Schulz, M. C.; Baygents, J. C.; Farrell, J., (2009). Laboratory and pilot testing of electrocoagulation for removing scale-forming species from industrial process waters. Int. J. Environ. Sci. Tech., 6(4), 521–526 (6pages).
Twort, A.; Ratnayaka, D.; Brandt, M., (2000). Water Supply, IWA Publishing, UK.
Ugurlu, M., (2004). The removal of some inorganic compounds from paper mill Effluents by the electrocoagulation method. G.U. J. Sci., 17(3), 85–99 (14 pages).
Vosoughifar, H. R.; Shamsi, A.; Ebadi, T., (2004). Discretization of flow contain nitrate in porous media by finite volume technique. Int. J. Environ. Sci. Tech., 1(4), 317–324 (7 pages).
Xiong, Y.; Strunk, P.; Hongyun, X.; Xihai, Z.; Karlsson, H., (2001). Treatment of dye wastewater containing acid orange II using a cell with three-phase three-dimensional electrode. Water Res., 35, 4226–4230 (6 pages).
Zvinowanda, C. M.; Okonkwo, J. O.; Shabalala, P. N.; Agyei, N. M., (2009). A novel adsorbent for heavy metal remediation in aqueous environments. Int. J. Environ. Sci. Tech., 6(3), 425–434 (10 pages).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Malakootian, M., Yousefi, N. & Fatehizadeh, A. Survey efficiency of electrocoagulation on nitrate removal from aqueous solution. Int. J. Environ. Sci. Technol. 8, 107–114 (2011). https://doi.org/10.1007/BF03326200
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03326200