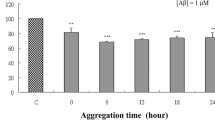
Abstract
β-amyloid is strongly implicated in Alzheimer’s pathology, and mitochondria play an important role in neurodegenerative disorders. Dactylorhin B [short for bis(4-β-D-glucopyranosyloxybenzyl)-2-β-D-glucopyranosyl-2-isobutyltartrate (DHB)] is an active compound isolated from Coeloglossum viride. (L.) Hartm. var. bracteatum (Willd.) and may have neuroprotective effects. In the present study, we investigated the damage of rat brain mitochondrial function induced by β-amyloid and the protective effect of DHB. Isolated rat brain mitochondria were incubated with amyloid-β peptide (Aβ)25–35 alone or together with DHB. reactive oxygen species production, pyruvate dehydrogenase complex activity, α-ketoglutarate dehydrogenase complex activity, cytochrome c oxidase activity, mitochondrial swelling, mitochondrial membrane potential and the release of cytochrome c from mitochondria were measured. Data showed that Aβ25–35 directly disrupted mitochondrial function, inhibited the key enzymes and contributed to apoptosis and the deficiency of energy metabolism. Co-incubation of DHB attenuated Aβ25–35-induced pathological changes. The significance of DHB in the management of mitochondria-related disorders is discussed.






Similar content being viewed by others
References
Ball CR (1966) Estimation and identification of thiols in rat spleen after cysteine or glutathione treatment: relevance to protection against nitrogen mustards. Biochem Pharmacol 15:809–816
Beal MF (2000) Energetics in the pathogenesis of neurodegenerative diseases. Trends Neurosci 23:298–304
Berman SB, Watkins SC, Hastings TG (2000) Quantitative biochemical and ultrastructural comparison of mitochondrial permeability transition in isolated brain and liver mitochondria: evidence for reduced sensitivity of brain mitochondria. Exp Neurol 164:415–425
Bernardi P, Petronilli V (1996) The permeability transition pore as a mitochondrial calcium release channel: a critical appraisal. J Bioenerg Biomembr 28:131–138
Berridge MV, Tan AS (1993) Characterization of the cellular reduction of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT): subcellular localization, substrate dependence, and involvement of mitochondrial electron transport in MTT reduction. Arch Biochem Biophys 303:474–482
Bolanos JP, Heales SJ, Land JM, Clark JB (1995) Effect of peroxynitrite on the mitochondrial respiratory chain: differential susceptibility of neurones and astrocytes in primary culture. J Neurochem 64:1965–1972
Casley CS, Canevari L, Land JM, Clark JB, Sharpe MA (2002) Beta-amyloid inhibits integrated mitochondrial respiration and key enzyme activities. J Neurochem 80:91–100
Chandrasekaran K, Hatanpaa K, Brady DR, Rapoport SI (1996) Evidence for physiological down-regulation of brain oxidative phosphorylation in Alzheimer’s disease. Exp Neurol 142:80–88
Cook DG, Foeman MS, Sung C, Leight S, Kolson DL, Iwatsubo T, Lee VM, Doms RW (1997) Alzheimer’s A beta(1–42) is generated in the endoplasmic reticulum/intermediate compartment of NT2N cells. Nat Med 3:1021–1023
Crow JP (1997) Dichlorodihydrofluorescein and dihydrorhodamine 123 are sensitive indicators of peroxynitrite in vitro: implications for intracellular measurement of reactive nitrogen and oxygen species. Nitric Oxide 1:145–157
Elnageh KM, Gaitonde MK (1988) Effect of a deficiency of thiamine on brain pyruvate dehydrogenase: enzyme assay by three different methods. J Neurochem 51:1482–1489
Gabuzda D, Busciglio J, Chen LB, Matsudaira P, Yankner BA (1994) Inhibition of energy metabolism alters the processing of amyloid precursor protein and induces a potentially amyloidogenic derivative. J Biol Chem 269:13623–13628
Gorman AM, Orrenius S, Ceccateli S (1998) Apoptosis in neuronal cells: role of caspases. Neuroreport 9:49–55
Gu QB, Zhao JX, Fei J, Schwarz W (2004) Modulation of Na(+),K(+) pumping and neurotransmitter uptake by beta-amyloid. Neuroscience 126:61–67
Hirai K, Hayako H, Kato K, Miyamoto M (1998) Idebenone protects hippocampal neurons against amyloid beta-peptide-induced neurotoxicity in rat primary cultures. Naunyn Schmiedebergs Arch Pharmacol 358:582–585
Hirai K, Aliev G, Nunomura A, Fujioka H, Russell RL, Atwood CS, Johnson AB, Kress Y, Vinters HV, Tabaton M, Shimohama S, Cash AD, Siedlak SL, Harris PL, Jones PK, Petersen RB, Perry G, Smith MA (2001) Mitochondrial abnormalities in Alzheimer’s disease. J Neurosci 21:3017–3023
Huang SY, Li GQ, Shi JG, Mo SY (2004) Chemical constituents of the rhizomes of Coeloglossum viride var. bracteatum. J Asian Nat Prod Res 6:49–61
Ishida H, Higashijima N, Hirota Y, Genka C, Nakazawa H, Nakaya H, Sato T (2004) Nicorandil attenuates the mitochondrial Ca2+ overload with accompanying depolarization of the mitochondrial membrane in the heart. Naunyn Schmiedebergs Arch Pharmacol 369:192–197
Iversen LL, Mortishire-Smith RJ, Pollack SJ, Shearman MS (1995) The toxicity in vitro of beta-amyloid protein. J Biochem 311:1–16
Jiang S, Cai J, Wallace DC, Jones DP (1999) Cytochrome c-mediated apoptosis in cells lacking mitochondrial DNA. Signaling pathway involving release and caspase 3 activation is conserved. J Biol Chem 274:29905–29911
Jiangsu New Medical College (1977) Dictionary of Traditional Chinese Medicine. Shanghai People’s Publishing House, Shanghai, pp. 436–437
Jurgensmeier JM, Xie Z, Deveraux Q, Ellerby L, Bredesen D, Reed JC (1998) Bax directly induces release of cytochrome c from isolated mitochondria. Proc Natl Acad Sci 95:4997–5002
Kubo T, Nishimura S, Kumagae Y, Kaneko I (2002) In vivo conversion of Racemized-Amyloid((D-Ser26)A beta1–40) to truncated and toxic fragments ([D-Ser 26]A beta25–35/40) and Fragment presence in the brains of Alzheimer’s patients. J Neurosci Res 70:474–483
Lai JC, Clark JB (1979) Preparation of synaptic and nonsynaptic mitochondria from mammalian brain. Meth Enzymol 55:51–60
Lai JC, Cooper AJ (1986) Brain alpha-ketoglutarate dehydrogenase complex: kinetic properties, regional distribution, and effects of inhibitors. J Neurochem 47:1376–1386
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Mark RJ, Lovell MA, Markesbery WR, Uchida K, Mattson MP (1997) A role for 4-hydroxynonenal, an aldehydic product of lipid peroxidation, in disruption of ion homeostasis and neuronal death induced by amyloid beta-peptide. J Neurochem 68:255–264
Mastrogiacoma F, Lindsay JG, Bettendorff L, Rice J, Kish SJ (1996) Brain protein and alpha-ketoglutarate dehydrogenase complex activity in Alzheimer’s disease. Ann Neurol 39:592–598
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Muszbek L, Szabo T, Fesus L (1997) A high sensitive method for the measurement of ATPase activity. Anal Biochem 77:286–288
Myhre O, Vestad TA, Sagstuen E, Aarnes H, Fonnum F (2000) The effects of aliphatic (n-nonane), naphtenic (1,2,4-trimethylcyclohexane), and aromatic (1,2,4-trimethylbenzene) hydrocarbons on respiratory burst in human neutrophil granulocytes. Toxicol Appl Pharmacol 167:222–230
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Palmeira CM, Moreno AJ, Madeira VM, Wallace KB (1996) Continuous monitoring of mitochondrial membrane potential in hepatocyte cell suspensions. J Pharmacol Toxicol Methods 35:35–43
Pereira C, Santos MS, Oliveira C (1999) Involvement of oxidative stress on the impairment of energy metabolism induced by a beta peptides on PC12 cells: protection by antioxidants. Neurobiol Dis 6:209–219
Petronilli V, Costantini P, Scorrano R, Passamonti S, Bernardi P (1994) The voltage sensor of the mitochondrial permeability transition pore is tuned by the oxidation-reduction state of vicinal thiols. J Biol Chem 269:16638–16642
Pike CJ, Burdick D, Walencewicz AJ, Glabe CG, Cotman CW (1993) Neurodegeneration induced by beta-amyloid peptides in vitro: the role of peptide assembly state. J Neurosci 13:1676–1687
Poirier J, Danik M, Blass JP (1999) Pathophysiology of the Alzheimer syndrome. In: Gautier S (ed), Clinical diagnosis and management of Alzheimer’s disease. Martin Dunitz, London, pp.17–32
Prehn JH (1998) Mitochondrial transmembrane potential and free radical production in excitotoxic neurodegeneration. Naunyn Schmiedebergs Arch Pharmacol 357:316–322
Rodrigues CM, Sola S, Silva R, Brites D (2000) Bilirubin and amyloid-beta peptide induce cytochrome c release through mitochondrial membrane permeabilization. Mol Med 6:936–946
Selkoe DJ (1999) Translating cell biology into therapeutic advances in Alzheimer’s disease. Nature 399:23–31
Skulachev VP (1998) Cytochrome c in the apoptotic and antioxidant cascades. FEBS Lett 423:275–280
Sureda FX, Escubedo E, Gabriel C, Camarasa J, Camins A (1996) Effect of glutamate receptor ligands on mitochondrial membrane potential in rat dissociated cerebellar cells. Naunyn Schmiedebergs Arch Pharmacol 354:420–423
Wharton DC, Tzagoloff A (1967) Cytochrome oxidase from beef heart mitochondria. Meth Enzymol 10:245–250
Zhang D, Liu GT, Shi JG, Zhang JJ (2006) Effects of Coeloglossum. viride var. bracteatum extract on memory deficits and pathological changes in senescent mice. Basic Clin Pharmacol Toxicol 98:55–60
Zoratti M, Szabo I (1995) The mitochondrial permeability transition. Biochim Biophys Acta 1241:139–176
Acknowledgments
This work was supported by research grants from the Natural Science Foundation of China (30472179) and the National High-tech R&D Program (863 Program) (2004AA2Z3815). We thank Dr. Xiaoliang Wang for reading the manuscript and providing helpful comments and suggestions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, D., Zhang, Y., Liu, G. et al. Dactylorhin B reduces toxic effects of β-amyloid fragment (25–35) on neuron cells and isolated rat brain mitochondria. Naunyn-Schmied Arch Pharmacol 374, 117–125 (2006). https://doi.org/10.1007/s00210-006-0095-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-006-0095-9