Summary
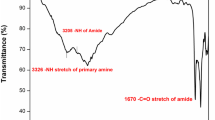
Aromatic-aliphatic polyamides were prepared from 2, 5-bis(4-carboxyethylcarbonylphenyl)-3,4-diphenylthiophene (BCDT) with few commercial diamines by using Yamazaki's phosphorylation reaction. The polymers were obtained in good yields and were characterized by solubility tests, viscosity measurements, IR spectroscopy, thermal analysis and X-ray diffraction studies. These polyamides had inherent viscosities in the range of 0.35 to 0.70 dl/g and were amorphous in nature. All the polyamides were readily soluble in solvents such as N,N-dimethylacetamide, N-methyl-2-pyrrolidinone, N,N-dimethylformamide, nitrobenzene, hexamethylphosphoramide, m-cresol, sulphuric acid and did not lose weight below 300°C in air.
Similar content being viewed by others
References
Imai, Y; Maldar, N.N.; and Kakimoto, M; J. Polym. Sci; Polym. Chem. Ed; 23 1797 (1985).
Scariah, K.J.; Krishnamurthy, V.N.; Rao, K.V.C.; and Srinivasan, M; Macromol. Chem; 186, 2427 (1985).
Srinivasan, P.R.; Mahadevan, V; and Srinivasan, M; J. Polym. Sci; Polym. Chem. Ed; 19, 2275 (1981).
Feld, W. A.; Ramalingam, B; and Harris, F. W.; J.Poly. Sci; Polym. Chem. Ed; 21, 319 (1983).
Mahajan, S.S.; Wadgaonkar, P.P.; and Chavan, N.N.; Intern. J. Polymeric Mater; 12, 101 (1988).
Imai, Y; Maldar, N.N.; and Kakimoto, M; J. Polym. Sci.; Polym. Chem. Ed; 22, 2189 (1984).
de Meester, P; Maldar, N.N.; Hosmane, N.S.; Chu, S.S.C; Acta Crystallogr. Sect. C: Cryst. Struct. Commun; C42, 363 (1986).
Yamazaki, N; Matsumoto, M; and Higashi, F; J. Polym. Sci; Polym. Chem. Ed: 13, 1373 (1975).
Author information
Authors and Affiliations
Additional information
NCL Communication No:-4815
Rights and permissions
About this article
Cite this article
Mahajan, S.S., Sarwade, B.D. & Maldar, N.N. Synthesis and characterization of aromatic-alipathic polyamides containing tetraphenylthiophene units into the backbone. Polymer Bulletin 24, 143–149 (1990). https://doi.org/10.1007/BF00297310
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00297310