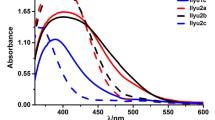
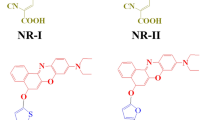
Abstract
This study is carried out to design diarylamine-fluorene dyes by incorporating electron‐deficient thiadiazole derivatives. Quantum chemical calculations are performed to study the geometries, electronic structures and absorption spectra of the dyes. Interaction of dyes with TiO2 cluster is also studied. The effects of the electron‐deficient units on the spectra and electrochemical properties have been investigated. Dyes D1–D4 display remarkably enhanced spectral response in the red portion of the solar spectrum as compared with reference compound D0. The newly designed dyes demonstrate desirable energetic and spectroscopic parameters and may lead to efficient metal‐free organic dye sensitizers for DSSCs.




Similar content being viewed by others
References
O’Regan B, Gratzel M (1991) Nature 353:737
Hagfeldt A, Boschloo G, Sun L, Kloo L, Pettersson H (2010) Chem Rev 110:6595
Clifford JN, Martinez-Ferrero E, Viterisi A, Palomares E (2011) Chem Soc Rev 40:1635
Abdullah MI, Janjua MRSA, Mahmood A, Ali S, Ali M (2013) Bull Korean Chem Soc 34:2093–2098
Abdullah MI, Janjua MRSA, Nazar MF, Mahmood A (2013) Bull Chem Soc Jpn 86:1272–1281
Hagfeldt A, Ute B Cappel, Boschloo G, Sun L, Kloo L, Pettersson H, Gibson EA (2013) Solar Cells
Nazeeruddin MK, Angelis FD, Fantacci S, Selloni A, Viscardi G, Liska P, Ito S, Takeru B, Gratzel M (2005) J Am Chem Soc 127:16835
Funaki T, Funakoshi H, Kitao O, Onozawa-Komatsuzaki N, Kasuga K, Sayama K, Sugihara H (2012) Angew Chem Int Ed 51:7528
Mishra A, Fischer MKR, Bauerle P (2009) Angew Chem Int Ed 48:2474
Liang M, Chen J (2013) Chem Soc Rev 42:3453
Tian H, Yang X, Chen R, Zhang R, Hagfeldt A, Sun L (2008) J Phys Chem C 112:11023–11033
Cai N, Moon S-J, Cevey-Ha L, Moehl T, Humphry-Baker R, Wang P, Zakeeruddin SM, Grätzel M (2011) Nano Lett 11:1452–1456
Zhu W, Wu Y, Wang S, Li W, Li X, Chen J, Wang Z-S, Tian H (2011) Adv Funct Mater 21:756–763
Chen K-F, Chang C-W, Lin J-L, Hsu Y-C, Yeh M-CP, Hsu C-P, Sun S-S (2010) Chem Eur J 16:12873–12882
Ning Z, Zhang Q, Wu W, Pei H, Liu B, Tian H (2008) J Org Chem 73:3791–3797
Qin P, Yang X, Chen R, Sun L, Marinado T, Edvinsson T, Boschloo G, Hagfeldt A (2007) J Phys Chem C 111:1853–1860
Hagberg DP, Marinado T, Karlsson KM, Nonomura K, Qin P, Boschloo G, Brinck T, Hagfeldt A, Sun L (2007) J Org Chem 72:9550–9556
Wu W, Yang J, Hua J, Tang J, Zhang L, Long Y, Tian H (2010) J Mater Chem 20:1772–1779
Chen C-H, Hsu Y-C, Chou H-H, Thomas KRJ, Lin JT, Hsu C-P (2010) Chem Eur J 16:3184–3193
Zhang J, Li H-B, Sun S-L, Geng Y, Wu Y, Su Z-M (2012) J Mater Chem 22:568–576
Zeng WD, Cao YM, Bai Y, Wang YH, Shi YS, Zhang M, Wang FF, Pan CY, Wang P (2010) Chem Mater 22:1915–1925
Tian HN, Yang XC, Chen RK, Zhang R, Hagfeldt A, Sun LC (2008) J Phys Chem C 112:11023–11033
Choi H, Choi H, Paek S, Song K, Kang M-S, Ko J (2010) Bull Korean Chem Soc 31:125–132
Li Y-T, Chen C-L, Hsu Y-Y, Hsu H-C, Chi Y, Chen B-S, Liu W-H, Lai C-H, Lin T-Y, Chou P-T (2010) Tetrahedron 66:4223–4229
Chen C-H, Hsu Y-C, Chou H-H, Thomas KRJ, Lin JT, Hsu C-P (2010) Chem Eur J 16:3184–3193 168
Lin LY, Tsai CH, Wong KT, Huang TW, Wu CC, Chou SH, Lin F, Chen SH, Tsai AI (2011) J Mater Chem 21:5950–5958
Lee DH, Lee MJ, Song HM, Song BJ, Seo KD, Pastore M, Anselmi C, Fantacci S, De Angelis F, Nazeeruddin MK, Grätzel M, Kim HK (2011) Dyes Pigm 91:192–198
Thomas KRJ, Baheti A, Hsu Y-C, Ho K-C, Lin JT (2011) Dyes Pigm 91:33–43
Velusamy M, Thomas KRJ, Lin JT, Hsu YC, Ho KC (2005) Org Lett 7:1899–1902
Xu MF, Li RZ, Pootrakulchote N, Shi D, Guo J, Yi ZH, Zakeeruddin SM, Grätzel M, Wang P (2008) J Phys Chem C 112:19770–19776
Lin JT, Chen P-C, Yen Y-S, Hsu Y-C, Chou H-H, Yeh M-CP (2009) Org Lett 11:97–100
Yen Y-S, Hsu Y-C, Lin JT, Chang C-W, Hsu C-P, Yin D-J (2008) J Phys Chem C 112:12557–12567
Zhou H, Yang L, Stoneking S, You W (2010) ACS Appl Mater Interfaces 2:1377–1383
Zhou H, Yang L, Price SC, Knight KJ, You W (2010) Angew Chem Int Ed 49:7992–7995
Zhou H, Yang L, Liu S, You W (2010) Macromolecules 43:10390–10396
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA, Peralta JE Jr, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski J, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) GAUSSIAN 09 (Revision A.2), Gaussian, Inc., Wallingford, CT
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Becke AD (1993) J Chem Phys 98:5648–5652
Gordon MS (1980) Chem Phys Lett 76:163–168
Hay PJ, Wadt WR (1985) J Chem Phys 82:270–283
Chen J, Bai F-Q, Wang J, Hao L, Xie Z-F, Pan Q-J, Zhang H-X (2012) Dyes Pigm 94:459–468
Yanai T, Tew DP, Handy NC (2004) Chem Phys Lett 393:51–57
Zhang J, Li H-B, Wu Y, Geng Y, Duan Y-A, Liao Y, SU Z-M (2011) Chem. J. Chinese U. 32:1343–1348
Barone V, Cossi M (1998) J Phys Chem A 102:1995–2001
Qin P, Yang XC, Chen RK, Sun LC, Marinado T, Edvinsson T, Boschloo G, Hagfeldt A (2007) J Phys Chem C 111:1853–1860
Gratzel M (2001) Nature 414:338–344
Zhang GL, Bai Y, Li RZ, Shi D, Wenger S, Zakeeruddin SM, Gratzel M, Wang P (2009) Energy Environ Sci 2:92–95
Nalwa HS (2001) Handbook of advanced electronic and photonic materials and devices. Academic, San Diego, CA
Sanchez-de-Armas R, San Miguel MA, Oviedo J, Sanz JF (2012) Phys Chem Chem Phys 14:225–233
Zhang J, Kan Y-H, Li H-B, Geng Y, Wu Y, Su Z-M (2012) Dyes Pigm 95:313–321
Katoh R, Furube A, Yoshihara T, Hara K, Fujihashi G, Takano S, Murata S, Arakawa H, Tachiya M (2004) J Phys Chem B 108:4818–4822
Preat J, Michaux C, Jacquemin D, Perpete EA (2009) J Phys Chem C 113:16821–16833
Pearson RG (1988) Inorg Chem 27:734
Acknowledgments
The authors would like to sincerely appreciate the Deanship of Scientific Research at King Saud University for its funding of this research through the Research Group Project no RGP-VPP-255.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khan, S.UD., Mahmood, A., Rana, U.A. et al. Utilization of electron‐deficient thiadiazole derivatives as π-spacer for the red shifting of absorption maxima of diarylamine-fluorene based dyes. Theor Chem Acc 134, 1596 (2015). https://doi.org/10.1007/s00214-014-1596-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-014-1596-0