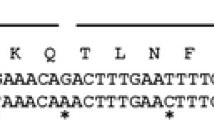
Abstract
Easy and low-cost protein purification methods for the mass production of commonly used enzymes that play important roles in biotechnology are in high demand. In this study, we developed a fast, low-cost recombinant protein purification system in the methylotrophic yeast Pichia pastoris using the family 3 cellulose-binding module (CBM3)-based affinity tag. The codon of the cbm3 gene from Clostridium thermocellum was optimized based on the codon usage of P. pastoris. The CBM3 tag was then fused with enhanced green fluorescent protein (CBM3-EGFP) or with inulinase and expressed in P. pastoris to demonstrate its ability to function as an affinity tag in a yeast expression system. We also examined the effects of glycosylation on the secreted CBM3-tag. The secreted wild-type CBM3-EGFP was glycosylated; however, this had little influence on the adsorption of the fusion protein to the regenerated amorphous cellulose (RAC; maximum adsorption capacity of 319 mg/g). Two CBM3-EGFP mutants lacking glycosylation sites were also constructed. The three CBM3-EGFPs expressed in P. pastoris and the CBM3-EGFP expressed in Escherichia coli all had similar RAC adsorption capacity. To construct a tag-free recombinant protein purification system based on CBM3, a CBM3-intein-EGFP fusion protein was expressed in P. pastoris. This fusion protein was stably expressed and the self-cleavage of intein was efficiently induced by DTT or l-cysteine. In this study, we were able to purify the recombinant fusion protein with high efficiency using both intein and direct fusion-based strategies.






Similar content being viewed by others
References
Ahn J, Choi E, Lee H, Hwang S, Kim C, Jang H, Haam S, Jung J (2004) Enhanced secretion of Bacillus stearothermophilus L1 lipase in Saccharomyces cerevisiae by translational fusion to cellulose-binding domain. Appl Microbiol Biotechnol 64:833–839
Arnau J, Lauritzen C, Petersen GE, Pedersen J (2006) Current strategies for the use of affinity tags and tag removal for the purification of recombinant proteins. Protein Expr Purif 48(1):1–13
Babu KS, Antony A, Muthukumaran T, Meenakshisundaram S (2008) Construction of intein-mediated hGMCSF expression vector and its purification in Pichia pastoris. Protein Expres Purif 57(2):201–205. doi:10.1016/j.pep.2007.10.004
Boraston AB, McLean BW, Guarna MM, Amandaron-Akow E, Kilburn DG (2001a) A family 2a carbohydrate-binding module suitable as an affinity tag for proteins produced in Pichia pastoris. Protein Expres Purif 21(3):417–423
Boraston AB, Warren RAJ, Kilburn DG (2001b) Glycosylation by Pichia pastoris decreases the affinity of a family 2a carbohydrate-binding module from Cellulomonas fimi: a functional and mutational analysis. Biochem J 358:423–430
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chong S, Xu MQ (1997) Protein splicing of the Saccharomyces cerevisiae VMA intein without the endonuclease motifs. J Biol Chem 272(25):15587–15590
Chong S, Shao Y, Paulus H, Benner J, Perler FB, Xu MQ (1996) Protein splicing involving the Saccharomyces cerevisiae VMA intein. The steps in the splicing pathway, side reactions leading to protein cleavage, and establishment of an in vitro splicing system. J Biol Chem 271(36):22159–22168
Elleuche S, Poggeler S (2010) Inteins, valuable genetic elements in molecular biology and biotechnology. Appl Microbiol Biot 87(2):479–489. doi:10.1007/s00253-010-2628-x
Esposito D, Chatterjee DK (2006) Enhancement of soluble protein expression through the use of fusion tags. Curr Opin Biotechnol 17(4):353–358
Fong BA, Wu WY, Wood DW (2010) The potential role of self-cleaving purification tags in commercial-scale processes. Trends Biotechnol 28(5):272–279. doi:10.1016/j.tibtech.2010.02.003
Gong F, Sheng J, Chi ZM, Li J (2007) Inulinase production by a marine yeast Pichia guilliermondii and inulin hydrolysis by the crude inulinase. J Ind Microbiol Biotechnol 34(3):179–185. doi:10.1007/s10295-006-0184-2
Guerreiro CIPD, Fontes CMGA, Gama M, Domingues L (2008) Escherichia coli expression and purification of four antimicrobial peptides fused to a family 3 carbohydrate-binding module (CBM) from Clostridium thermocellum. Protein Expres Purif 59(1):161–168. doi:10.1016/j.pep.2008.01.018
Hartley JL (2006) Cloning technologies for protein expression and purification. Curr Opin Biotechnol 17(4):359–366
Hong J, Tamaki H, Kumagai H (2007a) Cloning and functional expression of thermostable beta-glucosidase gene from Thermoascus aurantiacus. Appl Microbiol Biot 73(6):1331–1339
Hong J, Ye XH, Zhang YHP (2007b) Quantitative determination of cellulose accessibility to cellulase based on adsorption of a nonhydrolytic fusion protein containing CBM and GFP with its applications. Langmuir 23(25):12535–12540
Hong J, Wang YR, Ye XH, Zhang YHP (2008a) Simple protein purification through affinity adsorption on regenerated amorphous cellulose followed by intein self-cleavage. J Chromatogr A 1194(2):150–154
Hong J, Ye XH, Wang YR, Zhang YHP (2008b) Bioseparation of recombinant cellulose-bindning module-proteins by affinity adsorption on an ultra-high-capacity cellulosic adsorbent. Anal Chim Acta 621(2):193–199
Kavoosi M, Meijer J, Kwan E, Creagh AL, Kilburn DG, Haynes CA (2004) Inexpensive one-step purification of polypeptides expressed in Escherichia coli as fusions with the family 9 carbohydrate-binding module of xylanase 10A from T. maritima. J Chromatogr B 807:87–94
Laemmli UK (1970) Cleavage of structure proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Levy I, Shoseyov O (2002) Cellulose-binding domains biotechnological applications. Biotechnol Adv 20(3–4):191–213
Levy I, Shoseyov O (2004) Cross bridging proteins in nature and their utilization in bio- and nanotechnology. Curr Protein Pept Sc 5(1):33–49
Miller GO (1959) Use of dinitrosaiicyiic acid reagent for determination of reducing sugar. Anal Biochem 31:426–428
Murashima K, Kosugi A, Doi RH (2003) Solubilization of cellulosomal cellulases by fusion with cellulose-binding domain of noncellulosomal cellulase engd from Clostridium cellulovorans. Proteins 50(4):620–628
Przybycien TM, Pujar NS, Steele LM (2004) Alternative bioseparation operations: life beyond packed-bed chromatography. Curr Opin Biotechnol 15:469–478
Ramirez C, Fung J, Miller RC, Antony R, Warren J, Kilburn DG (1993) A bifunctional affinity linker to couple antibodies to cellulose. Nat Biotech 11(12):1570–1573
Shoseyov O, Shani Z, Levy I (2006) Carbohydrate binding modules: biochemical properties and novel applications. Microbiol Mol Biol R 70(2):283–295
Tomme P, Boraston AB, McLean B, Kormos JM, Creagh AL, Sturch K, Gilkes NR, Haynes CA, Warren RA, Kilburn DG (1998) Characterization and affinity applications of cellulose-binding domains. J Chromatogr B Biomed Sci Appl 715:283–296
Tormo J, Lamed R, Chirino AJ, Morag E, Bayer EA, Shoham Y, Steitz TA (1996) Crystal structure of a bacterial family-III cellulose-binding domain: a general mechanism for attachment to cellulose. EMBO J 15(21):5739–5751
Zhao Z, Lu W, Dun B, Jin D, Ping S, Zhang W, Chen M, Xu MQ, Lin M (2008) Purification of green fluorescent protein using a two-intein system. Appl Microbiol Biot 77(5):1175–1180. doi:10.1007/s00253-007-1233-0
Zhou X, Cai S, Hong A, You Q, Yu P, Sheng N, Srivannavit O, Muranjan S, Rouillard JM, Xia Y, Zhang X, Xiang Q, Ganesh R, Zhu Q, Matejko A, Gulari E, Gao X (2004) Microfluidic PicoArray synthesis of oligodeoxynucleotides and simultaneous assembling of multiple DNA sequences. Nucleic Acids Res 32(18):5409–5417. doi:10.1093/nar/gkh879
Acknowledgments
This work was supported by a grant-in-aid from the Anhui Provincial Natural Science Foundation (grant no. 090413075), the State Education Ministry and Specialized Research Fund for the Doctoral Program of Higher Education of China (grant no. 20093402120027), the National Basic Research Program of China (grant no. 2011CBA00801), and the Fundamental Research Funds for the Central Universities (grant no. WK2070000007). The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 49 kb)
Rights and permissions
About this article
Cite this article
Wan, W., Wang, D., Gao, X. et al. Expression of family 3 cellulose-binding module (CBM3) as an affinity tag for recombinant proteins in yeast. Appl Microbiol Biotechnol 91, 789–798 (2011). https://doi.org/10.1007/s00253-011-3373-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-011-3373-5