Abstract
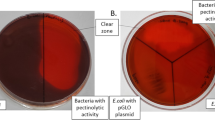
Novel thermophilic and alkaliphilic bacteria for the processing of bast fibres were isolated using hemp pectin as substrate. The strain PB94A, which showed the highest growth rate (µ = 0.5/h) was identified as Geobacillus thermoglucosidasius (DSM 21625). The strain grew optimally at 60°C and pH 8.5. During growth on citrus pectin, the strain produced pectinolytic lyases, which were excreted into the medium. In contrast to the commercially available pectinase Bioprep 3000 L, the enzymes from G. thermoglucosidasius PB94A converted pectin isolated from hemp fibres. In addition to hemp pectin, the culture supernatant also degraded citrus, sugar beet and apple pectin and polygalacturonic acid. When hemp fibres were incubated with the cell-free fermentation broth of G. thermoglucosidasius PB94A, the fineness of the fibres increased. The strain did not produce any cellulases, which is important in order to avoid damaging the fibres during incubation. Therefore, these bacteria or their enzymes can be used to produce fine high-quality hemp fibres.





Similar content being viewed by others
References
Akin DE, Slomczynski D, Rigsby LL, Eriksson KEL (2002) Retting flax with endopolygalacturonase from Rhizopus oryzae. Text Res J 72(1):27–34
Atlas (2004) Handbook of microbiological media. CRC, Boca Raton
Blumenkrantz N, Asboe-Hansen G (1973) New method for quantitative determination of uronic acids. Anal Biochem 54(2):484–489
Brühlmann F, Kim KS, Zimmerman W, Fiechter A (1994) Pectinolytic enzymes from actinomycetes for the degumming of ramie bast fibers. Appl Environ Microb 60(6):2107–2112
Cao J, Zheng L, Chen S (1992) Screening of pectinase producer from alkalophilic bacteria and study on its potential application in degumming of ramie. Enzyme Microb Technol 14:1013–1016
Collmer A, Ried JL, Mount MS (1988) Assay methods for pectic enzymes. In: Wood WA, Kellogg ST (eds) Methods in enzymology, vol 161. Academic, New York, pp 329–335
Cronier D, Monties B, Chabbert B (2005) Structure and chemical composition of bast fibers isolated from developing hemp stem. J Agric Food Chem 53:8279–8289
Dreyer J, Müssig J, Koschke N, Ibenthal W-D, Harig H (2002) Comparison of enzymatically separated hemp and nettle fibre to chemically separated and steam exploded hemp fibre. J Industrial Hemp 7(1):43–59
Foulk JA, Akin DE, Dodd RB (2001) Processing techniques for improving enzyme-retting of flax. Ind Crop Prod 13:239–248
Kashyap DR, Vohra PK, Chopra S, Tewari R (2001) Applications of pectinases in the commercial sector: a review. Bioresource Technol 77:215–227
Kessler RW, Becker U, Kohler R, Goth B (1998) Steam explosion of flax—a superior technique for upgrading fibre value. Biomass Bioenerg 14(3):237–249
Kluskens LD, van Alebeek GJWM, Voragen AGJ, de Vos WM, van der Oost J (2003) Molecular and biochemical characterization of the thermoactive family 1 pectate lyase from the hyperthermophilic bacterium Thermotoga maritima. Biochem J 370:651–659
Kobayashi T, Hatada Y, Higaki N, Lusterio DD, Ozawa T, Koike K, Kawai S, Ito S (1999) Enzymatic properties and deduced amino acid sequence of a high-alkaline pectate lyase from an alkaliphilic Bacillus isolate. Biochim Biophys Acta 1427:145–154
McGovern JN (1983) Fibres (vegetable). In: Grayson M (ed) Encyclopedia of composite materials and components. Wiley-Interscience, New York, pp 497–512
Nazina TN, Tourova TP, Poltaraus AB, Novikova EV, Grigoryan AA, Ivanova AE, Lysenko AM, Petrunyaka VV, Osipov GA, Belyaev SS, Ivanov MV (2001) Taxonomic study of aerobic thermophilic bacilli: descriptions of Geobacillus subterraneus gen. nov., sp. nov. and Geobacillus uzenensis sp. nov. from petroleum reservoirs and transfer of Bacillus stearothermophilus, Bacillus thermocatenulatus, Bacillus thermoleovorans, Bacillus kaustophilus, Bacillus thermoglucosidasius und Bacillus thermodenitrificans to Geobacillus as the new combinations G. stearothermophilus, G. thermocatenulatus, G. thermoleovorans, G. kaustophilus, G. thermoglucosidasius and G. thermodenitrificans. Int J Syst Evol Micr 51:433–446
Ogawa A, Sawada K, Saito K, Hakamada Y, Sumitomo N, Hatada Y, Kobayashi T, Ito S (2000) A new high-alkaline and high-molecular-weight pectate lyase from a Bacillus isolate: enzymatic properties and cloning of the gene for the enzyme. Biosci Biotechnol Biochem 64(6):1133–1141
Sanio C (1995) Zur Mikrobiologie der Flachsröste: bakteriologische, biochemische und technologische Untersuchungen. Dissertation Westfälische Wilhelms-Universität Münster, Germany
Schols HA, Voragen AGJ (2003) Pectic polysaccharides. In: Whitaker JR, Voragen AGJ, Wong DWS (eds) Handbook of food enzymology. Marcel Dekker, New York, pp 829–843
Schols HA, Geraeds CCJM, Searle-van Leeuwen MF, Kormelink FJM, Voragen AGJ (1990) Rhamnogalacturonase: a novel enzyme that degrades the hairy regions of pectins. Carbohyd Res 206:105–115
Sharma HSS, Lefevre J, Boucaud J (1992) Role of microbial enzymes during retting and their effect on fibre characteristics. In: Sharma HSS, van Sumere CF (eds) The biology and processing of flax. M Publications, Belfast, pp 199–212
Sharma HSS, Whiteside L, Kernaghan K (2005) Enzymatic treatment of flax fibre at the roving stage for production of wet-spun yarn. Enzyme Microb Technol 37:386–394
Soares MMCN, da Silva R, Gomes E (1999) Screening of bacterial strains for pectinolytic activity: characterization of the polygalacturonase produced by Bacillus sp. Rev Microb 30:299–303
Takao M, Nakaniwa T, Yoshikawa K, Terashita T, Sakai T (2000) Purification and characterization of thermostable pectate lyase with protopectinase activity from thermophilic Bacillus sp. TS 47. Biosci Biotechnol Biochem 64(11):2360–2367
Teather RM, Wood PJ (1982) Use of Congo red-polysaccharide interactions in enumeration and characterization of cellulolytic bacteria from the bovine rumen. Appl Environ Microb 43(4):777–780
Van Alebeek G-JWM, Christensen TMIE, Schols HA, Mikkelsen JD, Voragen AGJ (2002) Mode of action of pectin lyase A of Aspergillus niger on differently C6-substituted oligogalacturonides. J Biol Chem 227(29):25929–25936
Van Sumere CF (1992) Retting of flax with special reference to enzymatic retting. In: Sharma HSS, van Sumere CF (eds) The biology and processing of flax. M Publications, Belfast, pp 157–198
Vignon MR, Garcia-Jaldon C (1996) Structural features of the pectic polysaccharides isolated from retted hemp bast fibres. Carbohyd Res 296:249–260
Von Drach V, Kloß KD, Hensel K-H (1999) Ultraschallbehandlung von Pflanzenfasern—Herstellung und Einsatz. In: 2nd International Wood and Natural Fibre Composites Symposium, Kassel, vol. 23, pp 1–28
Yavuz E, Gunes H, Harsa S, Yenidunya AF (2004) Identification of extracellular enzyme producing thermophilic bacilli from Balcova (Agamemnon) geothermal site by ITS rDNA RFLP. J Appl Microbiol 97:810–817
Zhai C, Cao J, Wang Y (2003) Cloning and expression of a pectate lyase gene from Bacillus alcalophilus NTT33. Enzyme Microb Technol 33:173–178
Zhang J, Henriksson H, Szabo IJ, Henriksson G, Johansson G (2005) The active component in the flax-retting system of the zygomycete Rhizopus oryzae sb is a family 28 polygalacturonase. J Ind Microbiol Biotechnol 32:431–438
Acknowledgements
This study was supported by the Bundesministerium für Bildung und Forschung with the project 0312640A and by the Fachagentur Nachwachsende Rohstoffe with the project 22006303. We thank Dr. rer. nat. Holger Fischer and Dr. Jörg Müssig from Faserinstitut Bremen (FIBRE) for the analysis of the fibre fineness.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Valladares Juárez, A.G., Dreyer, J., Göpel, P.K. et al. Characterisation of a new thermoalkaliphilic bacterium for the production of high-quality hemp fibres, Geobacillus thermoglucosidasius strain PB94A. Appl Microbiol Biotechnol 83, 521–527 (2009). https://doi.org/10.1007/s00253-009-1969-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-009-1969-9