Abstract
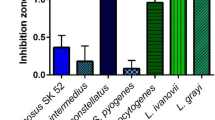
Recently, we reported the synthesis of five bacteriocin-like inhibitor substances (Bt-BLIS: morricin 269, kurstacin 287, kenyacin 404, entomocin 420, and tolworthcin 524) by Mexican strains of Bacillus thuringiensis. Here we show that, collectively, these Bt-BLIS have a moderate to broad spectrum of antibacterial activity, being toxic to clinically significant against Gram-positive and Gram-negative bacteria, including common etiological agents of human diseases, such as strep throat and scarlet fever, septicemia, pneumonia, urinary tract infection, and emetic and gastrointestinal syndromes. Although synthesis of the five Bt-BLIS was independent of the presence of a target inducing bacterium, we demonstrated for the first time that a proteinaceous component(s) secreted by, or liberated by proteolytic cleavage of Bacillus cereus 183 following treatment with proteinase K, enhanced Bt-BLIS synthesis.



Similar content being viewed by others
References
Ahern M, Verschueren S, van Sinderen D (2003) Isolation and characterization of a novel bacteriocin produced by Bacillus thuringiensis strain B439. FEMS Microbiol Lett 220:127–131
Alvarado C, Garcia-Almendarez BE, Martin SE, Regalado C (2005) Anti-Listeria monocytogenes bacteriocin-like inhibitory substances from Enterococcus faecium UQ31 isolated from artisan Mexican-style cheese. Curr Microbiol 51:110–115
Barboza-Corona JE, Vázquez-Acosta H, Bideshi DK, Salcedo-Hernández R (2007) Bacteriocin-like inhibitor substances produced by Mexican strains of Bacillus thuringiensis. Arch Microbiol 187:117–126
Barboza-Corona JE, Reyes-Rios DM, Salcedo-Hernandez R, Bideshi DK (2008) Molecular and biochemical characterization of an endochitinase (ChiA-HD73) from Bacillus thuringiensis subsp. kurstaki HD–73. Mol Biotechnol 39:29–37
Biswas SR, Ray P, Johnson C, Ray B (1991) Influence of growth conditions on the production of a bacteriocin, pediocin AcH, by Pediococcus acidilactici AcH. Appl Environ Microbiol 57:1265–1267
Chehimi S, Delalande F, Sablé S, Hajlaoui MR, Van Dorsselaer A, Limam F, Pons AM (2007) Purification and partial amino acid sequence of thuricin S, a new anti-Listeria bacteriocin from Bacillus thuringiensis. Can J Microbiol 53:284–290
Chen H, Hoover DG (2003) Bacteriocins and their food applications. Comprehensive reviews in food science and food safety. Institute of Food Technologist 2:82–100
Cherif A, Ouzari H, Cherif H, Daffonchio , Ben-Slama K, Hassen A, Jaoua A, Boudabous A (2001) Thuricin 7: a novel bacteriocin produced by Bacillus thuringiensis BMG1.7, a new strain isolated from soil. Lett Appl Microbiol 32:243–247
Cherif A, Chehimi S, Limen F, Hansen BM, Hendriksen NB, Daffonchio D, Boudabous A (2003) Detection and characterization of the novel bacteriocin entomocin 9, and safety evaluation of its producer, Bacillus thuringiensis ssp. Entomocidus HD-9. J Appl Microbiol 95:990–1000
De la Fuente-Salcido N, Salcedo-Hernández R, Alanis-Guzmán MG, Bideshi DK, Barboza-Corona JE (2007) A new rapid fluorogenic method for measuring bacteriocin activity. J Microbiol Meth 70(1):196–199
Delgado A, Brito D, Fevereiro P, Tenreiro R, Peres C (2005) Bioactivity quantification of crude bacteriocin solutions. J Microbiol Meth 62:121–124
Diep DB, Havarstein LS, Nes IF (1995) A bacteriocin-like peptide induces bacteriocin synthesis in Lactobacillus plantarum C11. Mol Microbiol 18:631–639
Diep DB, Axelsson L, Grefsli C, Nes IF (2000) The synthesis of the bacteriocin sakacin A is a temperature-sensitive process regulated by a pheromone peptide through a three-component regulatory system. Microbiol 146:155–2160
Favret ME, Yousten AA (1989) Thuricin: the bacteriocin produced by Bacillus thuringiensis. J Invertebr Pathol 53:206–216
Gray EJ, Lee KD, Souleimanov AM, Di Falco MR, Zhou X, Ly A, Charles TC, Driscoll BT, Smith DL (2006a) A novel bacteriocin, thuricin 17, produced by plant growth promoting rhizobacteria strain Bacillus thuringiensis NEB17: isolation and classification. J Appl Microbiol 100:545–554
Gray EJ, Di Falco MR, Souleimanov A, Smith DL (2006b) Proteomic analysis of the bacteriocin thuricin 17 produced by Bacillus thuringiensis NEB 17. FEMS Microbiol Lett 255:27–32
Kamoun F, Mejdoub H, Aouissaoui H, Reinbolt J, Hammami A, Jaoua S (2005) Purification, amino acid sequence and characterization of Bacthuricin F4, a new bacteriocin produced by Bacillus thuringiensis. J Appl Microbiol 98:881–888
Kleerebezen M, Quadri LEN (2001) Peptide pheromone-dependent regulation of antimicrobial peptide production in Gram-positive bacteria: a case of multicellular behaviour. Peptides 22:1579–1596
Klein C, Kaletta C, Entian KD (1993) Biosynthesis of the lantibiotic subtisilin is regulated by a histidine kinase/response regulator system. Appl Environ Microbiol 59:296–303
Kuipers OP, Beerthuyzen MB, de Ruyter PGGA, Luesink EJ, de Vos WM (1995) Autoregulation of nisin biosynthesis in Lactococcus lactis by signal transduction. J Biol Chem 270:27299–27304
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Maldonado A, Ruiz-Barba JL, Jiménez-Díaz R (2004) Production of plantaricin NC8 by Lactobacillus plantgarum NC8 is induced in presence of different types of gram-positive bacteria. Arch Microbiol 181:8–16
Matthewes KR (2004) Here, there, everywhere: antibiotic-resistant foodborne pathogens. Food Technol 58:104
Morales de laVega L, Barboza-Corona JE, Aguilar-Uscanga MG, Ramírez-Lepe M (2006) Purification and characterization of an exochitinase from Bacillus thuringiensis ssp. aizawai and its action against phytopathogenic fungi. Can J Microbiol 52:651–657
Nilsen T, Nes IF, Holo H (1998) An exporter inducer peptide regulates bacteriocins production in Enterococcus faecium CTC492. J Bacteriol 180:1848–1854
Olasupo NA, Fitzgerald DJ, Gasson MJ, Narbad A (2003) Activity of natural antimicrobial compounds against Escherichia coli and Salmonella enterica serovar typhimurium. Lett Appl Microbiol 37:448–451
Paik HD, Bae SS, Park SH, Pan JG (1997) Identification and partial characterization of tochicin, a bacteriocin produced by Bacillus thuringiensis subsp. tochigiensis. J Ind Microbiol Biotechnol 19:294–298
Prescott LM, Harley JP, Klein DA (2005) The epidemiology of infectious disease. In: Prescott LM, Harley JP, Klein DA (eds) Microbiology, 6th edn, McGraw-Hill, New York, pp 821–843
Schnepf E, Crickmore N, VanRie J, Lereclus D, Baum J, Feitelson J, Zeigler DR, Dean DH (1998) Bacillus thuringiensis and its pesticidal crystal proteins. Microbiol Mol Biol Rev 62:775–806
Silo-Suh LA, Stabb EV, Raffel S, Hendelsman J (1998) Target range of zwittermicin A, an aminopolyol antibiotic from Bacillus cereus. Curr Microbiol 37:6–11
Simon L, Frémaux C, Cenatiempo Y, Berjeaud JM (2002) Sakacin G, a new type of antilisterial bacteriocin. Appl Environ Microbiol 12:6416–6420
Swiecicka I (2008) Natural occurrence of Bacillus thuringiensis in eukaryotic organisms: a case for symbiosis. Biocontrol Sci Technol 18:221–239
Yang R, Ray B (1994) Factors influencing production of bacteriocins by lactic acid bacteria. Food Microbiol 11:281–291
Acknowledgments
We thank Gabriela Morales-Pérez and Roberto Ortega Monzon, undergraduate students from Universidad de Guanajuato, Mexico, and Janeth Adriana Martínez-Cardeñas from Universidad Juarez Autonoma de Tabasco, Mexico, for their technical assistance. Norma de la Fuente-Salcido is a graduate student supported by a fellowship sponsored by CONACYT-México. The authors are grateful for the essential material provided by Jorge Ibarra, Irais Sánches-Ortega and Ma. Alejandra Chavira-Zuñiga. This research was supported partially by a grant (DINPO 2006) from the Universidad de Guanajuato to JEB-C.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Erko Stackebrandt.
Rights and permissions
About this article
Cite this article
de la Fuente-Salcido, N., Guadalupe Alanís-Guzmán, M., Bideshi, D.K. et al. Enhanced synthesis and antimicrobial activities of bacteriocins produced by Mexican strains of Bacillus thuringiensis . Arch Microbiol 190, 633–640 (2008). https://doi.org/10.1007/s00203-008-0414-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-008-0414-2