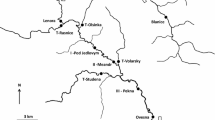
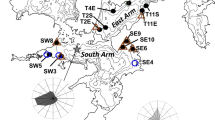
Abstract
Anthropogenic disturbances often cause decline and extinction of threatened species. The present study investigated how gravid freshwater mussels, Margaritifera margaritifera, were affected by turbidity and water temperature, and by mussel density. At an early date of mussel spawning, there were lower proportions of gravid mussels in streams with evidence of mussel recruitment than in streams without mussel recruitment. At a late spawning date, this pattern was reversed. Higher water temperature in streams without recruitment was probably responsible for this difference. The combination of high water temperature and turbidity may be one reason for reduced growth of gravid mussels in streams without recruitment. There was a positive relationship between adult mussel density and the proportions of gravid mussels. Early gravidity may lead to early release of larvae, early infestation on the host fish and an earlier start of the benthic phase, which may reduce survival rates. Clear-cutting of forests and global warming are factors that are likely to cause increased turbidity/sedimentation and water temperatures in streams. One restoration measure that reduces sediment input and water temperatures is maintaining or restoring riparian zones, but these are long-term measures that require many years before they have an effect in streams.






Similar content being viewed by others
References
Arvidsson BL, Karlsson J, Österling ME (2012) Recruitment of the threatened mussel Margaritifera margaritifera in relation to mussel population size, mussel density and host density. Aquat Conserv Mar Freshw Ecosyst 22:526–532
Bauer G (1987a) Reproductive strategy of the freshwater pearl mussel Margaritifera margaritifera. J Anim Ecol 56:691–704
Bauer G (1987b) The parasitic stage of the freshwater pearl mussel (Margaritifera margaritifera L.) 3. Host relationships. Arch Hydrobiol 76:413–423
Downing JA, Rochon Y, Perusse M, Harvey H (1993) Spatial aggregation, body-size, and reproductive success in the fresh-water mussel Elliptio complanata. J N Am Benthol Soc 12:148–156
Feld CK (2013) Response of three lotic assemblages to riparian and catchment-scale land use: implications for designing catchment monitoring programmes. Freshw Biol 58:715–729
Floury M, Usseglio-Polatera P, Ferreol M, Delattre C, Souchon Y (2013) Global climate change in large European rivers: long-term effects on macroinvertebrate communities and potential local confounding factors. Global Change Biol 19:1085–1099
Galbraith HS, Vaughn CC (2009) Temperature and food interact to influence gamete development in freshwater mussels. Hydrobiologia 636:35–47
Haag WR, Warren ML (1998) Role of ecological factors and reproductive strategies in structuring freshwater mussel communities. Can J Fish Aquat Sci 55:297–306
Hanssen-Bauer I, Achberger C, Benestad R, Chen D, Forland E (2005) Statistical downscaling of climate scenarios over Scandinavia. Clim Res 29:255–268
Hastie LC, Young MR (2001) Freshwater pearl mussel (Margaritifera margaritifera) glochidiosis in wild and farmed salmonid stocks in Scotland. Hydrobiologia 445:109–119
Hastie LC, Young MR (2003) Timing of spawning and glochidial release in Scottish freshwater pearl mussel (Margaritifera margaritifera) populations. Freshw Biol 48:2107–2117
Klemetsen A, Amundsen PA, Dempson JB, Jonsson B, Jonsson N, O'connell MF, Mortensen E (2003) Atlantic salmon Salmo salar L., brown trout Salmo trutta L. and Arctic charr Salvelinus alpinus (L.): a review of aspects of their life histories. Ecol Freshw Fish 12:1–59
Kasangaki A, Chapman LJ, Balirwa J (2008) Land use and the ecology of benthic macroinvertebrate assemblages of high-altitude rainforest streams in Uganda. Freshw Biol 53:681–697
Kreutzweiser DP, Capell SS (2001) Fine sediment deposition in streams after selective forest harvesting without riparian buffers. Can J Forest Res 31:2134–2142
Kreutzweiser DP, Capell SS, Holmes SB (2009) Stream temperature responses to partial-harvest logging in riparian buffers of boreal mixedwood forest watersheds. Can J Forest Res 39:497–506
Larsen S, Ormerod SJ (2010a) Low-level effects of inert sediments on temperate stream invertebrates. Freshw Biol 55:476–486
Larsen S, Ormerod SJ (2010b) Combined effects of habitat modification on trait composition and species nestedness in river invertebrates. Biol Conserv 143:2638–2646
Larsen S, Vaughan IP, Ormerod SJ (2009) Scale-dependent effects of fine sediments on temperate headwater invertebrates. Freshw Biol 54:203–219
Österling EM (2011) Test and application of a non-destructive photo-method investigating the parasitic stage of the threatened mussel Margaritifera margaritifera on its host fish Salmo trutta. Biol Conserv 144:2984–2990
Österling EM, Högberg JO (2014) The impact of land use on the mussel Margaritifera margaritifera and its host fish Salmo trutta. Hydrobiologia 735:213–220. doi:10.1007/s10750-013-1501-1
Österling EM, Bergman E, Greenberg LA, Baldwin BS, Mills EL (2007) Turbidity-mediated interactions between invasive filter-feeding mussels and native bioturbating mayflies. Freshw Biol 52:1602–1610
Österling EM, Greenberg LA, Arvidsson BL (2008) Relationship of biotic and abiotic factors to recruitment patterns in Margaritifera margaritifera. Biol Conserv 141:1365–1370
Österling ME, Arvidsson BL, Greenberg LA (2010) Habitat degradation and the decline of the threatened mussel Margaritifera margaritifera: influence of turbidity and sedimentation on the mussel and its host. J Appl Ecol 47:759–768
Quinn GP, Keough MJ (2002) Experimental design and data analysis for biologists. Cambridge University Press, Cambridge, pp 1–537
Quinn JM, Wright-Stow AE (2008) Stream size influences stream temperature impacts and recovery rates after clearfell logging. For Ecol Manag 256:2101–2109
Richter BD, Braun DP, Mendelson MA, Master LL (1997) Threats to imperiled freshwater fauna. Conserv Biol 11:1081–1093
Strayer DL, Downing JA, Haag WR, King TL, Layzer JB, Newton TJ, Nichols SJ (2004) Changing perspectives on pearly mussels, North America’s most imperiled animals. Bioscience 54:429–439
Underwood AJ (1997) Experiments in ecology: their logical design and interpretation using analysis of variance. Cambridge University Press, Cambridge, pp 1–504
Wilcove DS, Rothstein D, Dubow J, Phillips A, Losos E (1998) Quantifying threats to imperiled species in the United States. Bioscience 48:607–615
Wood PJ, Armitage PD (1997) Biological effects of fine sediment in the lotic environment. Environ Managem 21:203–217
Young M, Williams J (1984) The reproductive biology of the freshwater pearl mussel Margaritifera margaritifera (Linn.) in Scotland 1. Field studies. Arch Hydrobiol 99:405–422
Acknowledgments
I thank Fortums’ Nordiska Miljöfond, the Knowledge Foundation and Karlstad University for financing this study, all the landowners for permission to work on their properties, Linda Bång for field assistance, the Editor, one anonymous reviewer, Larry Greenberg and John J Piccolo for commenting earlier versions of this manuscript, and Håkan Söderberg and Andreas Karlberg at the County Administration in Västernorrland for information about the mussel populations. The study was performed with permission from the County Administration in Västernorrland.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Österling, E.M. Timing, growth and proportion of spawners of the threatened unionoid mussel Margaritifera margaritifera: influence of water temperature, turbidity and mussel density. Aquat Sci 77, 1–8 (2015). https://doi.org/10.1007/s00027-014-0366-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00027-014-0366-3