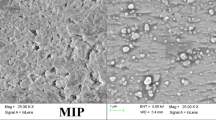
Abstract
An As(III)-imprinted chitosan resin (As-ICR) was successfully prepared to selectively remove arsenic from Laminaria japonica Aresch (LJA) juice. Batch adsorption experiments and column adsorption experiments were performed to evaluate the adsorption conditions, selectivity, and reusability. The results showed that the minimum total arsenic concentration of the juice was 4.16 mg/L at pH 6.0, 40 °C, with equilibrium time 4 h. The selectivity of As-ICR for arsenic was much higher than that of non-imprinted chitosan resin. As-ICR could be reused for 10 times with only about 13% regeneration loss. FTIR spectra demonstrated that the adsorption of As-ICR for arsenic occurred on amino and hydroxyl groups. Total arsenic concentration of the juice decreased significantly, while the other metal ions decreased except Zn2+ insignificantly. This suggests that As-ICR is a very promising sorbent for selective removal of arsenic from the juice.






Similar content being viewed by others
References
Fendorf S, Michael HA, Geen AV (2010) Spatial and temporal variations of groundwater arsenic in south and southeast asia. Science 328:1123–1127
Tapio S, Grosche B (2006) Arsenic in the aetiology of cancer. Mutat Res 612:215–246
Rahmana MA, Hasegawa H, Rahman MM, Miah MAM, Tasmind A (2008) Arsenic accumulation in rice (Oryza sativa L.): human exposure through food chain. Ecotox Environ Safe 69:317–324
Hassan KM, Fukuhara T, Hai FI, Bari QH, KhMdS Islam (2009) Development of a bio-physicochemical technique for arsenic removal from groundwater. Desalination 249:224–229
Zhao Y, Zouboulis AI, Matis KA (1996) Removal of molybdate and arsenate from aqueous solutions by flotation. Sep Sci Technol 31:769–785
Bhattacharya D, AB Jumawan, Grieves RB (1979) Separation of toxic heavy metals by sulfide precipitation. Sep Sci Technol 14:441–452
Baskan MB, Pala A (2010) A statistical experiment design approach for arsenic removal by coagulation process using aluminum sulfate. Desalination 254:42–48
Balarama Krishna MV, Chandrasekaran K, Karunasagar D, Arunachalam J (2001) A combined treatment approach using Fenton’s reagent and zero valent iron for the removal of arsenic from drinking water. J Hazard Mater 84:229–240
Kim J, Benjamin MM (2004) Modeling a novel ion exchange process for arsenic and nitrate removal. Water Res 38(8):2053–2062
Guan BH, Ni WM, Wu ZB, Lai Y (2009) Removal of Mn(II) and Zn(II) ions from flue gas desulfurization wastewater with water-soluble chitosan. Sep Purif Technol 65:269–274
Huang LL, Sui WP, Wang YX, Jiao Q (2010) Preparation of chitosan/chondroitin sulfate complex microcapsules and application in controlled release of 5-fluorouracil. Carbohyd Polym 80(1):168–173
Volda IMN, Varum KM, Guibal E, Smidsrod O (2003) Binding of ions to chitosan selectivity studies. Carbohyd Polym 54:471–477
Copello GJ, Varela F, Martínez VR, Díaz LE (2008) Immobilized chitosan as biosorbent for the removal of Cd (II), Cr(III) and Cr(VI) from aqueous solutions. Bioresour Technol 99:6538–6544
Vijaya Y, Popuri SR, Boddu VM, Krishnaiah A (2008) Modified chitosan and calcium alginate biopolymer sorbents for removal of nickel(II) through adsorption. Carbohyd Polym 72:261–271
Sun SL, Wang AQ (2006) Adsorption properties of N–succinyl–chitosan and cross–linked N–succinyl–chitosan resin with Pb(II) as template ions. Sep Purif Technol 51:409–415
Krishnapriya KR, Kandaswamy M (2009) Synthesis and characterization of a crosslinked chitosan derivative with a complexing agent and its adsorption studies toward metal (II) ions. Carbohyd Res 344(13):1632–1638
Guibal E (2004) Interactions of metal ions with chitosan–based sorbents: a review. Sep Purif Technol 38:43–74
Ramesh A, Hasegawa H, Sugimoto W, Maki T, Ueda K (2008) Adsorption of gold(III), platinum(IV) and palladium(II) onto glycine modified crosslinked chitosan resin. Bioresour Technol 99:3801–3809
Zhou LM, Wang YP, Liu ZR, Huang QW (2009) Characteristics of equilibrium, kinetics studies for adsorption of Hg(II), Cu(II), and Ni(II) ions by thiourea–modified magnetic chitosan microspheres. J Hazard Mater 161:995–1002
Juang RS, Shao HJ (2002) A simplified equilibrium model for sorption of heavy metal ions from aqueous solutions on chitosan. Water Res 36:2999–3008
Claude B, Viron LC, Haupt K, Morin P (2010) Synthesis of a molecularly imprinted polymer for the solid-phase extraction of betulin and betulinic acid from plane bark. Phytochem Anal 21:180–185
Ahmadi SJ, Noori KO, Shirvani AS (2010) Synthesis and characterization of new ion-imprinted polymer for separation and preconcentration of uranyl (UO2 2+) ions. J Hazard Mater 175:193–197
Biju VM, Mary GJ, Prasada RT (2003) Ion imprinted polymer particles: synthesis, characterization and dysprosium ion uptake properties suitable for analytical applications. Anal Chim Acta 478:43–51
Garcia R, Pinel C, Madic C, Lemaire M (1998) Ionic imprinting effect in gadolinium/lanthanum separation. Tetrahedron Lett 39:8651–8654
Metilda P, Prasad K, Kala R, Gladis JM, Prasada RT, Naidu GRK (2007) Ion imprinted polymer based sensor for monitoring toxic uranium in environmental samples. Anal Chim Acta 582:147–153
Daniel S, Mary GJ, Prasada RT (2003) Synthesis of imprinted polymer material with palladium ion nanopores and its analytical application. Anal Chim Acta 488:173–182
Say R, Birlik E, Ersöz A, Yılmaz F, Gedikbey T, Denizli A (2003) Preconcentration of copper on ion-selective imprinted polymer microbeads. Anal Chim Acta 480:251–258
Chen H, Olmstead MM, Albright RL, Devenyi J, Fish RH (1997) Metal–ion–templated polymers: synthesis and structure of N–(4–Vinylbenzyl)–1,4,7–Triazacyclononanezinc(II) complexes, their copolymerization with divinylbenzene, and metal–ion selectivity studies of the demetalated resins–evidence for a sandwich complex in the polymer matrix. Angew Chem Int Edit Engl 36:642–645
Yoshida M, Uezu K, Goto M, Furusaki S (1999) Required properties for functional monomers to produce a metal template effect by a surface molecular imprinting technique. Macromolecules 32:1237–1243
Wulff G (1995) Molecular imprinting in cross–linked materials with the aid of molecular templates—a way towards artificial antibodies. Angew Chem Int Edit Engl 34:1812–1832
Ersöz A, Say R, Denizli A (2004) Ni(II) ion–imprinted solid-phase extraction and preconcentration in aqueous solutions by packed-bed columns. Anal Chim Acta 502:91–97
Rosatzin T, Andersson LI, Simon W, Mosbach K (1991) Preparation of Ca2+ selective sorbents by molecular imprinting using polymerisable ionophores. J Chem Soc Perk Trans 2 28:1261–1265
Dhal PK, Arnold FH (1991) Template-mediated synthesis of metal–complexing polymers for molecular recognition. J Am Chem Soc 113:7417–7418
Wang DF, Liu BJ (2010) A preparation technology of resin for removing residual heavy metal arsenic from LJA juice. CN patent No.201010119859.6
Sun SL, Wang AQ (2006) Adsorption properties of carboxymethyl–chitosan and cross–linked carboxymethyl-chitosan resin with Cu(II) as template ions. Sep Purif Technol 49:197–204
Reed BE, Vaughan R, Jiang L (2000) As(III), As(V), Hg and Pb removal by Fe–oxide impregnated activated carbon. J Environ Eng 126:869–873
Chen CY, Chang TH, Kuo JT, Chen YF, Chung YC (2008) Characteristics of molybdate–impregnated chitosan beads (MICB) in terms of arsenic removal from water and the application of a MICB–packed column to remove arsenic from wastewater. Bioresour Technol 99:7487–7494
Bhattacharyya KG, Sarma A (2003) Adsorption characteristics of the dye, brilliant green, on neem leaf powder. Dye Pigment 57:211–222
Jin L, Bai RB (2002) Mechanisms of lead adsorption on chitosan/PVA hydrogel beads. Langmuir 18:9765–9770
Acknowledgments
This work was financially supported by the Ocean Public Welfare Scientific Research Special Appropriation Project (No. 201005020), National Natural Science Foundation projects of China (No. 20807040 and No. 30972289) and the international cooperation project (No. 2010DFA31330).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, B., Wang, D., Gao, X. et al. Removal of arsenic from Laminaria japonica Aresch juice using As(III)-imprinted chitosan resin. Eur Food Res Technol 232, 911–917 (2011). https://doi.org/10.1007/s00217-011-1460-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-011-1460-6