Abstract
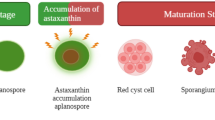
The unicellular green alga Chlorella zofingiensis has been proposed as a promising producer of natural astaxanthin, a commercially important ketocarotenoid. But the genetic toolbox for this alga is not available. In the present study, an efficient transformation system was established for C. zofingiensis. The transformation system utilized a modified norflurazon-resistant phytoene desaturase (PDS-L516F, with an leucine-phenylalanine change at position 516) as the selectable marker. Three promoters from endogenous PDS, nitrate reductase (NIT), and ribulose-1,5-bisphosphate carboxylase/oxygenase small subunit (RBCS) genes were tested, with the RBCS promoter demonstrating the highest transformation efficiency. Inclusion of the first intron of the PDS gene further enhanced the efficiency by 91 %. Both particle bombardment and electroporation methods were examined, and the latter gave a fourfold higher transformation efficiency. The introduction of PDS-L516F, which exhibited a 33 % higher desaturation activity than the unaltered enzyme, enabled C. zofingiensis to produce 32.1 % more total carotenoids (TCs) and 54.1 % more astaxanthin. The enhanced accumulation of astaxanthin in transformants was revealed to be related to the increase in the transcripts of PDS, β-carotenoid ketolase (BKT), and hydroxylase (CHYb) genes. Our study clearly shows that the modified PDS gene is a dominant selectable marker for the transformation of C. zofingiensis and possibly for the genetic engineering of the carotenoid biosynthetic pathway. In addition, the engineered C. zofingiensis might serve as an improved source of natural astaxanthin.







Similar content being viewed by others
References
Bryksin AV, Matsumura I (2010) Overlap extension PCR cloning: a simple and reliable way to create recombinant plasmids. Biotechniques 48:463–465
Cerutti H, Johnson AM, Gillham NW, Boynton JE (1997) Epigenetic silencing of a foreign gene in nuclear transformants of Chlamydomonas. Plant Cell 9:925–945
Chamovitz D, Sandmann G, Hirschberg J (1993) Molecular and biochemical-characterization of herbicide-resistant mutants of cyanobacteria reveals that phytoene desaturation is a rate-limiting step in carotenoid biosynthesis. J Biol Chem 268:17348–17353
Chow KC, Tung WL (1999) Electrotransformation of Chlorella vulgaris. Plant Cell Rep 18:778–780
Dawson HN, Burlingame R, Cannons AC (1997) Stable transformation of Chlorella: rescue of nitrate reductase-deficient mutants with the nitrate reductase gene. Curr Microbiol 35:356–362
Del Campo JA, Rodriguez H, Moreno J, Vargas MA, Rivas J, Guerrero MG (2004) Accumulation of astaxanthin and lutein in Chlorella zofingiensis (Chlorophyta). Appl Microbiol Biotechnol 64:848–854
Fraser PD, Bramley PM (2004) The biosynthesis and nutritional uses of carotenoids. Prog Lipid Res 43:228–265
Guerin M, Huntley ME, Olaizola M (2003) Haematococcus astaxanthin: applications for human health and nutrition. Trends Biotechnol 21:210–216
Hawkins RL, Nakamura M (1999) Expression of human growth hormone by the eukaryotic alga, Chlorella. Curr Microbiol 38:335–341
Huang JC, Liu J, Li YT, Chen F (2008) Isolation and characterization of the phytoene desaturase gene as a potential selective marker for genetic engineering of the astaxanthin-producing green alga Chlorella zofingiensis (Chlorophyta). J Phycol 44:684–690
Huang J, Zhong Y, Sandmann G, Liu J, Chen F (2012) Cloning and selection of carotenoid ketolase genes for the engineering of high-yield astaxanthin in plants. Planta 236:691–699
Huang J, Zhong Y, Liu J, Sandmann G, Chen F (2013) Metabolic engineering of tomato for high-yield production of astaxanthin. Metab Eng 17:59–67
Ip PF, Chen F (2005) Production of astaxanthin by the green microalga Chlorella zofingiensis in the dark. Process Biochem 40:733–738
Ip PF, Wong KH, Chen F (2004) Enhanced production of astaxanthin by the green microalga Chlorella zofingiensis in mixotrophic culture. Process Biochem 39:1761–1766
Jayaraj J, Devlin R, Punja Z (2008) Metabolic engineering of novel ketocarotenoid production in carrot plants. Transgenic Res 17:489–501
Kilian O, Benemann CSE, Niyogi KK, Vick B (2011) High-efficiency homologous recombination in the oil-producing alga Nannochloropsis sp. Proc Natl Acad Sci U S A 108:21265–21269
Kim DH, Kim YT, Cho JJ, Bae JH, Hur SB, Hwang I, Choi TJ (2002) Stable integration and functional expression of flounder growth hormone gene in transformed microalga, Chlorella ellipsoidea. Mar Biotechnol 4:63–73
Kovar JL, Zhang J, Funke RP, Weeks DP (2002) Molecular analysis of the acetolactate synthase gene of Chlamydomonas reinhardtii and development of a genetically engineered gene as a dominant selectable marker for genetic transformation. Plant J 29:109–117
Linden H (1999) Carotenoid hydroxylase from Haematococcus pluvialis: cDNA sequence, regulation and functional complementation. Biochim Biophys Acta 1446:203–212
Liu J, Huang J, Fan KW, Jiang Y, Zhong Y, Sun Z, Chen F (2010a) Production potential of Chlorella zofingienesis as a feedstock for biodiesel. Bioresour Technol 101:8658–8663
Liu J, Zhong Y, Sun Z, Huang J, Sandmann G, Chen F (2010b) One amino acid substitution in phytoene desaturase makes Chlorella zofingiensis resistant to norflurazon and enhances the biosynthesis of astaxanthin. Planta 232:61–67
Liu J, Huang J, Sun Z, Zhong Y, Jiang Y, Chen F (2011) Differential lipid and fatty acid profiles of photoautotrophic and heterotrophic Chlorella zofingiensis: assessment of algal oils for biodiesel production. Bioresour Technol 102:106–110
Liu J, Huang J, Jiang Y, Chen F (2012a) Molasses-based growth and production of oil and astaxanthin by Chlorella zofingiensis. Bioresour Technol 107:393–398
Liu J, Sun Z, Zhong Y, Huang J, Hu Q, Chen F (2012b) Stearoyl-acyl carrier protein desaturase gene from the oleaginous microalga Chlorella zofingiensis: cloning, characterization and transcriptional analysis. Planta 236:1665–1676
Liu J, Gerken H, Huang J, Chen F (2013a) Engineering of an endogenous phytoene desaturase gene as a dominant selectable marker for Chlamydomonas reinhardtii transformation and enhanced biosynthesis of carotenoids. Process Biochem 48:788–795
Liu J, Sun Z, Zhong Y, Gerken H, Huang J, Chen F (2013b) Utilization of cane molasses towards cost-saving astaxanthin production by a Chlorella zofingiensis mutant. J Appl Phycol 25:1447–1456
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCTmethod. Methods 25:402–408
Lumbreras V, Stevens DR, Purton S (1998) Efficient foreign gene expression in Chlamydomonas reinhardtii mediated by an endogenous intron. Plant J 14:441–447
Mann V, Harker M, Pecker I, Hirschberg J (2000) Metabolic engineering of astaxanthin production in tobacco flowers. Nat Biotechnol 18:888–892
Misawa N, Yamano S, Linden H, Defelipe MR, Lucas M, Ikenaga H, Sandmann G (1993) Functional expression of the Erwinia uredovora carotenoid biosynthesis gene crtI in transgenic plants showing an increase of beta-carotene biosynthesis activity and resistance to the beaching herbicide norflurazon. Plant J 4:833–840
Morris WL, Ducreux LJM, Fraser PD, Millam S, Taylor MA (2006) Engineering ketocarotenoid biosynthesis in potato tubers. Metab Eng 8:253–263
Niu YF, Zhang MH, Xie WH, Li JN, Gao YF, Yang WD, Liu JS, Li HY (2011) A new inducible expression system in a transformed green alga, Chlorella vulgaris. Genet Mol Res 10: 3427–3434
Ohresser M, Matagne RF, Loppes R (1997) Expression of the arylsulphatase reporter gene under the control of the nit1 promoter in Chlamydomonas reinhardtii. Curr Genet 31:264–271
Poulsen N, Kroger N (2005) A new molecular tool for transgenic diatoms - Control of mRNA and protein biosynthesis by an inducible promoter-terminator cassette. FEBS J 272:3413–3423
Radakovits R, Eduafo PM, Posewitz MC (2011) Genetic engineering of fatty acid chain length in Phaeodactylum tricornutum. Metab Eng 13:89–95
Ralley L, Enfissi EMA, Misawa N, Schuch W, Bramley PM, Fraser PD (2004) Metabolic engineering of ketocarotenoid formation in higher plants. Plant J 39:477–486
Randolph-Anderson BL, Sato R, Johnson AM, Harris EH, Hauser CR, Oeda K, Ishige F, Nishio S, Gillham NW, Boynton JE (1998) Isolation and characterization of a mutant protoporphyrinogen oxidase gene from Chlamydomonas reinhardtii conferring resistance to porphyric herbicides. Plant Mol Biol 38:839–859
Romer S, Fraser PD, Kiano JW, Shipton CA, Misawa N, Schuch W, Bramley PM (2000) Elevation of the provitamin A content of transgenic tomato plants. Nat Biotechnol 18:666–669
Schmidt I, Schewe H, Gassel S, Jin C, Buckingham J, Hümbelin M, Sandmann G, Schrader J (2011) Biotechnological production of astaxanthin with Phaffia rhodozyma/Xanthophyllomyces dendrorhous. Appl Microbiol Biotechnol 89:555–571
Shewmaker CK, Sheehy JA, Daley M, Colburn S, Ke DY (1999) Seed-specific overexpression of phytoene synthase: increase in carotenoids and other metabolic effects. Plant J 20:401–412
Steinbrenner J, Sandmann G (2006) Transformation of the green alga Haematococcus pluvialis with a phytoene desaturase for accelerated astaxanthin biosynthesis. Appl Environ Microbiol 72:7477–7484
Stewart CN, Via LE (1993) A rapid CTAB DNA isolation technique useful for rapid fingerprinting and other PCR applications. Biotechniques 14:748–751
Sun N, Wang Y, Li Y-T, Huang J-C, Chen F (2008) Sugar-based growth, astaxanthin accumulation and carotenogenic transcription of heterotrophic Chlorella zofingiensis (Chlorophyta). Process Biochem 43:1288–1292
Zhong Y-J, Huang J-C, Liu J, Li Y, Jiang Y, Xu Z-F, Sandmann G, Chen F (2011) Functional characterization of various algal carotenoid ketolases reveals that ketolating zeaxanthin efficiently is essential for high production of astaxanthin in transgenic Arabidopsis. J Exp Bot 62:3659–3669
Acknowledgments
This study was partially supported by a grant from the 985 Project of Peking University, by the State Oceanic Administration of China and by the National Research Foundation of Singapore.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 25 kb)
Rights and permissions
About this article
Cite this article
Liu, J., Sun, Z., Gerken, H. et al. Genetic engineering of the green alga Chlorella zofingiensis: a modified norflurazon-resistant phytoene desaturase gene as a dominant selectable marker. Appl Microbiol Biotechnol 98, 5069–5079 (2014). https://doi.org/10.1007/s00253-014-5593-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-014-5593-y