Abstract
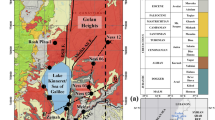
Deuterium, δ 18O, major ions and dissolved silica in groundwater from semi-arid Mayo-Tsanaga river basin in the Far North Province, Cameroon were used to trace hydrogeochemical processes that control their concentrations and to explore for usability of the water. Electrical conductivity ranges from 57–2,581 μs/cm with alternating low and high values along the hydraulic gradient. Waters from piedmont alluvium show low concentrations in major cations, which peak in Mg within basalt, Na within plain alluvium, and Ca within basalt and the sandy Limani-Yagoua ridge. The initial dominant groundwater composition is CaHCO3, which did not evolve within the basalt and piedmont alluvium, but evolved to NaHCO3 in the granite and plain alluvium. The main processes controlling the major ions composition include the following: (1) dissolution of silicates and fluorite; (2) precipitation of fluorite and carbonate; (3) cation exchange of Ca in water for Na in clay; (4) and anthropogenic activities. The δD and δ 18O ratios vary from −35 to 0.7 and −5.3 to 1.1‰, respectively. The lowest and highest isotope ratios are observed in groundwater within the downstream sandy Limani-Yagoua ridge and the upstream graintes respectively. Variation in isotope ratios depends on altitude effect of −0.48‰ per 100 m between 600 and 850 m asl, and on evaporation, which had insignificant effect on the water salinity. Seventy percent of the groundwater shows poor drinking quality and 90% is suitable for irrigation.







Similar content being viewed by others
References
Allison GB, Hughes MW (1983) The use of natural tracers as indicators of soil-water movement in a temperate semi-arid region. J Hydrol 60:157–173
Appelo CAJ, Postma D (1993) Geochemistry, groundwater, and pollution. Balkema, Netherlands, p 536
Betah SM (1976) Compliment sur la monographie nationale de l’eau (aspect eaux souterraines) Diredtion des mines et de la geologie, Cameroon
Boutrais J (1984) Le milieux naturels et L’occupation du sol. In: Jean B (ed) Le Nord du Cameroon: des homes, une region, pp 63–100. Collection memoires 102. Editions de L’ORSTOM, Paris
Budyko MI (1951) On climate factors of runoff. Prob. Fiz. Geogr. 16 in Russian Canadian Council of Ministers of the Environment (2001a) Canadian water quality guidelines for the protection of aquatic life. CCME water quality index 1.0, Technical report. http://www.ccme.ca
Canadian Council of Ministers of the Environment (2001b) Canadian water quality guidelines for the protection of aquatic life. CCME water quality index 1.0, user’s manual. http://www.ccme.ca
Clarke I, Fritz P (1997) Environmental isotopes in hydrogeology. Lewis, Boca Raton
Coleman ML, Stephard TJ, Durnham JJ, Rouse JE, Moore GR (1982) Reduction of water with zinc for hydrogen isotope analysis. Anal Chem 54:993–995
Craig H (1961) Isotopic variations in meteoric waters. Science 133:1702–1703
Demlie M, Wohnlich S, Wisotzky F, Gizaw B (2007) Groundwater recharge, flow and hydrogeochemical evolution in a complex volcanic aquifer system, central Ethopia. Hydrogeol J 15:1169–1181
Deutsch WJ (1997) Groundwater geochemistry: fundamentals and applications to contamination. Lewis, New York, p 221
Dobrzynski D (2005) Silica origin and solubility in groundwater from the weathered zone of sedimentary rocks of the Intra-Sudetic basin, SW Poland. Acta Geol Pol 55(4):445–462
Edmunds WM, Carrillo-Rivera JJ, Cardona A (2002) Geochemical evolution of groundwater beneath Mexico city. J Hydrol 258:1–24
Epstein S, Mayeda T (1953) Variation of oxygen-18 content of waters from natural sources. Geochim Cosmochim Acta 4:213–224
Garrels RM, Mackenzie FT (1967) Origin of the chemical composition of some springs and lakes. In: Gould RF (ed) Equillibrium concepts in natural water systems. American Chemical society, Washington, DC, pp 222–242
Gat JR (1996) Oxygen and hydrogen isotopes in the hydrologic cycle. Annu Rev Earth Planet Sci 24:225–262
Gee GW, Hillel D (1988) Groundwater recharge in arid region: review and critique of estimation methods. Hydrol Proc 2:255–266
Giggenbach WF (1990) Water and gas chemistry of Lake Nyos and its bearing on the eruptive process. J Volcanol Geothermal Res 42:337–362
Goni IB (2006) Tracing stable isotope values from meteoric water to groundwater in the southwestern part of the Chad basin. Hydrogeol J 14:742–752
Hem JD (1991) Study and interpretation of the chemical characteristics of natural water, 3rd edn. Book 2254. Scientific, Jodhpur
Herczeg AL, Simpson HJ, Mazor E (1993) Transport of soluble salts in a large semiarid basin. River Murray, Australia. J Hydro 144:59–84
Hervieu J (1969) Le Quaternaire Du Nord-Cameroon Schema D’evolution Geomorphologique et Relations Avec La Pedogenese. Ca. ORSTOM. Ser Pedol, vol 8, No. 3
IAEA (2007). Atlas of isotope hydrology. Africa–Vienna. ISBN 978-92-0-1072707-8 ImesJL
IAEA (2007) Global network of isotopes in precipitation (GNIP) Database IAEA/WMO, Vienna, Austria, http://www.isohis.iaea.org. Cited 22 April 2008
IGNP (1965) Carte de L’Afrique Centrale au 1/50000. Republique Federale du Cameroon, feuille NC-33-XIV-3a, 3b, 4b, and 4c
ISI: 11624(1986) Guidelines for quality of irrigation water, Bureau of Indian Standards, New Delhi
Jalali M (2006) Chemical characteristics of groundwater in parts of mountainous region, Alvand, Hamadan, Iran. Environ Geol 51:433–446
Kortatsi BK, Tay CK, Anornu G, Hayford E, Darty GA (2008) Hydrogeochemical evolution of groundwater in the lower Offin basin. Ghana Environ Geol 53:1651–1662
Kusakabe M, Ohba T, Issa, Yoshida Y, Satake H, Ohizumi T, Evanns WC, Tanyileke G, Kling GW (2008) Evolution of CO2 in Lakes Monoun and Nyos, Cameroon, before and during controlled degassing. Geochem J 42:93–118
McEachern S (2003) Processes of montagnard ethnogenesis in the northern mandaras mountain, Cameroon. PhD thesis. 433p. University of Calgary, Canada. ISBN 0-9544730-1-9. Mandaras Publishing
Moller, Detler (1990) The Na/Cl ratio rainwater and sea salt chloride cycle. Tellus B 3:254–262
Molua E1, Lambi CM (2006) Climate hydrology and water resources in Cameroon. CEEPA
Nagaraju A, Surresh S, Killam K, Hudson-Edwards K (2006) Hydrogeochemistry of waters of Mangampeta Barite Mining Area, Cuddapach Basin, Andhra Pradesh, India. Turk J Environ Sci 30:203–219
Ngako V, Njonfang E, Aka FT, Affaton P, Nnange JM (2006) The north–south Paleozoic to quaternary trend of alkaline magmatism from Niger–Nigeria to Cameroon: complex interaction between hotspots and Precambrian faults. J Afr Earth Sci 45:241–256
Ngounou-Ngatcha B, Murdry J, Wakponou A, Ekodeck GE, Njitchoua R, Sarrot-Reynauld J (2001) The Limani-Yagoua mega sand-ridge, northern Cameroon, and its hydrological importance. J Afr Earth Sci 32(4):889–898
Ngounou-Ngatcha B, Murdry J, Sarrot-Reynauld J (2007) Groundwater recharge from rainfall in the southern border of Lake Chad in Cameroon. World Appl Sci J 2:125–131
Njitchoua R, Ngounou-Ngatcha B (1997) Hydrogeochemistry and environmental isotope investigations of the north Diamare plain northern Cameroon. J Afr Earth Sci 25(2):307–316
Njitchoua R, Aranyossy JF, Fontes JC, Michelot JL, Naah E, Zuppi GM (1995) Oxygen-18, deuterium et chlorures dans les precipitations a Garoua (Nord-Cameroon): implications meteorolgiques. CR Acad Sci Paris, t 321, serie IIa: 853–860
Nouvelot JF (1972) Hydrologie des Mayos du Nord Cameroun. Monographie de la Tsanaga. Raport terminal 119p. ORSTOM
Peronne Y, Dumort JC (1968) Carte geologigue de reconnaissance de La Republique Federale Du Cameroon. Feuille No: NC 33 NO E62. Maroua
Petrides B, Cartwright I, Weaver TR (2006) The evolution of groundwater in the Tyrell catchment, south-central Murray Basin, Victoria, Australia. Hydrogeol J 14:1522–1543
Piper AM (1944) A graphic procedure in the geochemical interpretation of water analyses. Am Geophys Union Trans 25:914–923
Satake H, Kita Y, Hayashi H, Murata M (2007) Geochemical investigation around the Mozumi-Sukenobe fault survey tunnel. Geodynamics of atotsugawa fault system. In: Ando M (ed) pp 1–26
Saxena VK, Ahmed S (2001) Dissolution of fluoride in groundwater: a water-rock interaction study. Environ Geol 40:1084–1087
Schoellar H (1977) Geochemistry of groundwater. Ch. 15 In: Groundwater studies—an international guideline for research and practice, UNESCO, Paris pp 1–18
Srinivasamoorthy K, Chindambaram S, Prasanna MV, Vasanthavihar M, Peter J, Anandhan P (2008) Identification of major sources controlling groundwater chemistry from a hard rock terrain-A case study from Mettur taluk, Salem district, Tamil Nadu, India. J Earth Syst Sci 117(1):49–58
Subba Rao N (2006) Seasonal variation of groundwater quality in a part of Gunter District, Andhra Pradesh, India. Environ Geol 49:413–429
Thomas JM, Welch AH, Preissler AM (1989) Geochemical evolution of groundwater in Smith Creek Valley—a hydrologically closed basin in central Nevada, USA. Appl Geochem 4:493–510
Tillement B (1972) Hydrogeologie du Nord—Cameroun. Rapport 6, 294p. Direction des Mines et de la Geologie, Yaounde, Cameroon
Tsujimura M, Abe Y, Tanaka T, Shimade J, Higuchi S, Yamanaka T, Davaa G, Oyunbaatar D (2007) Stable isotopic and geochemical characteristics of groundwater in Kherlin River Basin: a semi-arid region in Eastern Mongolia. J Hydrol 333:47–57
UNDP (1975) Recherche et exploitation pilote d`eaux souterraines dans le Nord Cameroun. Contrat 58/72. Rapport final, Annexe technique No. 1–4. Hydrogeo. Roma-Pisa, Italy
Villholth KG (2006) Groundwater assessment and management: implications and opportunities of globalization. Hydrogeol J 14:330–339
Wen X, Wu Y, Zhang Y, Liu F (2005) Hydrochemical characteristics and salinity of groundwater in the Ejina basin, north western China. Environ Geol 48:665–675
WHO (1989) Health guidelines for the use of wastewater in agriculture and aquaculture. Report of WHO scientific group-Technical report series 778, WHO, Geneva, 74 pp
Wilson M (1988) Geomorphology and archaeological visibility in the northern mandara mountains and mora plain (Cameroon): Preliminary results. In: Daniel B, Henri T (eds) Collection colloques et seminaries, Editions de L’ORSTOM, Paris pp 9–50
Zhu GF, Su YH, Feng Q (2008) The hydrochemical characteristics and evolution of groundwater and surface water in the Heihe River Basin, northwest China. Hydrogeol J 16:167–182
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
254_2008_1629_MOESM3_ESM.tif
Plots showing variation of Na (Fig. 3a), Ca (Fig. 3b), Mg (Fig. 3c) K (Fig. 3d), F (Fig. 3e) and dissolved silica (3f) in groundwater within different rock types in study area (TIFF 21 kb)
254_2008_1629_MOESM4_ESM.tif
Plots showing variation of Na (Fig. 3a), Ca (Fig. 3b), Mg (Fig. 3c) K (Fig. 3d), F (Fig. 3e) and dissolved silica (3f) in groundwater within different rock types in study area (TIFF 25 kb)
254_2008_1629_MOESM5_ESM.tif
Plots showing variation of Na (Fig. 3a), Ca (Fig. 3b), Mg (Fig. 3c) K (Fig. 3d), F (Fig. 3e) and dissolved silica (3f) in groundwater within different rock types in study area (TIFF 22 kb)
254_2008_1629_MOESM6_ESM.tif
Plots showing variation of Na (Fig. 3a), Ca (Fig. 3b), Mg (Fig. 3c) K (Fig. 3d), F (Fig. 3e) and dissolved silica (3f) in groundwater within different rock types in study area[INSERT CAPTION HERE] (TIFF 21 kb)
254_2008_1629_MOESM7_ESM.tif
Plots showing variation of Na (Fig. 3a), Ca (Fig. 3b), Mg (Fig. 3c) K (Fig. 3d), F (Fig. 3e) and dissolved silica (3f) in groundwater within different rock types in study area (TIFF 21 kb)
254_2008_1629_MOESM8_ESM.tif
Plots showing variation of Na (Fig. 3a), Ca (Fig. 3b), Mg (Fig. 3c) K (Fig. 3d), F (Fig. 3e) and dissolved silica (3f) in groundwater within different rock types in study area (TIFF 19 kb)
254_2008_1629_MOESM11_ESM.tif
Variation of d-excess in groundwater as a function of altitude. The clusters ‘A’, ‘B’, and ‘C’ in figure 7 are observed (TIFF 63 kb)
254_2008_1629_MOESM12_ESM.tif
b. Saturation index with respect to fluorite showing that group 1 groundwater were undersaturated, while group 2 were supersaturated thus posing dissolution threat with time (TIFF 22 kb)
254_2008_1629_MOESM13_ESM.tif
a. Na/Ca ratio (meq/L) against fluoride (meq/L), showing a positive correlation and clustering the groundwater into groups 1 and 2 (TIFF 21 kb)
254_2008_1629_MOESM14_ESM.tif
Plots of groundwater samples in (a) Na2O- Al2O3- SiO2- H2O and (b) CaO-Al2O3-SiO2-H2O systems at 25°C. Thermodynamic data used in constructing these diagrams are those given in Fritz (1976) (TIFF 24 kb)
254_2008_1629_MOESM15_ESM.tif
Plots of groundwater samples in (a) Na2O- Al2O3- SiO2- H2O and (b) CaO-Al2O3-SiO2-H2O systems at 25°C. Thermodynamic data used in constructing these diagrams are those given in Fritz (1976) (TIFF 27 kb)
Rights and permissions
About this article
Cite this article
Fantong, W.Y., Satake, H., Ayonghe, S.N. et al. Hydrogeochemical controls and usability of groundwater in the semi-arid Mayo Tsanaga River Basin: far north province, Cameroon. Environ Geol 58, 1281–1293 (2009). https://doi.org/10.1007/s00254-008-1629-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00254-008-1629-x