Summary
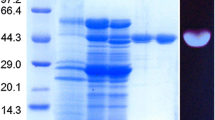
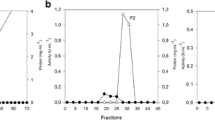
The β-xylosidase (EC 3.2.1.37) of Trichoderma reesei was purified and its characteristics and use in the hydrolysis of steamed birch xylan were studied. The enzyme was a glycoprotein with a molecular weight of 100000 as determined by SDS-gel electrophoresis and its isoelectric point was 4.7. The pH optimum was 4.0 and temperature optimum 60°C. β-Xylosidase was competitively inhibited by xylose and the inhibition constant was 2.3 mM. The purified enzyme also showed α-arabinofuranosidase activity.
Similar content being viewed by others
References
Dekker RFH (1983) Bioconversion of Hemicellulose: Aspects of Hemicellulase Production by Trichoderma reesei QM 9414 and Enzymatic Saccharification of Hemicellulose. Biotechnol Bioeng 30:1127–1146
Deleyn F, Claeyssens M, De Bruyne CK (1982) β-d-Xylosidase from Penicillium wortmanni. Meth Enzymol 83:639–644
Desphande V, Lachke A, Mishra C, Keskar S, Rao M (1986) Mode of action and properties of xylanase and β-xylosidase from Neurospora crassa. Biotechnol Bioeng 28:1832–1837
Van Doorslaer E, Kersters-Hilderson H, De Bruyne CK (1985) Hydrolysis of β-d-xylo-oligosaccharides by β-d-xylosidase from Bacillus pumilus. Carboh Res 140:342–346
Ebringerova A, Kramav A, Rendos F, Domansky R (1967) Die Stufenextraktion der Hemicellulosen aus dem Holz der Hagebuche (Carpinus betulus L.). Holzforschung 21:74–77
Esteban R, Chordi A, Villa TG (1983) Some aspects of a 1,4-β-d-xylanase and a β-xylosidase secreted by Bacillus coagulans strain 26. FEMS Microbiol Lett 17:163–166
Hodge JE, Hofreiter BT (1962) Determination of reducing sugars and carbohydrates. Meth Carboh Chem 1:390
Kersters-Hilderson H, Loontiens FG, Claeyssens M, De Bruyne, EK (1969) Partial purification and properties of an induced β-d-xylosidase of Bacillus pumilus 12. Eur J, Biochem 7:434–441
Kitpreechavanich V, Hayashi M, Nagai S (1986) Purification and characterization of extracellular β-xylosidase and β-glucosidase from Aspergillus fumigatus. Agr Biol Chem 50:1703–1711
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lappalainen A (1986) Purification and characterization of xylanolytic enzymes from Trichoderma reesei. Biotechnol Appl Biochem 8:437–448
Lowry OH, Rosebrough NH, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Matsuo M, Yasui T (1984a) Purification and some properties of β-xylosidase from Trichoderma viride. Agr Biol Chem 48:1845–1852
Matsuo M, Yasui T (1984b) Purification and some properties of β-xylosidase from Emericella nidulans. Agr Biol Chem 48:1853–1860
Oguntimein GB, Reilly PJ (1980) Properties of soluble and immobilized Aspergillus niger β-xylosidase. Biotechnol Bioeng 22:1143–1154
Poutanen K, Puls J, Linko M (1986) Hydrolysis of steamed birchwood hemicellulose by enzymes produced by Trichoderma reesei and Aspergillus awamori. Appl Microbiol Biotechnol 23:487–490
Poutanen K, Rättö M, Puls J, Viikari L (1987) Evaluation of microbial xylanolytic systems. J Biotechnol 6:49–60
Puls J, Poutanen K, Körner H-U, Viikari L (1985) Biotechnical utilization of wood carbohydrates after steaming pretreatment. Appl Microbiol Biotechnol 22:416–423
Roberts DV (1977) Enzyme kinetics. Cambridge Chemistry Texts, Cambridge
Rodionova NA, Tavobilov IM, Bezborodov AM (1983) β-Xylosidase from Aspergillus niger 15: Purification and properties. J Appl Biochem 5:300–312
Sumner JB, Somers GF (1949) Dinitrosalisylic method for glucose. In: Laboratory experiments in biological chemistry. Academic Press, New York, pp 38–39
Uziie M, Matsuo M, Yasui T (1985) Purification and some properties of Chaetomium trilaterale β-xylosidase. Agr Biol Chem 49:1159–1166
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Poutanen, K., Puls, J. Characteristics of Trichoderma reesei β-xylosidase and its use in the hydrolysis of solubilized xylans. Appl Microbiol Biotechnol 28, 425–432 (1988). https://doi.org/10.1007/BF00268208
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00268208