Abstract
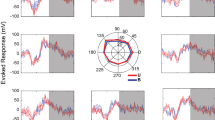
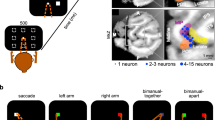
The motor output of the pontomedullary reticular formation (PMRF) was investigated to determine the reticulospinal system’s capacity for bilateral control of the upper limbs. Stimulus triggered electromyographic averages (StimulusTA) were constructed from muscles of both upper limbs while two awake monkeys (Macaca fascicularis) performed a reaching task using either arm. Extensor and flexor muscles were studied at the wrist, elbow, and shoulder; muscles acting on the scapula were also studied. Post-stimulus effects (PStEs) resulted from 435 (81%) of 535 sites tested. Of 1611 PStEs analyzed, 58% were post-stimulus suppression (PStS), and 42% were post-stimulus facilitation (PStF). Onset latency was earlier for PStF than PStS, duration was longer for PStS, and amplitude was larger for PStF. Ipsilateral and contralateral PStEs were equally prevalent; bilateral responses were typical. In the ipsilateral forelimb and shoulder, the prevalent pattern was flexor PStF and extensor PStS; the opposite pattern was prevalent contralaterally. Sites producing strong ipsilateral upper trapezius PStF were concentrated in a region caudal and ventral to abducens. The majority of muscles studied had no clear somatotopic organization. Overall, the results indicate the monkey PMRF has the capacity to support bilateral coordination of limb movements using reciprocal actions within a limb and between sides.








Similar content being viewed by others
References
Alstermark B, Eide E, Gorska T, Lundberg A, Pettersson LG (1984) Visually guided switching of forelimb target reaching in cats. Acta Physiol Scand 120:151–153
Alstermark B, Isa T, Lundberg A, Pettersson LG, Tantisira B (1993a) Characteristics of target-reaching in cats. II. Reaching to targets at different locations. Exp Brain Res 94:287–294
Alstermark B, Lundberg A, Pettersson LG, Tantisira B, Walkowska M (1993b) Characteristics of target-reaching in cats. I. Individual differences and intra-individual constancy. Exp Brain Res 94:279–286
Bannatyne BA, Edgley SA, Hammar I, Jankowska E, Maxwell DJ (2003) Networks of inhibitory and excitatory commissural interneurons mediating crossed reticulospinal actions. Eur J Neurosci 18:2273–2284
Belhaj-Saif A, Karrer JH, Cheney PD (1998) Distribution and characteristics of poststimulus effects in proximal and distal forelimb muscles from red nucleus in the monkey. J Neurophysiol 79:1777–1789
Bowden D, Dubach M, MacArthur E, Song E (2003) BrainInfo (2000), Neuroscience division, regional primate research center, University of Washington, accessed 31-Dec-2003
Buford JA, Davidson AG (2004) Movement-related and preparatory activity in the reticulospinal system of the monkey. Exp Brain Res DOI 10.1007/s00221-004-1956-4:
Canedo A (1997) Primary motor cortex influences on the descending and ascending systems. Prog Neurobiol 51:287–335
Cheney PD, Fetz EE (1985) Comparable patterns of muscle facilitation evoked by individual corticomotoneuronal (CM) cells and by single intracortical microstimuli in primates: evidence for functional groups of CM cells. J Neurophysiol 53:786–804
Cheney PD, Fetz EE, Palmer SS (1985) Patterns of facilitation and suppression of antagonist forelimb muscles from motor cortex sites in the awake monkey. J Neurophysiol 53:805–820
Cheney PD, Mewes K, Widener G (1991) Effects on wrist and digit muscle activity from microstimuli applied at the sites of rubromotoneuronal cells in primates. J Neurophysiol 66:1978–1992
Cowie RJ, Robinson DL (1994) Subcortical contributions to head movements in macaques. I. Contrasting effects of electrical stimulation of a medial pontomedullary region and the superior colliculus. J Neurophysiol 72:2648–2664
Cowie RJ, Smith MK, Robinson DL (1994) Subcortical contributions to head movements in macaques. II. Connections of a medial pontomedullary head-movement region. J Neurophysiol 72:2665–2682
Davidson AG (2004) Bilateral actions of the reticulospinal tract in the monkey. Dissertation, The Ohio State University, Columbus, OH, USA
Davidson AG, Buford JA (2004a) Motor outputs from the primate reticular formation to shoulder muscles as revealed by stimulus triggered averaging. J Neurophysiol 92:83–95
Davidson AG, Buford JA (2004b) Spike and stimulus triggered averaging of arm and shoulder muscles bilaterally from the reticular formation in the monkey. Program No.755.14.2004. Abstract viewer/itinerary planner. Society for Neuroscience, Washington, DC (Online)
Denny-Brown D (1966) The cerebral control of movement. Charles C Thomas, Springfield, IL
Dewald JP, Pope PS, Given JD, Buchanan TS, Rymer WZ (1995) Abnormal muscle coactivation patterns during isometric torque generation at the elbow and shoulder in hemiparetic subjects. Brain 118(Pt 2):495–510
Drew T (1991) Functional organization within the medullary reticular formation of the intact unanesthetized cat. III. Microstimulation during locomotion. J Neurophysiol 66:919–938
Drew T, Prentice S, Schepens B (2004) Cortical and brainstem control of locomotion. Prog Brain Res 143:251–261
Drew T, Rossignol S (1984) Phase-dependent responses evoked in limb muscles by stimulation of medullary reticular formation during locomotion in thalamic cats. J Neurophysiol 52:653–675
Drew T, Rossignol S (1990a) Functional organization within the medullary reticular formation of intact unanesthetized cat. I. Movements evoked by microstimulation. J Neurophysiol 64:767–781
Drew T, Rossignol S (1990b) Functional organization within the medullary reticular formation of intact unanesthetized cat. II. Electromyographic activity evoked by microstimulation. J Neurophysiol 64:782–795
Fetz EE, Cheney PD (1980) Postspike facilitation of forelimb muscle activity by primate corticomotoneuronal cells. J Neurophysiol 44:751–772
Freund HJ, Hummelsheim H (1985) Lesions of premotor cortex in man. Brain 108(Pt 3):697–733
Gibson AR, Horn KM, Pong M, Van Kan PL (1998) Construction of a reach-to-grasp. Novartis Found Symp 218:233–245
Gossard JP, Floeter MK, Degtyarenko AM, Simon ES, Burke RE (1996) Disynaptic vestibulospinal and reticulospinal excitation in cat lumbosacral motoneurons: modulation during fictive locomotion. Exp Brain Res 109:277–288
Jankowska E, Hammar I, Slawinska U, Maleszak K, Edgley SA (2003) Neuronal basis of crossed actions from the reticular formation on feline hindlimb motoneurons. J Neurosci 23:1867–1878
Kably B, Drew T (1998) Corticoreticular pathways in the cat. I. Projection patterns and collaterization. J Neurophysiol 80:389–405
Krutki P, Jankowska E, Edgley SA (2003) Are crossed actions of reticulospinal and vestibulospinal neurons on feline motoneurons mediated by the same or separate commissural neurons? J Neurosci 23:8041–8050
Kuypers HG (1958) Anatomical analysis of cortico-bulbar connexions to the pons and the lower brain stem in the cat. J Anat 92:198–218
Kuypers HG (1981) Anatomy of descending pathways. Handbook of physiology. Sect. I. The nervous system, vol. II. Motor control, pt 1, vol II. Motor control, pt 1. American Physiological Society, Bethesda, MD, pp 597–666
Lemon RN, Mantel GW, Muir RB (1986) Corticospinal facilitation of hand muscles during voluntary movement in the conscious monkey. J Physiol 381:497–527
Martin RF, Bowden DM (1996) A stereotaxic template atlas of the macaque brain for digital imaging and quantitative neuroanatomy. Neuroimage 4:119–150
Matsuyama K, Mori F, Nakajima K, Drew T, Aoki M, Mori S (2004) Locomotor role of the corticoreticular-reticulospinal-spinal interneuronal system. Prog Brain Res 143:239–249
McKiernan BJ, Marcario JK, Karrer JH, Cheney PD (1998) Corticomotoneuronal postspike effects in shoulder, elbow, wrist, digit, and intrinsic hand muscles during a reach and prehension task. J Neurophysiol 80:1961–1980
Nyberg-Hansen R (1965) Sites and mode of termination of reticulo-spinal fibers in the cat. An experimental study with silver impregnation methods. J Comp Neurol 124:71–99
Park MC, Belhaj-Saif A, Gordon M, Cheney PD (2001) Consistent features in the forelimb representation of primary motor cortex in rhesus macaques. J Neurosci 21:2784–2792
Perreault MC, Rossignol S, Drew T (1994) Microstimulation of the medullary reticular formation during fictive locomotion. J Neurophysiol 71:229–245
Peterson BW (1979) Reticulospinal projections to spinal motor nuclei. Ann Rev Physiol 41:127–140
Peterson BW, Maunz RA, Pitts NG, Mackel RG (1975) Patterns of projection and branching of reticulospinal neurons. Exp Brain Res 23:333–351
Peterson BW, Pitts NG, Fukushima K (1979) Reticulospinal connections with limb and axial motoneurons. Exp Brain Res 36:1–20
Petras JM (1967) Cortical, tectal and tegmental fiber connections in the spinal cord of the cat. Brain Res 6:275–324
Quessy S, Freedman EG (2004) Electrical stimulation of rhesus monkey nucleus reticularis gigantocellularis: I. Characteristics of evoked head movements. Exp Brain Res 156:342–356
Schepens B, Drew T (2004) Independent and convergent signals from the pontomedullary reticular formation contribute to the control of posture and movement during reaching in the cat. J Neurophysiol 92:2217–2238
Schwerin SC, Dewald JPA (2004) Ipsilateral hemisphere control of upper extremity muscles following stroke using transcranial magnetic stimulation. Program number 878.4.2004. Abstract viewer/itinerary planner, accessed 27-Oct-2004
Shapovalov AI (1972) Extrapyramidal monosynaptic and disynaptic control of mammalian alpha-motoneurons. Brain Res 40:105–115
Shapovalov AI (1973) Extrapyramidal control of primate motoneurons. In: Desmedt JE (ed) Human reflexes, pathophysiology of motor systems, methodology of human reflexes, vol 3. Karger, Basel, pp145–158
Shinoda Y, Ohgaki T, Sugiuchi Y, Futami T (1992) Morphology of single medial vestibulospinal tract axons in the upper cervical spinal cord of the cat. J Comp Neurol 316:151–172
Shinoda Y, Sugiuchi Y, Izawa Y, Hata Y (2005) Long descending motor tract axons and their control of neck and axial muscles. Prog Brain Res 151:527–563
Sprague JM, Chambers WW (1954) Control of posture by reticular formation and cerebellum in the intact, anesthetized and unanesthetized and in the decerebrated cat. Am J Physiol 176:52–64
Sugiuchi Y, Kakei S, Izawa Y, Shinoda Y (2004) Functional synergies among neck muscles revealed by branching patterns of single long descending motor-tract axons. Prog Brain Res 143:411–421
Szabo J, Cowan WM (1984) A stereotaxic atlas of the brain of the cynomolgus monkey (Macaca fascicularis). J Comp Neurol 222:265–300
Torvik A, Brodal A (1957) The origin of reticulospinal fibers in the cat; an experimental study. Anat Rec 128:113–137
Ackowledgments
We thank Stephanie Moran for excellent technical assistance in all aspects of the project and Neena Joseph for assistance in developing software for control of the task. Thanks also to Wendy Herbert, Melanie McClain, Kristen Keen, Cheryl Fortney, and Chris Figgins for helpful comments on a draft of the manuscript. This work was supported in part by NIH NINDS R01 NS037822 to JAB.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Davidson, A.G., Buford, J.A. Bilateral actions of the reticulospinal tract on arm and shoulder muscles in the monkey: stimulus triggered averaging. Exp Brain Res 173, 25–39 (2006). https://doi.org/10.1007/s00221-006-0374-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-006-0374-1